Original research |
Peer reviewed |
An observational study on tail biting in commercial grower-finisher barns
Spyridon K. Kritas, DVM, PhD; Robert B. Morrison, DVM, MBA, PhD
SKK: Clinic of Medicine, School of Veterinary Medicine, University of Thessaly, 43100 Karditsa, Greece; RBM: Department of Clinical and Population Sciences, College of Veterinary Medicine, University of Minnesota, St Paul, MN 55108. Corresponding author: Dr S.K. Kritas, Clinic of Medicine, School of Veterinary Medicine, University of Thessaly, 43100 Karditsa, Greece; Tel: 24410-66003; Fax: 24410-66055; E-mail: skritas@vet.uth.gr
Cite as: Kritas SK, Morrison RB. An observational study on tail biting in commercial grower-finisher barns. J Swine Health Prod. 2004;12(1):17-22.
Also available as a PDF.
Summary
Objectives: To describe the prevalence of bitten tails and determine risk factors at pig and pen level in barns with a history of high incidence of tail biting.
Methods: In two finishing barns, prevalence and severity of tail-biting lesions in docked pigs and association of bitten tails with gender were determined in a cross-sectional study. The associations with space allowance per pig, animal density, and barrow:gilt ratio per pen were examined. A matched case-control study determined the association between length of tail, severity of lesions on bitten tails, and chest girth (a surrogate measure of body weight).
Results: In a total of 1895 pigs (58% barrows, 42% gilts), the prevalence of bitten tails was 16.3% (barrows 21%; gilts 9.4%). The odds of being a bitten barrow was 2.6 times higher than being a bitten gilt (P < .001). Approximately 60% of affected pigs of both genders had severe lesions. Neither prevalence nor severity of lesions was associated with space allowance, animal density, or barrow:gilt ratio within pen. The prevalence of bitten barrows was positively correlated with the percentage of gilts in the pen (r = 0.56, P = .001). Severely bitten pigs had significantly smaller chest girth than less affected pigs. No association of the severity of lesions with length of the tail was observed.
Implications: Under the conditions of this study, the incidence of tail-biting lesions in barrows was more than twice that in gilts. Severely bitten pigs were smaller than pen mates. Sorting by gender may help reduce tail biting.
Keywords: swine, tail
biting, prevention
Search the AASV web site
for pages with similar keywords.
Received: October
12, 2002
Accepted: June
6, 2003
Tail biting is one of the most common problems in confinement herds, for which no satisfactory control or treatment program exists. In many cases, substantial costs are incurred due to secondary infection and deteriorating body condition of affected pigs. In an abattoir study, 8.5 to 9.2% of undocked pigs were tail bitten, and tail-bitten pigs had significantly more carcass damage compared to unbitten ones.1,2 Tail biting was determined as the primary cause of multi-site abscesses found in carcasses at slaughter.3 The cost of tail biting also includes increased labor for transferring bitten pigs to recovery pens and applying medical treatment.
Another consequence of tail biting is the negative effect on the health and welfare of bitten pigs.3.4 Pigs that become tail biters may also be negatively affected, as the frustration of living in a stressful environment may be the motivation for biting the tails of pen mates.5
Despite much research, the specific factors that precipitate tail biting are not fully understood. Imbalanced diet (particularly protein and minerals), discomfort (eg, excessive temperature variation, drafts, noise), overcrowding, noxious gases in the barn, and a high proportion of slatted flooring have all been associated with higher frequencies of tail biting.2,5-7 The length of the tail appears to be an important factor. Hunter et al1,2 reported that tail docking reduced the prevalence of tail biting from a range of 8.5 to 9.2% to a range of 2.4 to 3.1%. Other factors reported to reduce the probability of undocked pigs being tail bitten are light straw provision, use of natural ventilation or artificially controlled natural ventilation, mixed gender grouping, feeding of meal or liquid feed, the use of double or multi-space feeders,and low prevalence of diseases in the herd.2,7
Much of the information on tail biting has been derived from slaughterhouses and surveys. These approaches have some important benefits, such as the large number of cases, but they may not always accurately represent conditions in the barns of origin. Pigs with severely bitten tails are sometimes slaughtered locally and thus are not included in such studies.2 The purpose of this study was to describe the prevalence of bitten tails and determine risk factors at pig and pen levels in two barns where there had been a high incidence of tail biting.
