Original research |
Peer reviewed |
How effective is Mycoplasma hyopneumoniae vaccination in pigs less than three weeks of age?
Cuán tan efectiva es la vacunación de Mycoplasma hyopneumoniae en cerdos de menos de tres semanas de edad?
Quelle est l'efficacité de la vaccination contre Mycoplasma hyopneumoniae chez les porcs de moins de trois semaines d'âge?
Patricia K. Holyoake, PhD; Anthony P. L. Callinan, PhD
PKH: University of Sydney, Camden, New South Wales, 2570, Australia. APLC: Bendigo Scientific Data Analysts, Bendigo, Victoria, Australia. Dr Callinan has served as a paid consultant to Fort Dodge Australia Pty Ltd concerning statistical analysis of trials unrelated to the one reported here. Corresponding author: Dr Patricia K. Holyoake, 425 Werombi Road, University of Sydney, Camden, New South Wales, 2570, Australia; Tel: +61 2 93511617; Fax: +61 2 93511693; E-mail: trishh@camden.usyd.edu.au.
Cite as: Holyoake PK, Callinan APL. How effective is Mycoplasma hyopneumoniae vaccination in pigs less than three weeks of age? J Swine Health Prod. 2006;14(4):189-195.
Also available as a PDF.
SummaryObjective: To assess the cost effectiveness of administering a two-dose Mycoplasma hyopneumoniae (MH) vaccine to pigs before weaning at 13 to 17 days of age. Materials and methods: Two hundred litters of piglets on a commercial breeder farm (Site One) were randomly allocated to two treatment groups: nonvaccinated or vaccinated with MH vaccine at 4 days of age and at weaning (off-label use of the vaccine). At weaning, treatment groups were commingled in two nursery barns on Site Two, segregated by gender (875 castrates, 911 females). At 8 weeks, vaccinates and nonvaccinates were randomly allocated to either bedded shelters or concrete-floored barns at Site Three. Pigs were processed at 26 weeks of age and carcass weights, P2 backfat, and lung lesions for individuals recorded. Results: Carcass weight (95.91 kg) and P2 backfat (15.35 mm) in vaccinated pigs were greater (P < .05) than in nonvaccinated pigs (92.47 kg and 14.13 mm, respectively). Average lung score and incidence of chronic lung lesions were lower in vaccinates. Mortality rate, carcass weight variation, and incidence of pleurisy, acute lung lesions, and pleuropneumonia at processing did not differ between treatment groups. Profitability was greater in vaccinates by AU$5.14 per pig. Implications: Under the conditions of this study, vaccinating pigs for MH at 4 and 13 to 17 days of age increased profitability. Profitability of this strategy depends on price received for pig meat (influenced by backfat depth) and costs of vaccine and extra feed. | ResumenObjetivo: Valorar la eficiencia del costo de la administración de un vacuna de dos dosis contra Mycoplasma hyopneumoniae (MH por sus siglas en inglés) a cerdos antes del destete entre los 13 y 17 días de edad. Materiales y métodos: Se asignaron al azar doscientas camadas de lechones en una granja comercial (Sitio Uno) en dos grupos de tratamiento: no vacunados o vacunados con la vacuna de MH a los 4 días de edad y al destete (uso de la vacuna fuera de las recomendaciones de etiqueta). En el destete, los grupos tratados se mezclaron en dos edificios de destete Sitio Dos, segregados por sexo (875 castrados, 911 hembras). En el Sitio Tres, a las 8 semanas, los vacunados y los no vacunados se asignaron al azar a resguardos con cama o edificios con pisos de concreto. Los cerdos se sacrificaron a las 26 semanas de edad y se registraron el peso de la canal, la medición de grasa P2, y las lesiones pulmonares de cada individuo. Resultados: El peso de la canal (95.91 kg) y la medición de grasa P2 (15.35 mm) fueron mayores en los cerdos vacunados (P < .05) que en los cerdos no vacunados (92.47 kg y 14.13 mm, respectivamente). La puntuación promedio y la incidencia de lesiones pulmonares crónicas fueron más bajas en los vacunados. Al sacrificio, la mortalidad, la variación de peso de la canal, y la incidencia de pleuresía, lesiones pulmonares agudas, y pleuroneumonía no difirió entre los tratamientos. La rentabilidad fue mayor en los vacunados por $5.14 AU por cerdo. Implicaciones: