Case report |
Peer reviewed |
A presumptive case of vomiting and wasting disease in a swine nucleus herd
Un presunto caso de enfermedad de vómito y desgaste en una granja núcleo porcina
Cas présumé de vomissements et de maladie du dépérissement chronique au sein d'un troupeau de reproduction de porcs
Janet E. Alsop, DVM
Canadian Food Inspection Agency, Woodstock, Ontario, Canada. Dr Janet E. Alsop, Canadian Food Inspection Agency, 685545 Highway 2, RR 5, Woodstock, Ontario, Canada N4S 7V9; Tel: 519-539-8505; Fax: 519-539-7694 ; E-mail: alsopj@inspection.gc.ca.
Cite as: Alsop JE. A presumptive case of vomiting and wasting disease in a swine nucleus herd. J Swine Health Prod. 2006;14(2):97-100.
Also available as a PDF.
SummaryAn outbreak of vomiting and wasting disease was presumptively diagnosed in a 650-sow genetic nucleus herd in January 2002. Clinical signs included inappetence, coughing, and pyrexia in nursing sows, and vomiting, huddling, and pyrexia in piglets. More than 500 weaned pigs were euthanized because of anorexia and wasting. No clinical signs were observed in gestating sows or in pigs in the finishing barns. The clinical diagnosis was presumptively confirmed by signs in piglets and weaned piglets. However, the cause of the outbreak was not determined, as the herd had been seropositive for hemagglutinating encephalomyelitis virus (HEV) prior to the onset of clinical signs. Since February 18, 2002, there have been no clinical signs of HEV in the herd. | ResumenUn brote de la enfermedad de vómito y desgaste fue presuntamente diagnosticado en una granja núcleo productora de genética de 650 hembras en enero de 2002. Los signos clínicos incluyeron inapetencia, tos, y pirexia en las hembras lactantes y vómito, amontonamiento, y pirexia en los lechones. Más de 500 lechones fueron sacrificados debido a la inapetencia y desgaste. No se observaron signos clínicos en las hembras gestantes o en los cerdos de la engorda. El diagnóstico clínico presuntamente se confirmó por los signos clínicos de los lechones lactantes y destetados. Sin embargo, la causa del brote no fue determinada ya que la piara era positiva al virus de la encefalitis hemoaglutinante (HEV, pos sus siglas en inglés) antes de la presentación de los signos clínicos. Desde febrero 18, 2002, no se han presentado signos clínicos de HEV en la piara. | ResuméEn janvier 2002, on a diagnostiqué une éclosion présumée de vomissements et de maladie du dépérissement chronique au sein d'un troupeau de reproduction comptant 650 truies. Parmi les signes cliniques, on notait l'inappétence, la toux, et la pyrexie chez les truies allaitantes ainsi que des vomissements, le pelotonnement, et la pyrexie chez les porcelets. Plus de 500 porcelets sevrés ont dû être euthanasiés en raison de l'anorexie et du dépérissement. Aucun signe clinique n'a été observé chez les truies en gestation et chez les porcs des granges de finition. Le diagnostic clinique aurait été confirmé par les signes présents chez les porcelets et les porcelets sevrés. Il n'a toutefois pas été possible de déterminer la cause de cette éclosion puisque le troupeau était séropositif pour l'encéphalomyélite porcine [hemagglutinating encephalomyelitis virus: HEV] avant l'émergence des signes cliniques. Aucun signe clinique du HEV n'a été décelé au sein du troupeau depuis le 18 février 2002. |
Keywords: swine, hemagglutinating
encephalomyelitis virus, vomiting and wasting disease
Search the AASV web site
for pages with similar keywords.
Received: January
11, 2005
Accepted: May
6, 2005
Hemagglutinating encephalomyelitis virus (HEV), a member of the Coronaviridae family, was originally identified in Canada in 1962,1 and has since been observed in pig-raising countries around the world.2 The pig is the natural host, although the virus has been adapted experimentally to replicate in laboratory rodents.2 The virus is endemic in many swine populations3,4 and may be isolated from the respiratory tracts of normal pigs.5 Infection is usually subclinical and the disease is of low economic importance. Only one serotype of HEV is known to exist. Sequencing data indicate that it has remained antigenically and genetically stable since its first isolation.6,7 No vaccine is available.