Materials and methods
Housing
This study was conducted in two identical finishing barns of a large swine enterprise. The barns were located 50 m apart at the same site. Each barn had 16 pens on each side (32 pens total per barn). Pens were 3 m x 7.2 m (21.6 m2), with fully slatted floors. Each pen had one five-space feeder and two bowl-type drinkers. Four pens in each barn were initially reserved for sick pigs. Barns had automatically controlled natural ventilation, with curtains on both sides of the building. Six large circulating fans were distributed along the south (long) side of each building.
Management
At 3 to 5 days of age, all pigs were tail docked and male pigs were castrated. Pigs were weaned at approximately 21 days old, and at approximately 70 days old, they were transferred from the nurseries to the finishing barns, where they stayed until slaughter age. Pigs were allocated to finisher pens (except the sick pens), irrespective of gender, in groups of approximately 36 to 38. Feed was provided ad libitum.All in-all out flow was applied by site. All pigs originated from the daughter nucleus herd of the enterprise. Pigs were vaccinated for Salmonella cholerasuis and Lawsonia intracellularis 3 weeks after entering the finishing barns.
Cross-sectional study
A cross-sectional study was designed to determine the prevalence and severity of bitten tails within the barns, and association of bitten tails with gender. For this purpose, we recorded gender and individual tail score of all pigs in each pen in both barns on one occasion, 7 weeks after placement in finishing barns. A scale from 0 to 4 was used for tail scoring, with a score of 4 for the most severe lesions, as described in Table 1 and illustrated in Figures 1A through 1F.

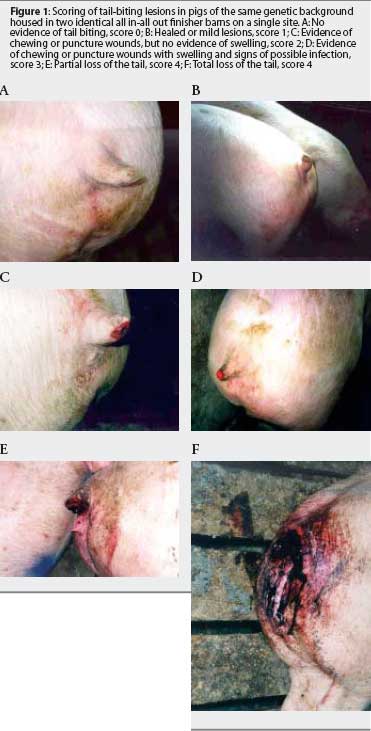
Chi square analysis was used to test the association between gender and having a bitten tail. The severity of lesions on bitten tails was recorded collectively per pen by calculating a pen biting score as follows: Pen biting score = ([1 x Number of pigs in pen scored 1] + [2 x Number of pigs in pen scored 2] + [3 x Number of pigs in pen scored 3] + [4 x Number of pigs in pen scored 4]) (Number of all pigs in pen). Multiple regression analysis was conducted,using pen as the unit, to investigate associations of prevalence of bitten tails and severity of lesions (pen biting score) with three factors: space allowance per pig, number of pigs in the pen (animal density), and the barrow:gilt ratio within each pen. All pens were included except six in Barn 1 and 10 in Barn 2, which served as sick pens at the time of measurement.
Case-control study
To determine the association of the severity of lesions of tail biting with body weight and the length of the tails, a matched case-control study was designed. In addition to tail score and tail length, chest girths of bitten pigs and of matched nonbitten control pigs were measured. The matched control was the first nonbitten pig in the pen observed approaching the drinker after each measurement of a bitten pig. In pens where the number of tail-bitten pigs exceeded the number of unbitten pigs, the necessary matched control pigs were selected from the adjoining pen using the same principle.
Tail length was measured as the distance from the base of the tail to the end of the tail, with the ventral side of the tail stretched onto a rigid ruler. Body weight was estimated by measuring chest girth.
To correlate chest girth (measured in cm) with body weight in pounds, a pilot study was conducted in 130 pigs of the same genetic background in a finishing barn of a different system. A tape measure was used to measure the perimeter of the chest just behind the front limbs in pigs weighing 45 to 125 kg. Pigs were then weighed individually on an electronic scale accurate to 1 lb, and weights were converted to kg. The correlation between girth and body weight was 0.94 (P < .001). Linear regression was performed to determine the following equation that was used to transform girth (cm) to body weight: Body weight (kg) = (1.76 x girth) - 94.