Bajo las condiciones de este estudio, vacunar a los cerdos contra MH a los 4 y 13 a 17 días de edad incrementó la rentabilidad. La rentabilidad de esta estrategia depende del precio pagado por la carne de cerdo (influenciado por el grosor de grasa) y los costos de la vacuna y alimento extra. | ResuméObjectif: Évaluer le coût bénéfice de l'administration de deux doses de vaccin anti-Mycoplasma hyopneumoniae (MH) à des porcs avant le sevrage à l'âge de 13 à 17 jours. Matériels et méthodes: Deux cent portées de porcelets provenant d'une ferme de type naisseur (Site Un) ont été reparties de manière aléatoire à deux groupes de traite-ment: non-vacciné et vacciné avec un vaccin MH à 4 jours d'âge et au sevrage (usage non homologué du vaccin). Au sevrage, les deux groupes ont été mélangés dans deux pouponnières au Site Deux, et séparés selon le genre (875 castrés, 911 femelles). À l'âge de 8 semaines, les animaux vaccinés et non-vaccinés ont été répartis de manière aléatoire sur le Site Trois pour être logés soit dans un abri avec litière ou dans une étable avec plancher de ciment. À 26 semaines d'âge, les porcs ont été abattus et le poids de la carcasse, le gras dorsal P2, et les lésions pulmonaires ont été enregistrés pour chaque individu. Résultats: Chez les porcs vaccinés, le poids de la carcasse (95.91 kg) et le gras dorsal P2 (15.35 mm) étaient significativement plus élevés (P < .05) que chez les animaux non-vaccinés (valeurs de 92.47 kg et 14.13 mm, respectivement). Les pointages moyens de lésions pulmonaires et l'incidence de lésions pulmonaires chroniques étaient plus faibles chez les animaux vaccinés. Le taux de mortalité, la variation du poids des carcasses, et l'incidence de pleurésie, de lésions pulmonaires aiguës, et de pleuropneumonie au moment de l'abattage ne différaient pas entre les deux groupes. La marge de profit était supérieure de AU$5.14 par animal chez les porcs vaccinés. Implications: Dans les conditions expérimentales de cette étude, la vaccination des porcs contre MH à 4 et entre 13 et 17 jours d'âge augmentait le profit. La rentabilité de cette stratégie dépend du prix reçu pour la viande (influencé par l'épaisseur du gras dorsal) et les coûts du vaccin et de la nourriture supplémentaire. |
Keywords: swine, Mycoplasma
hyopneumoniae, vaccine
Search the AASV web site
for pages with similar keywords.
Received: July
11, 2005
Accepted: October
14, 2005
Pneumonia due to Mycoplasma hyopneumoniae (MH) is an insidious disease with the potential to cause health problems and suboptimal production parameters, including depressed growth, poor feed conversion efficiency (feed:gain; F:G), and greater variation in sale weights than in noninfected pigs. In the presence of other interacting pathogenic bacteria (eg, Haemophilus parasuis, Actinobacillus pleuropneumoniae [APP], Pasteurella multocida), it can also result in deaths among infected animals.1-3 North American and European readers should note that two major viral respiratory pathogens, porcine reproductive and respiratory syndrome virus and swine influenza virus, are exotic to Australia. Pointon et al3 reported that enzootic pneumonia initiated by MH added approximately AU$2.80 to the cost of every pig produced. It is estimated that mean average daily gain decreases by approximately 23 to 37 g for every 10% of the pig's lung affected by pneumonia.4,5
In an attempt to improve the health of pigs, many production systems in the early 1990s adopted segregated early weaning (SEW) management. Weaning pigs at < 21 days of age was an attempt to prevent transmission of pathogens from the sow herd to the progeny herd. Although these systems worked well experimentally to prevent pathogen transfer, they were often not as successful commercially. As a result, some large SEW commercial pork production systems are plagued with performance-limiting diseases in the progeny, including MH-induced pneumonia. In these systems, the best time to administer individual animal therapeutics and prophylactics (such as parenteral vaccines), for ease of handling and to minimize labor costs, is before the pigs are weaned.