In enzootically infected herds, most pigs are protected by colostral antibodies for 4 to 18 weeks (median 10.5 weeks),8 and subsequently develop an active immunity. However, in naive herds, infection causes severe clinical signs that may be manifested as vomiting and wasting disease (VWD), a neurological syndrome, or, rarely, both.9 Both syndromes commonly occur in pigs less than 3 weeks of age, although older swine may occasionally vomit and have a brief period of inappetence, listlessness, and central nervous system (CNS) disturbance.2 Occurrence of only one syndrome in a particular outbreak is probably attributable to susceptibility of the infected pigs, exposure level, and strain differences in the virulence of the virus.10 In North America, the differential diagnoses for HEV infection include porcine reproductive and respiratory syndrome (PRRS), swine influenza, pseudorabies, paramyxovirus infection, porcine enterovirus infection, rabies, and transmissible gastroenteritis (TGE).
The primary sign in a VWD outbreak is repeated retching and vomiting in piglets, although there may be mild respiratory signs initially. Piglets huddle and are listless, and there may be a transient pyrexia.2 Young nursing piglets often become severely dehydrated and die. Older nursing or weaned piglets become cachectic due to inadequate feed intake and may persist in a wasting state for several weeks before dying. The abdomen may be distended due to gaseous distension of the stomach and intestines.11
The encephalomyelitic form may begin as a VWD outbreak.2 The first signs may include vomiting at 4 to 7 days of age, but this is rarely severe and pigs do not become dehydrated. Alternatively, piglets may show acute depression and huddling. Coughing and sneezing may be observed and the piglets rapidly lose weight.2 After 1 to 3 days, more severe clinical signs occur, including generalized muscle tremors, hyperesthesia, posterior paresis, and convulsions.2 Mortality may reach 100% in neonatal piglets. The clinical course in a herd is usually 2 to 3 weeks, coinciding with the time required for sows to develop immunity and pass this on to their offspring through colostral antibodies.2
Clinical report
In January 2002, sows and piglets in a recently established high-health, 650-sow production nucleus herd (purebred Landrace and Large White x Landrace) in southwestern Ontario began to exhibit anorexia, vomiting, fever, and coughing. The first piglets had been farrowed in October 2001.
The herd receives monthly veterinary visits. The sows are not routinely blood tested; serological testing is carried out only if there are clinical signs of disease. Each month, pigs in the finishing barn are serologically monitored for PRRS virus (PRRSV) (Idexx HerdChek ELISA; Idexx Laboratories, Westbrook, Maine), Mycoplasma hyopneumoniae (DAKO monoclonal blocking ELISA), Actinobacillus pleuropneumoniae (serotypes 1, 5a, and 5b; ELISA) and toxigenic Pasteurella multocida (ELISA). At the time of the outbreak, the sow herd was seronegative for M hyopneumoniae, Actinobacillus pleuropneumoniae(serotypes 1, 5a, and 5b), TGE virus (TGEV), porcine respiratory coronavirus (PRCV), toxigenic Pasteurella multocida, and swine influenza virus (SIV) H1N1, and seropositive for PRRSV (vaccine strain), Lawsonia intracellularis, Streptococcus suis, SIV H3N2, and porcine circovirus type 2 (PCV2). The breeding animals in the herd are vaccinated against PRRSV, parvovirus, six Leptospira serovars, Erysipelothrix rhusiopathiae, and Escherichia coli during each parity period. The sow barn is located in a wooded area 2 km from the nearest swine unit. Farrowing rooms are operated on an all in-all out basis. Piglets receive no vaccines, and are weaned at 14 to 17 days of age and transferred to a 2250-head offsite nursery located 14 km away. After approximately 8 weeks in the nursery, they are transferred to one of two 2000-head finishing barns located 18 km from the sow herd and approximately 12 km from other unrelated swine facilities.