A total of 119 of 136 bitten pigs and 119 matched controls in Barn 1 and 94 of 173 bitten pigs and 94 matched controls in Barn 2 were included in the weight analysis. Some bitten pigs were not included because it was not possible to get a measurement. This was particularly true in Barn 2, as approximately 80 of the bitten pigs had been transferred to sick pens where they were not easily approached. Pigs in sick pens were housed in groups of 2 to 11 in Barn 1 and in groups of 1 to 20 in Barn 2.
All measurements and scoring throughout the study were performed by the same person. A main effects ANOVA was performed to determine the association between chest girth and tail length as dependent variables and individual tail score as independent variable, while controlling for the effects of barn and gender. Duncan's multiple range test was used to compare mean chest girth and tail length measurements among tail scores. The level of significance was set at P < .05.
All statistical analyses were performed using Statistica Software (Kernel Release 5.5, Edition 99; StatSoft Inc, Tulsa, Oklahoma).
Results
Cross-sectional study
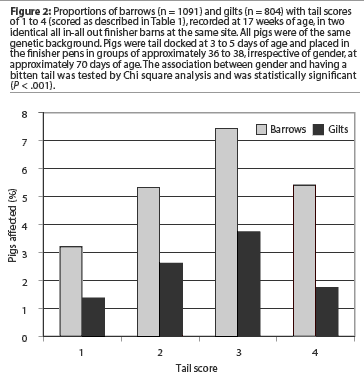
A total of 1895 pigs, of which 58% were barrows and 42% were gilts, were evaluated in both barns (Table 2). The average prevalence of bitten tails was 13.5% in Barn 1 and 19.4% in Barn 2 (P < .05), and the average prevalence on the site was 16.3%. The range of within-pen prevalence on the site was 0 to 66.7%. The prevalence of bitten tails was higher in barrows than in gilts (Table 2). The odds of being a bitten barrow was 2.6 times higher than being a bitten gilt (P < .05). Approximately 60% of the affected pigs of both genders had severely bitten tails, scored as 3 or 4. In both genders, the most frequently recorded score was 3 (Figure 2). Most bitten pigs were in good condition and alert at the time of measurement, but in some cases, severely bitten pigs (mainly scored as 4) were moribund, sometimes bleeding continuously from the tail (Figure 1F), or suffering from infection apparent associated with the traumatized tail (Figure 1E).

For the pen-basis analysis, a total of 1711 pigs, of which 993 (58%) were barrows and 718 (42%) were gilts, were counted in 48 pens (22 to 44 pigs per pen). Space allowance ranged from 0.49 to 0.98 m2 per pig, and the frequency distribution of pigs per pen was approximately normal (Figure 3). The average prevalence of bitten tails per pen was 12.9% for Barn 1, 16.5% for Barn 2, and 14.5% for the site. Neither prevalence nor severity of lesions on bitten tails (pen biting score) was associated with space allowance, animal density, or the ratio of barrows to gilts within each pen. The prevalence of bitten barrows was positively correlated with the percentage of gilts in the pen (r = 0.56, P = .001).

Case-control study
In Barn 1, average estimated body weight was 80.6 kg for barrows (n=163) and 77.4 kg for gilts (n=75). Average body weight was higher (P < .05) in Barn 2: 86.2 kg for barrows (n=102) and 82.2 kg for gilts (n=86).
Tail score was significantly associated with chest girth. Pigs with tail score 1 had larger girth measurements than pigs with score 0 (P < .05), and as tail score increased from 1 to 4, girth measurement decreased. Severely affected pigs (scores 3 and 4) had smaller girth measurements than pigs that were less severely affected or unbitten (P < .05) (Figure 4).

No clear association between severity of tail-biting lesions and length of the tail of the affected pigs was detected (P > .05).
Discussion
Tail biting is a complex behavioral condition that may cause substantial economic losses due to slow growth, carcass devaluation, and increased cost for labor and medication. A barren or uncomfortable environment (eg, uncontrolled air draughts) appears to provide the initial stimulus for this behavior,with subsequent attraction of pen mates to the blood.8,9 Enrichment of the environment appears to be critical in reducing tail biting.2,7,10-12
Several abattoir studies have recorded a high prevalence of tail-biting lesions in undocked pigs (7.4 to 11.6% on average) compared to 2.4 to 3.1% in pigs with docked tails.1,2,13,14 Furthermore, Hunter and co-workers1,2 compared farms with variable management practices with regard to the prevalence of tail biting, and observed that prevalence may be lower where there is optimal combination of such practices. Thus, for example, farms with natural or artificially controlled natural ventilation and light straw provision had a prevalence of 4.3% when tail docking was not performed, and 1.2% when tail docking was performed.1,2 In the present on-farm study, however, although all pigs were docked and several parameters reported to be beneficial were applied (eg, artificially controlled natural ventilation, meal-type feed, multi-space feeders), the prevalence of bitten tails was six times higher than that reported in docked pigs in the other studies. It may be that this difference is simply due to a "dilution effect" of severely affected barns mixed with less affected in the slaughterhouse studies. It could also be that some risk factors, such as the barren environment described earlier, predispose to others. Recently, Moinard and co-workers7 studied risk factors for tail biting among farms and described provision of light straw as the most important, followed by the level of postweaning mortality in the farm, type of floor, tail docking, type of feeder, and stocking density.