In Australia, three commercial MH vaccines are currently available: Suvaxyn M. hyo (Fort Dodge Australia Pty Ltd, Baulkham Hills, NSW, Australia) and Respisure and Respisure 1 (Pfizer Animal Health, West Ryde, NSW, Australia). Most studies demonstrate that in herds or groups where MH vaccine is used, pigs have less severe lung lesions and better F:G, and either higher growth rate, less medication used, or both, than in herds or groups where MH vaccine is not used.6-8 Manufacturers of all three vaccines recommend that they be administered to pigs > 7 days of age, with at least a 14-day interval between doses.9,10 The efficacy of these vaccines in unweaned pigs £ 7 days of age in a commercial environment has not been determined. This is likely to depend on a number of factors, including the age at which pigs are infected, pathogen "load," and environmental conditions.11,12
The aim of this experiment was to determine the efficacy and commercial success of administering a two-dose MH vaccine to unweaned piglets at 4 days and 13 to 17 days of age on a commercial breeding farm undertaking SEW.
Materials and methods
Facilities and study herds
This experiment was conducted at three commercial production sites operated by the same company in Victoria, Australia. Site One was a 3000-sow breeder-farrowing site, rearing pigs to 13 to 17 days of age. Site Two was a nursery, receiving weaned pigs from Site One and from other breeder-farrowing sites, and rearing them to 8 weeks of age. Site Three was a grower-finisher site receiving pigs from Site Two and rearing them to processing at approximately 26 weeks of age.
Site One. The breeder herd was endemically infected with MH and APP serovars 1, 7, and 15 and had occasional APP-induced pneumonia deaths among gilts. Herd health status was determined by the consulting veterinarian on the basis of clinical signs, necropsy data, results of serological testing (for MH and APP), and confirmatory diagnostic tests (culture and serotyping of APP isolated from the lungs of pigs found dead on the farm or from lung lesions detected during postmortem inspection at slaughter). Replacement gilts, selected from a multiplier herd infected with APP serovar 7, were vaccinated with an autogenous APP vaccine containing serovars 1, 7, and 15 (Intervet, Bendigo, Australia), first at selection (approximately 22 weeks of age) and again on arrival at Site One (approximately 24 weeks of age). Pregnant females were given a third dose 2 weeks before farrowing in an attempt to boost piglets' passively acquired immunity. The breeding herd was not vaccinated against MH.
Site Two. The nursery was situated approximately 500 km from the breeder-farrowing site. This site consisted of 13 bedded shelters, each 20 m ´ 9 m with no internal penning, and each housing approximately 1000 pigs for a total of 13,000 pigs. Shelters were constructed of galvanized iron frames with wood and sandwich-panel walls, internally lined with industrial-quality rubber matting. Roofs were constructed of two layers of canvas. Floors were compacted clay, covered with rice hulls to a depth of approximately 1 m, with a raised concrete pad on each side of the shed where the feeders and drinkers were located. Multi-space feeders were located on one side of the shed and provided dry pelleted feed, and single-space bowl drinkers were located on the opposite wall. Exhaust fans forcibly removed air from the buildings, and air was passively drawn in through side vents. Three other nurseries of similar size, owned by the same commercial company and housing pigs at different stages of growth, were situated within a radius of approximately 5 km of the experimental nursery and received pigs from the same breeder sources.