Biosecurity is excellent. Replacement animals are housed in an isolation facility for 6 to 8 weeks and tested serologically (PRRS ELISA) before entering the main herd. Personnel entry is restricted: all staff and visitors must shower and change clothing before entering the facilities. Transport trailers delivering or collecting animals are washed, disinfected, and dried before arrival. Supplies and equipment are removed from exterior packaging, disinfected, or both before being brought into the barns.
No abnormalities were noted during a routine herd health visit on January 7, 2002. As barn staff had reported a mild, sporadic cough in a small percentage of the animals (before the outbreak of illness in the piglets), sera were collected from 14 gestating sows and submitted to the Animal Health Laboratory (AHL; University of Guelph, Guelph, Ontario, Canada) for testing for PRRSV, SIV strains H1N1 and H3N2 (hemagglutination inhibition; HI), and M hyopneumoniae. Sows were seronegative for SIV H1N1 and M hyopneumoniae, and had low positive titers for PRRSV and SIV H3N2.
On January 14, a power failure lasting 4 hours occurred, and the stand-by generator failed to start automatically. As a result, temperatures and relative humidity in the mechanically ventilated farrowing rooms were elevated. In the breeding and gestation area, room temperature remained within normal range because the curtains automatically dropped by 15 cm as part of the failsafe program. Following restoration of the power supply, temperature and humidity levels in the farrowing rooms returned to normal over the course of 6 to 8 hours.
On January 16, the nursing sows were not eating well. This was not considered significant, as poor feed quality had been associated with an average consumption of only 65% to 70% of the lactation feed intake target of 5 kg per sow per day. However, on January 21, there were more anorexic nursing sows than expected, and approximately 5% had a mild cough. A sample of the lactation ration was submitted to Agri-Food Laboratories (Guelph, Ontario, Canada) for mycotoxin testing, and the feed in the bins was replaced.
On January 24, vomiting was noticed in 10% to 15% of the piglets. During a herd health visit on January 28, the herd veterinarian observed clinical signs in most piglets, including fever (up to 43°C), shivering, huddling, hyperemic skin, and vomiting. Approximately 50% of the nursing sows were severely anorexic, and 15% to 20% were coughing and febrile, with rectal temperatures of 40°C to 41°C. Gestating sows appeared unaffected. On the same day, similar signs were observed in nursery pigs that had been weaned on January 24. Four weaned piglets exhibiting clinical signs were euthanized, and tissue samples and sera were submitted to AHL.
On the basis of the respiratory signs and anorexia in the sows and the vomiting and fever in the piglets, the differential diagnoses at this time included TGE, swine influenza, and PRRS. Lung tissue was negative for influenza virus type A by fluorescent antibody testing, and both lung and serum were negative for PRRSV by reverse transcriptase-polymerase chain reaction (PCR). Pooled lung samples were weakly positive for PCV2 by PCR. There was no significant bacterial growth in lung, liver, spleen, or kidney. Histologically, there was mild chronic interstitial pneumonia suggestive of viral infection, and renal tubular necrosis secondary to dehydration. There was no histologic evidence of infection with SIV or of bacterial pneumonia-septicemia. No virus was isolated from lung samples.
Breeding-stock sales from the nursery and finishing barns ceased until the etiology of the problem could be determined. Between January 21 and 28, approximately 50% of the affected sows and piglets were treated with procaine penicillin G (30 mg per kg, IM, once daily) and the remainder were injected with oxytetracycline HCl (10 mg per kg, IM, once daily), with no response to either treatment. On January 29, acetylsalicylic acid (ASA) was administered to anorexic sows in the drinking water, and their piglets were injected with flunixin meglumine (8.3 mg, IM). Within 24 hours, pyrexia had resolved in pigs and sows. Some sows were already agalactic and were weaned within 3 to 8 days of farrowing. Affected litters were supplemented with liquid milk replacer and creep feed.