Prompted by previous observations that tail docking reduced tail biting,1,2 we thought that tail length would be an important risk factor in our study. Thus, we expected that the longer the tail, the higher the biting score would be. It is possible that tail length or the variability of tail lengths observed in this study (range 5 to 9.5 cm) was not large enough to affect tail biting or to permit clearer observations. Hunter and co-workers1,2 examined tails with more variable lengths, eg, docked (shorter than 10 cm) and undocked or tipped (longer than 10 cm). In contrast, others have observed that tail biting increases with tail docking.7,15 Apparently such different observations are largely due to the different sources of data (slaughterhouse versus farm, surveys versus studies, recording of simply biting versus multiple severe biting).
An interesting finding was that the prevalence of tail biting in barrows was twice that observed in gilts. This confirms observations in slaughterhouse surveys.1,13,14,16 It is not clear why this occurs. Our impression was that most gilts preferred face-to-face contact with strangers (eg, the person measuring chest girth), instead of turning to face the opposite direction, as was the case with most barrows. Wallgren and Lindahl17 suggested that during fighting, females confront face-to-face. If this occurs in a conflict situation, the barrow tails will be readily exposed to the biters (ie, when attacked by a gilt, a barrow will turn his back rather than face her, but another gilt will meet her face-to-face). Moreover, the finding that the prevalence of bitten barrows - but not that of bitten gilts - increased with increasing proportion of gilts in a pen may indicate that gilts are more prone to bite the tails of barrows in mixed pens. However, other researchers did not find evidence that a particular gender ratio encourages tail-biting behavior.18 These results suggest that it is better to separate genders and, if this is not possible, to avoid placing a much larger proportion of gilts in the pens. Studies should be designed for confirming behavior of genders, possibly also considering the genetic backgrounds of the animals. For example, in the herd in this study, the owner observed that tail biting constantly occurred in non-select breeding stock and multiplication barrows, but not in commercial pigs. Moinard and co-workers7 have suggested that genetics is one factor associated with tail biting.
Our finding that severely bitten pigs were significantly lighter (ie, had smaller chest girth measurements) than less severely bitten and unbitten pigs raises the question of whether low body weight is a cause or a result of severe tail biting. It has been suggested that larger pigs are usually better biting targets and that the initial biters are the smallest pigs of the group.19 It may be that the fast growing, less active, and less anxious pigs are generally less responsive to having other pigs nosing or chewing on their bodies, and only after they incur severe injuries do their growth rates decline. On the other hand, however, smaller pigs or sick pigs may be more reluctant to defend themselves against being bitten. It has been reported that tail biting is more prevalent in farms with high postweaning mortality and high incidence of respiratory disease.7 Prospective behavioral studies to observe development of tail biting activity from the time the pigs are placed in the finisher until close to slaughter age might clarify this issue.
The size of the group may affect tail biting via hierarchy establishment. An unstable social hierarchy may contribute to social stress and discomfort and may be observed to a greater degree in medium-sized groups (20 to 40 pigs) than in smaller or larger groups.20 It has also been suggested that reduction in space allowance is far more important in establishment of a social hierarchy than increase in group size.20 It has been observed that a stocking density greater than 110 kg per m2 increased the risk of tail biting by a factor of 2.7.7 In our study, both group size (22 to 44 pigs per pen) and stocking density (85 to 170 kg per m2) were not optimal and therefore constituted important risk factors for tail biting. However, neither group size nor space allowance appeared to affect prevalence and severity of tail-biting lesions, possibly because variability in these parameters was not wide enough to permit detection of differences.