Every 8 weeks, a batch of pigs moved through the nursery site, which was managed all in-all out (AIAO) by shelter. The site was filled over a 2-week period with pigs weaned at 13 to 17 days of age. Weaned pigs were usually sourced from 11 separate off-site breeder farms which were all part of the same production system owned by one company, resulting in some commingling of pigs from different breeder farms. After 6 weeks in the nursery, pigs were moved to several off-site grower-finisher farms, and the nursery site was emptied over a 2-week period. Shelters were cleaned using a high-pressure washer and disinfected between batches of pigs. Soiled bedding was replaced with clean bedding.
Site Three. The grower-finisher site, situated 15 km from the nursery, consisted of two traditional (concrete-floored) barns and four rice-hull-based shelters similar to those described for the nursery, each approximately 20 m ´ 9 m and housing approximately 400 pigs. Traditional barns were curtain-sided and all buildings were naturally ventilated. The flooring in the traditional barns was constructed of solid concrete at the front of the pens and concrete slats at the rear of the pens. Each pen was provided with a wet-dry feeder. Pigs were housed in groups of 20 per pen with a space allowance of 0.75 m2 per pig. In contrast, each of the four shelters was transversely subdivided into two pens, each approximately 9 m ´ 10 m, resulting in a group size of approximately 200 pigs per pen. Wet-dry feeders were situated at the boundary of the subdivision. Pigs were housed by gender (castrates or females) in each barn or shelter. Pigs were moved to the grower-finisher site at 8 weeks of age and moved out at approximately 26 weeks of age in an AIAO manner (by barn and site). Soiled bedding was removed from the shelters and all barns were cleaned and disinfected between batches of pigs. After cleaning, fresh bedding was added to the rice-hull based shelters.
Experimental design
This experiment was undertaken from July to December 2002 (winter to early summer in Australia). All experimental procedures were undertaken in consultation with the Animal Ethics Committee responsible for the welfare of the animals at the farm sites. At Site One (the breeder site), 200 litters of pigs were randomly allocated into vaccinated or nonvaccinated groups, with 100 litters per group. Treatment groups contained an equal distribution of sows of each parity housed on the farm. Piglets in the vaccinated litters were administered an intramuscular injection of MH vaccine (Suvaxyn M. hyo) at 4 days of age and at weaning (13 to 17 days of age). Vaccinated pigs were identified with a hole punched in the ear. Any pig transferred from Site One to Site Two (the nursery) without a hole punch was assumed to be nonvaccinated. Identification, vaccination, and transfer of pigs from Site One to Site Two were supervised by the principal investigator.
At weaning, 1786 pigs were transferred from Site One to Site Two, where they were allocated by gender to one of two shelters. One shelter housed 875 castrated pigs (423 vaccinated, 452 nonvaccinated) and the other housed 911 females (448 vaccinated, 463 nonvaccinated). Vaccinated and nonvaccinated pigs were commingled. The other 11 shelters on Site Two, which were not included in the experiment, each housed approximately 1000 pigs originating from other off-site breeder farms owned by the same company.
The average weaning weight of pigs in each treatment group was determined on arrival at Site Two by weighing the loaded truck, subtracting the empty truck weight, and dividing this weight by the number of pigs on the truck. The average live weight of pigs in each group transferred out of the nursery shelters was recorded in a similar manner. No attempts were made to weigh individual pigs. The numbers of nursery deaths by treatment group were recorded.