On February 4, the herd veterinarian revisited the sow herd and nursery. Coughing in the nursing sows had resolved. The only affected litter in the newly farrowed piglets was in a room into which older affected piglets had been fostered onto newly farrowed sows: this was not the normal practice. Nursery pigs weaned after January 24 had received ASA in the drinking water between January 30 and February 2. Piglets were thin but bright and alert. Postweaning feed intake was much lower than normal.
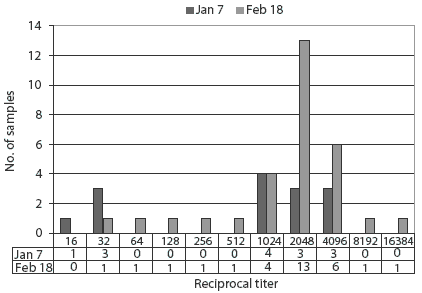
On February 18, 510 weaned piglets in the nursery were euthanized because of anorexia and irreversible wasting, and blood samples for convalescent titres were collected from 30 sows that had exhibited clinical signs. Sera collected on January 7 were used for acute titres. Because of the severe clinical signs in the weaned piglets, HEV infection was added to the list of differential diagnoses. Sows were seronegative for TGE-PRCV (differential ELISA), M hyopneumoniae, and SIV H1N1, and sero-positive for PRRSV and SIV H3N2. Reciprocal HI titers for HEV ranged from 16 to 4096 in the sows tested on January 7, with titers of 1024 or greater in 11 of the 14 sows tested (78.6%). In the February 18 samples, titers ranged from 32 to 16,384, with titers of 2048 or greater in 21 of the 30 sows tested (70%) (Figure 1). Statistical analysis (paired t test; STATISTIX 7.0; Analytical Software, Tallahassee, Florida) revealed no significant difference in sow HEV HI titres before and after the outbreak.
Figure 1: Reciprocal hemagglutination inhibition titers for hemagglutinating encephalomyelitis virus in sows in a nucleus herd in samples collected before and after an apparent outbreak of vomiting and wasting disease. Nursing sows showed anorexia and mild respiratory disease, and vomiting and wasting occurred in nursing and weaned pigs beginning approximately January 14. Samples were collected from 14 clinically normal sows on January 7 and 30 affected sows on February 18. There was no statistical difference (P = .35; paired t test) in the number of sows in each titer category before and after the outbreak of disease.
|
The lactation-feed sample tested negative for vomitoxin.
On the basis of clinical signs, the presumptive diagnosis was VWD caused by HEV. No further clinical signs have occurred in the herd. Clinical signs related to the outbreak were never observed in animals in the breeding-gestation barn on the same site as the farrowing barn or in the finishing barns. Breeding-stock sales resumed in March 2002.
Discussion
In this case, two confounding issues complicated the diagnosis of HEV. First, the initial clinical sign, anorexia in lactating sows, was consistent with the anorexia that had been occurring intermittently since farrowings had begun in October. Second, the mild cough heard in the nursing sows was attributed to the adverse environment caused by malfunction of the ventilation system. It was only when piglets also began to exhibit clinical signs that it became obvious that the problem was caused by an infectious disease rather than a nutritional or environmental problem.
Hemagglutinating encephalomyelitis virus is transmitted via nasal secretions7 and replicates in epithelial cells of the nasal mucosa, tonsil, lungs, and small intestine.12 After local replication, the virus spreads from the peripheral nervous system to the central nervous system.2 Vomiting associated with the disease is presumed to result from altered function of vagal and gastric intramural plexus neurons secondary to viral infection.11 Persistence of VWD is believed to be due to viral-induced neuronal death,10 and delayed stomach emptying probably plays an important role in development of wasting.2
Virus can be isolated from the tonsils and respiratory tract during the incubation period, which lasts for approximately 5 days.13 Isolation of HEV may be attempted on tonsil, brain, and lung samples from affected piglets. However, sensitivity is low unless samples are collected from acute cases within 1 or 2 days of the onset of clinical signs. It is suspected that hemagglutination inhibiting and seroneutralizing antibodies preclude virus isolation at later stages of the disease.13,14 In this case, the piglets sampled were no longer in the acute stage of the disease, and even if they had been infected with HEV, the virus might not have been recovered from the lungs.