The higher the prevalence of tail-biting lesions, particularly of severe lesions (9.7% in our study), the more individual treatments (and associated higher costs) and the more movements of pigs are necessary. Also, a marked delay in the growth of pigs and increased mortality may be expected. According to the farm personnel in the study herd, 60 to 70% of dead pigs had bitten tails.
Implications
- Under the conditions of this study, the incidence of tail-biting lesions was more than twice as high in barrows as in gilts.
- Severely bitten pigs were significantly smaller than their pen mates.
- Allocation of pigs to pens by gender may help to reduce tail biting.
Acknowledgements
We would like to thank Jeff Feder, DVM (Swine Vet Clinic, St Peter, Minnesota) and Bob Baarsch (Next Generation Pork, Spring Valley, Minnesota) for their help. This study was supported by funds from the Minnesota Pork Producers Association.
References
1. Hunter EJ, Jones TA, Guise HJ, Penny RH, Hoste S. Tail biting in pigs 1: the prevalence at six UK abattoirs and the relationship of tail biting with docking, sex and other carcass damage. Pig J. 1999;43:18-32.
2. Hunter EJ, Jones TA, Guise HJ, Penny RH, Hoste S. The relationship between tail biting in pigs, docking procedure and other management practices. Vet J. 2001;161:72-79.
3. Huey RJ. Incidence, location and interrelationships between the sites of abscesses recorded in pigs at a bacon factory in Northern Ireland. Vet Rec. 1996;138:511-514.
4. van Dijk WP, Klaver J, Verstegen MW. [Incidence of various disorders in fattening pigs and their effects on growth and carcass quality.] Tijdschr Diergeneeskd. 1984;109:539-548.
5. Schroder-Petersen DL, Simonsen HB. Tail biting in pigs. Vet J. 2001;162:196-210.
6. Gonyou HW, Lemay SP, Zhang Y. Effects of the environment on productivity and disease. In: Straw BE, Allaire SD, Mengeling WL, Taylor DJ, eds. Diseases of Swine. 8th ed. Ames, Iowa: Iowa State University Press. 1999;1017-1027.
7. Moinard C, Mendl M, Nicol CJ, Green LE. A case control study of on-farm risk factors for tail biting in pigs. Appl Anim Behav Sci. 2003;81:333-355.
8. Fraser D. Attraction to blood as a factor in tail-biting by pigs. Appl Anim Behav Sci. 1987;17:61-68.
9. Scheepens CJM, Hessing MJC, Laarakker E, Schouten WGP, Tielen MJM. Influences of intermittent daily draught on the behaviour of weaned pigs. Appl Anim Behav Sci. 1991;31:69-82.
10. Feddes JJR, Fraser D. Non-nutritive chewing by pigs: Implications for tail-biting and behavioral enrichment. Trans Am Soc Agric Eng. 1994;37:947-950.
11. Petersen V, Simonsen HB, Lawson LG. The effect of environmental stimulation on the development of behaviour in pigs. Appl Anim Behav Sci. 1995;45:215-224.
12. Beattie VE, Sneddon IA, Walker N, Weatherup RN. Environmental enrichment of intensive pig housing using spent mushroom compost. Anim Sci. 2001;72:35-42.
13. Penny RHC, Hill FWG. Observations of some conditions in pigs at the abattoir with particular reference to tail biting. Vet Rec. 1974;94:174-180.
14. Penny RHC, Walters JR, Tredget SJ. Tail biting in pigs: A sex frequency between boars and gilts. Vet Rec. 1981;108:35.
15. Chambers C, Powell L, Wilson E, Green LE. A postal survey of tail biting in pigs in south west England. Vet Rec. 1995;136:147-148.
16. Penny RHC, Hill FWG, Field JE, Plush JT. Tail biting in pigs: A possible sex incidence. Vet Rec. 1972;91:482-483.
17. Wallgren P, Lindahl E. The influence of tail biting on performance of fattening pigs. Acta Vet Scan. 1996;37:453-460.
18. Blackshaw JK. Some behavioural deviations in weaned domestic pigs: persistent inguinal nose thrusting, and tail and ear biting. Anim Prod. 1981;33:325-332.
19. Smith WJ, Penny RHC. Behavioral problems including vices and cannibalism. In: Leman AD, Straw BE, Glock RD, Mengeling WL, Penny RHC, Scholl E, eds. Diseases of Swine. 6th ed. Ames, Iowa: Iowa State University Press. 1986;762-772.
*20. Gonyou HW. The influences of group size on behavioral vices and cannibalism. Proc AD Leman Swine Conf. 1998;237-239.
*Non-refereed reference.