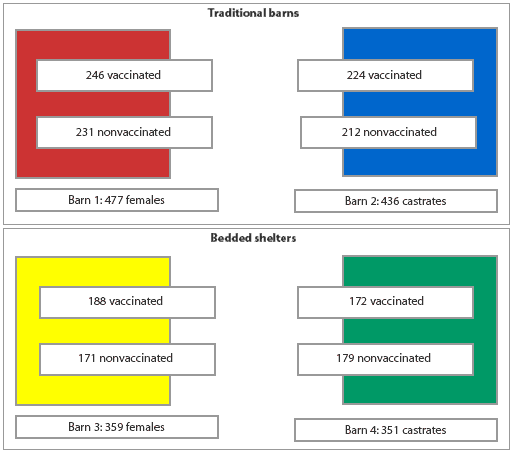
At 8 weeks of age, 1623 trial pigs were randomly selected for transfer into either bedded shelters (710 pigs) or traditional (concrete-floored) grower-finisher barns (913 pigs) at Site Three. Within these housing groups, pigs were randomly assigned to separate pens in the grower phase by treatment and gender (Figure 1). Mortalities and treatments were recorded for all groups during the grower-finisher period.
Figure 1: Allocation by gender at the grower-finisher facility of study groups of pigs either vaccinated with a commercial Mycoplasma hyopneumoniae vaccine at 4 days of age and at weaning (13 to 17 days of age) or nonvaccinated. Traditional barns had partially slatted concrete floors. Pigs were housed at appoximately 20 per pen (0.75 m2 per pig). Shelters had clay floors bedded with rice hulls, and were divided into two pens (9 m ´ 10 m), each housing appoximately 200 pigs. Vaccinated and nonvaccinated pigs were commingled both in shelters and in traditional barns.
|
Necropsies to determine cause of death were not undertaken by a veterinarian during this experiment. However, necropsies on nursery pigs prior to this study suggested that most pigs died from ill thrift due to starvation, Glasser's disease (Haemophilus parasuis), or both.
Routine medications
All pigs on Site Two were fed a diet containing 400 g per tonne tilmicosin (Pulmotil; Elanco Animal Health, West Ryde, NSW, Australia) and 200 g per tonne tiamulin (Tiamulin Fumarate; Lienert Australia, Roseworthy, SA, Australia) for the first 7 days post weaning. Pigs were then fed a diet containing lincomycin-spectinomycin (Lincospectin Antibiotic Premix; Pfizer Animal Health, West Ryde, NSW, Australia) at 44 g per tonne for 2 weeks and 22 g per tonne for the next 2 weeks. Amoxicillin (Amoxi-sol 10%; Dox-al Italia, Sulbiate, Italy) was added to the water continuously for the first 7 days after weaning at a dose rate of 10 mg per kg body weight.
All pigs were medicated with amoxicillin (Amoxi-sol 10%) at 10 mg per kg in the water for the first 3 days after moving to Site Three. Tylosin phosphate (Tylan 100 Premix; Elanco Animal Health, West Ryde, NSW, Australia) was added to the feed at 40 g per tonne for pigs 8 to 16 weeks of age and at 20 g per tonne from 16 weeks of age until processing at 26 weeks of age.
Processing data
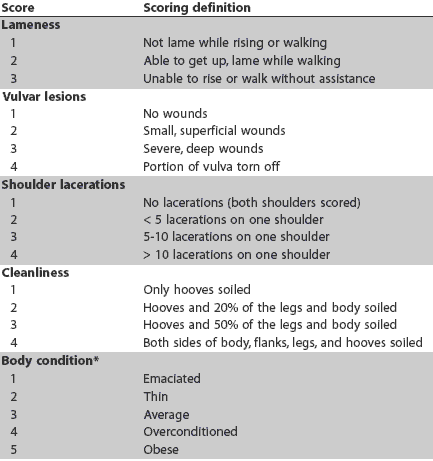
At the end of the grower-finisher period, at an average of 182 days of age (179 to 184 days), all pigs in the bedded shelters were sent to slaughter on one day, and all pigs in the traditional barns on the following day. Measurements at slaughter included hot carcass weight (HCW) and P2 backfat thickness. Average lung score was calculated on 350 vaccinated and 348 nonvac-cinated pigs which were randomly selected during slaughter from each experimental group. Lungs were graded using a scoring system described previously by Straw et al,13 with each lung set assigned a score out of 55 reflecting the proportion of lung affected by bronchopneumonia. From this, numbers of sets of lungs in each of three lung-score categories (< 10, 10-20, ³ 21) were determined. Pneumonia was determined as "acute" or "chronic" on the basis of visual inspection. Acute pneumonia was characterized by red-purple lesions, raised above the surface or at the surface of the rest of the lung, "wet," and with a liver-like consistency. In contrast, chronic pneumonia lesions were grey-white, shrunken below the surface, and had a firmer texture. Lungs with both acute and chronic lesions were graded as acute.