Because of the widespread subclinical nature of infection, paired sera must be collected to demonstrate a fourfold increase in titer in convalescent samples. Acute sera must be collected as soon as possible after the onset of clinical signs, as high antibody titers develop rapidly. In this case, paired sera were not collected. The initial samples were collected randomly from gestating sows for routine monitoring, whereas the follow-up samples were collected specifically from sows that had been clinically affected during the outbreak. Although some sows tested in the second set of sera had HEV titers higher than those in the first set of samples, this could be explained by laboratory variation or small sample size. Since the clinical signs in the weaned piglets were consistent with HEV infection, a presumptive diagnosis was made.
Respiratory signs in sows related to an outbreak of VWD have not been reported in the literature, although Greig and Girard15 observed anorexia in sows in affected herds. However, coughing was also observed in sows as an initial sign of a VWD outbreak in another Ontario herd in 1986 (K. Richardson, written communication, 2005).
The positive PCV2 PCR on lung tissue from nursery pigs was not unexpected. The gilts in this herd had been purchased from a PCV2-positive herd, and many pig herds in Ontario are seropositive. Porcine circovirus type 2 has been implicated in postweaning multisystemic wasting syndrome (PMWS), in which some clinical signs, including weight loss, emaciation, and CNS disturbances, resemble those of HEV. The literature clearly supports enhanced disease in growing pigs concurrently infected with PCV2 and other pathogens,16-18 and PCV2 might have been a cofactor in this case. However, nursing piglets in this herd showed clinical signs, and as nursing piglets rarely are affected by PMWS,19 no additional tests for circovirus were performed.
The reason for the apparent outbreak of VWD has not been determined. Serological results from January 7 indicated that the sows were already HEV-seropositive prior to the outbreak. Since the breeding herd was very young at this stage (all gilts or parity-one animals), it may have included both seropositive and subclinically infected animals and naive animals, even though all had been purchased from the same source. Replacement gilts entered the farm on November 7, 2001, and it is possible that the animals might have been recently infected with a more virulent strain of HEV. However, as the incubation period of HEV is only 4 to 7 days,2 the timing of the outbreak is not likely to have been associated with new additions to the herd. Another possibility is that the virus was transmitted by fomites. In common with other coronaviruses, HEV is most stable at low temperatures, and in winter, the virus can survive for extended periods of time on boots, clothing, transport trailers, and other fomites. However, because of the excellent biosecurity on the farm, fomite transmission is unlikely. The third possibility is that the ventilation breakdown triggered the outbreak, and this may explain why the nursing sows, and not the gestating sows, exhibited clinical signs. Whether the environmental changes acted as a stressor and cofactor to induce clinical disease is difficult to prove or refute.
The duration of clinical signs (3 weeks) was consistent with that reported in the literature. Despite the severity of the problems, preweaning mortality did not exceed 12% during the outbreak. This differs from reports in the literature2 of significantly higher mortality, approaching 100% in affected litters. The excellent nursing care provided by barn personnel (eg, antipyretics and milk supplementation) contributed to the low mortality in the nursing piglets. However, due to the nature of the disease and its impact on gastric function, it was necessary to euthanize a large number of affected piglets in the postweaning period. Approximately half of these animals were potential breeding stock; therefore, the economic loss to the farm was significant.
Implications
- Hemagglutinating encephalomyelitis virus should be included as a differential diagnosis when piglets exhibit vomiting and pyrexia.
- When unusual clinical signs suggesting HEV infection occur in a swine population, diagnostic sampling should be initiated during the acute phase of the disease, as a definitive diagnosis cannot be made without paired serum samples.