Numbers of pigs with chronic and acute pneumonia lesions were recorded. As MH infection may predispose pigs to other respiratory pathogens (eg, Pasteurella multocida, APP), the incidence of pleurisy and pleuropneumonia was recorded to determine if Mycoplasma vaccination impacted these other respiratory tract lesions. Pleurisy lesions between lung lobes were given a score of 1 and lesions between the lungs and the pleural cavity were given a score of 2.
Statistical analysis
Average weaning weight was determined for each group by the difference between the weight of a loaded transport truck and the empty truck, divided by the number of pigs on the truck. As there was no measure of variance between and within treatment groups, no statistical inference could be made about treatment differences in average weights in and out of the nursery site. Differences in the proportion of deaths between genders and within gender between treatments were analysed using the chi-squared test.
The experimental unit was the barn or shelter. Despite the differences in the housing type (concrete-based versus bedded), no inference could be made about the effect of housing type on vaccine efficacy, as housing was confounded with slaughter date. Backfat (P2), HCW, and average lung score were analyzed by analyses of variance (ANOVA), with housing or day of slaughter and gender as block structures. Mortalities, numbers of pigs with pleurisy (scores 1 and 2), acute and chronic pneumonia lesions, and pleuropneumonia were analyzed in generalized linear models or linear mixed models with binomial errors and with treatment as a fixed effect and barn or day of slaughter and gender as random effects. Numbers of pigs in the three lung-score categories were analyzed in a generalized linear model with multinomial errors. The association between HCW and average lung score, and numbers of pigs with acute and chronic pneumonia lesions, were analyzed by linear regression. Variances within each of the eight experimental units were fitted as response variables in ANOVA. For all statistical methods, a P value of < .05 was considered significant.
Results
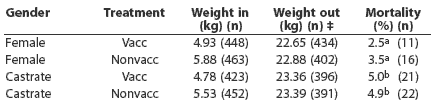
Average weaning weight of pigs in the vaccinated litters was less than in the nonvaccinated pigs by 950 g for females and 750 g for castrates. This weight difference had no impact on growth performance during the nursery phase, with the weights of vaccinated (22.65 kg) and nonvaccinated females (22.88 kg) and vaccinated (23.36 kg) and nonvaccinated castrates (23.39 kg) approximately equal on transfer to the grower site at 8 weeks of age.
There were no significant differences in nursery mortality between treatments for either females or castrates (Table 1). At the grower-finisher site, 58 study pigs died, with cause of death recorded for 29. Of the recorded deaths, the most common reason attributed was euthanasia due to ill thrift (six pigs) or arthritis (nine pigs). As a result, 692 pigs were transported for processing from the bedded shelters and 873 pigs from the traditional barns.
Table 1: Average weights* at transfer into and out of the nursery site and nursery mortality (expressed as a % of each population) for pigs either vaccinated (Vacc) or nonvaccinated (Nonvacc) for Mycoplasma hyopneumoniae†
* Average weights for each group determined by the difference between the weight of a loaded pig transport truck and the empty pig truck, divided by the number of pigs on the truck. Without a measure of variance between and within treatment groups, no statistical inference can be made about treatment differences in average weights in and out of the nursery site. † Pigs were vaccinated at 4 days of age and at weaning (13 to 17 days of age) with Suvaxyn M. hyo (Fort Dodge Australia Pty Ltd, Baulkham Hills, NSW, Australia). ‡ Weight out of the nursery includes only the 1623 pigs that were selected to remain in the study for the grower-finisher period. ab Within a column, values with different superscripts are significantly different (P < .05; c2 test). |
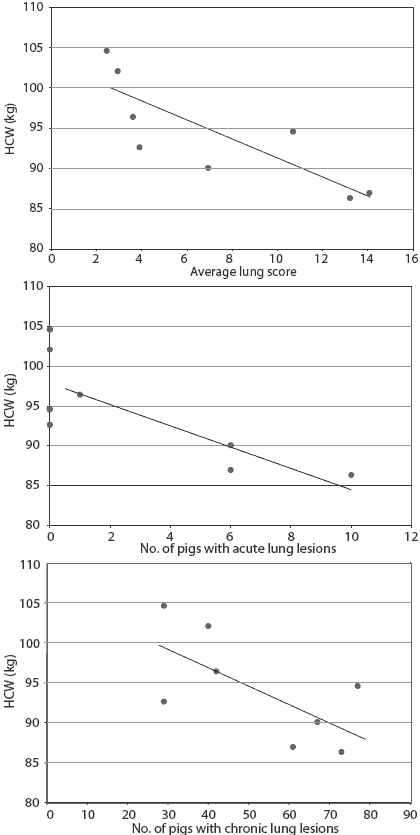
Hot carcass weight and P2 backfat at processing were higher in the vaccinated than in nonvaccinated pigs (Table 2); there was no difference between treatment groups in carcass weight variability. There was a trend (P < .10) for a lower average lung score among vaccinated pigs compared to nonvac-cinated pigs (Table 2). Hot carcass weight was negatively associated with average lung score (Figure 2). Upon inspection of lungs at processing, the incidences of pleurisy, acute pneumonic lesions, and pleuropneumonia did not differ between vaccinated and nonvaccinated groups. There was a trend (P < .10) for fewer chronic lung lesions among vaccinated pigs compared to nonvaccinated pigs. Hot carcass weight was negatively associated with the numbers of pigs with acute lesions, and there was a trend for the same association for chronic lesions (Figure 2). Mortality rates did not differ among vaccinated and nonvaccinated pigs (P > .05). Proportionately fewer vaccinated pigs were included in the higher lung-score categories (P < .001) (Table 3). There were no significant interactions between vaccination and gender of pigs in any of the variables measured (P > .05).
Table 2: Predicted means of P2 back fat, average lung score, and hot carcass weight (HCW) for pigs either vaccinated (350 pigs) with a commercial Mycoplasma hyopneumoniae vaccine at 4 days of age and at weaning (13 to 17 days of age) or not vaccinated (348 pigs)
* Carcasses were randomly selected from each treatment group at slaughter. Back fat (P2), average lung score, and HCW were analyzed by analyses of variance with housing or day of slaughter and gender as block structures. † Average lung score calculated using a scoring system described by Straw et al,13 with each lung set assigned a score out of 55 reflecting the proportion of bronchopneumonia. |
Figure 2: Association between hot carcass weight (HCW) and average lung score (lung-scoring system described in Table 2), average number of pigs with acute lung lesions, and average number of pigs with chronic lung lesions in a group of 698 pigs shipped from the same grower-finisher site, analyzed by linear regression. Hot carcass weight was negatively associated with average lung score (P < .05; adjusted r2 = 60%) and average number of pigs with acute lesions (P < .05; adjusted r2 = 57.4%). There was a trend for the same association for average number of pigs with chronic lesions (P < .10; adjusted r2 = 35%).