References
1. Greig AS, Mitchell D, Corner AH, Bannister GL, Meads EB, Julian RJ. A hemagglutinating virus producing encephalomyelitis in baby pigs. Can J Comp Med. 1962;26:49-56.
2. Pensaert MB. Hemagglutinating encephalomyelitis virus. In: Straw BE, D'Allaire S, Mengeling WL, Taylor DJ, eds. Diseases of Swine. 8th ed. Ames, Iowa: Iowa State University Press; 1999:151-157.
3. Mengeling WL. Incidence of antibody for hemagglutinating encephalomyelitis virus in serums from swine in the United States. Am J Vet Res. 1975;36:821-823.
4. Hirano N, Suzuki Y, Haga S. Pigs with highly prevalent antibodies to human coronavirus and swine hemagglutinating encephalomyelitis virus in the Tohoku district of Japan. Epidemiol Infect. 1999;122:545-551.
5. Huxtable CR. The nervous system. In: Jubb KVF, Kennedy PC, Palmer N. Pathology of Domestic Animals. 4th ed. New York: Academic Press; 1993:267-439.
6. Paul PS, Halbur P, Janke B, Joo H, Nawagitgul P, Singh J, Sorden S. Exogenous porcine viruses. Curr Top Microbiol Immunol. 2003;278:125-183.
7. Sasseville AM, Gelinas AM, Sawyer S, Boutin M, Dea S. Biological and molecular characteristics of an HEV isolate associated with recent acute outbreaks of encephalomyelitis in Quebec pig farms. Adv Exp Med Biol. 2001;494:57-62.
8. Paul PS, Mengeling WL. Persistence of passively acquired antibodies to hemagglutinating encephalomyelitis virus infection in swine. Am J Vet Res. 1984;45:932-934.
9. Sims LD. The nervous system. In: Sims LD, Glastonbury JRW, eds. Pathology of the Pig: A Diagnostic Guide. Barton, Australia: The Pig Research and Development Corporation; 1996:287-312.
10. Mengeling WL, Cutlip RC. Pathogenicity of field isolants of hemagglutinating encephalomyelitis virus for neonatal pigs. JAVMA. 1976;168:236-239.
11. Storts RW, Montgomery DL. The nervous system. In: McGavin MD, Carlton WW, Zachary JF. Thomson's Special Veterinary Pathology. 3rd ed. St Louis, Missouri: Mosby; 2001:381-459.
12. Andries K, Pensaert MB. Immunofluorescence studies on the pathogenesis of hemagglutinating encephalomyelitis virus infection in pigs after oronasal inoculation. Am J Vet Res. 1980;41:1372-1378.
13. Andries K, Pensaert M, Callebaut P. Pathogenicity of hemagglutinating encephalomyelitis (vomiting and wasting disease) virus of pigs, using different routes of inoculation. Zentralbl Veterinarmed B. 1978;25:461-468.
14. Cutlip RC, Mengeling WL. Lesions induced by hemagglutinating encephalomyelitis virus strain 67N in pigs. Am J Vet Res. 1972;33:2003-2009.
15. Greig AS, Girard A. Serological comparison of hemagglutinating encephalomyelitis viruses isolated from different outbreaks. Can J Comp Med. 1969;33:25-28.
*16. Segals J, Domingo M. Diagnosis, prevention and control of PCV2-associated diseases. Proc AASV. Orlando, Florida. 2003;371-376.
*17. Halbur PG, Opriessnig T, Yu S, Thacker E. PMWS: Laboratory experiences. Proc Iowa State Univ Swine Disease Conf Swine Pract. 2002;76-82.
18. Harms PA, Halbur PG, Sorden SD. Three cases of porcine respiratory disease complex associated with porcine circovirus type 2 infection. J Swine Health Prod. 2002;10:27-30.
19. Lukert PD. Porcine circovirus. In: Straw BE, D'Allaire S, Mengeling WL, Taylor, DJ, eds. Diseases of Swine. 8th ed. Ames, Iowa: Iowa State University Press; 1999:119-124.
* Non-refereed references.