|
Table 3: Distribution in three pneumonia lung-score categories of pigs either vaccinated (Vacc) with a commercial Mycoplasma hyopneumoniae vaccine at 4 days of age and at weaning (13 to 17 days of age) or not vaccinated (Nonvacc)*
* Carcasses randomly selected from each treatment group at slaughter. Average lung score calculated using a scoring system described by Straw et al,13 with each set of lungs assigned a score out of 55 reflecting the proportion of bronchopneumonia. Numbers of sets of lungs in each of three lung-score categories (< 10, 10-20, ³ 21) were determined. ab Within a column, values with different superscripts are significantly different (P < .001; generalized linear model with multinomial errors). |
Discussion
The results of this experiment support administration of a commercial two-dose MH vaccine to unweaned pigs at 4 and 13 to 17 days of age to increase carcass weight and maximize the profitability of this pork production system. The profits to be gained from MH vaccination will depend on the price received per kg of pig meat minus additional costs involved in rearing vaccinated pigs. Price received will be affected by current market prices and the abattoirs' pricing grid (affected by weight and P2 backfat), and additional costs will be affected by vaccine cost, feed price, and feed efficiency.
It is estimated that for every additional 10% of affected lung tissue (lung score 5.5), F:G worsens by 2.5%.14 In this experiment, the difference in lung scores between vaccinates and nonvaccinates was 6.0 (11% of affected lung); therefore, F:G would be expected to worsen by about 2.5% in nonvaccinates (for example, F:G of 3.20 in vaccinated pigs and 3.28 in nonvaccinated pigs).15 If we assume a feed cost of $AU0.35 per kg, then the cost of extra feed is $AU1.27 per pig (vaccinated pig feed cost = 95.91 kg ´ 3.2 F:G ´ $AU0.35 = $AU107.42, minus nonvaccinated pig feed cost = 92.47 kg ´ 3.28 F:G ´ $AU0.35 = $AU106.15 per pig). The potential profits from MH vaccination per pig can be calculated as [price received ($AU2.50 per kg ´ 3.44 kg = $AU8.60)] minus [vaccine cost ($AU1.25) + reduced % lean meat from extra fat ($AU0.95) + extra feed cost ($AU1.27)] = $AU5.13.
This experiment demonstrated the efficacy of administering a two-dose MH vaccine to very young pigs (< 3 weeks of age). These results are in agreement with those of Smith et al,16 who found significantly fewer lung lesions among pigs administered a single-dose MH vaccine at 7 to 10 days of age compared to nonvaccinated pigs, and a trend towards faster growth (618.5 g per day) in vaccinated pigs compared to nonvaccinated pigs (607.3 g per day).16
The impact that MH has on the overall performance of pigs in a herd will depend on season, herd size, proximity to other infected farms, pig flow (continuous versus AIAO), and air quality.17,18 Thus, the results of this experiment will not be representative of all pork production systems.
It is likely that MH infection pressure was higher at the start of the experiment (ie, in the winter months), when ventilation rates were minimized to keep the nursery pigs warm, than at the end of the experiment in the early summer. Jorsal and Thomsen17 reported an increased incidence of MH infections in Denmark in the autumn and winter months that may be due to decreased ventilation rates and a buildup of airborne pathogens and atmospheric contaminants in the environment.19
In this experiment, nursery pigs were housed in shelters containing approximately 1000 pigs per shelter, with 13 shelters per site. In addition, three other nurseries of similar size housing pigs at different stages of growth were situated within a radius of approximately 5 km of the experimental nursery. Cargill et al12 undertook a widespread epidemiological study of 32 naturally ventilated farms in Australia and reported positive correlations between the number of pigs in the barn and pleurisy prevalence, pneumonia prevalence, and coughing rate.12 Hence, the large population density present at the experimental nursery would have increased the MH infection pressure on pigs and the likelihood of respiratory disease.17
Systems that incorporate AIAO management and that clean facilities between batches of pigs maximize hygiene and air quality.11,20 Pigs housed in AIAO barns that were cleaned thoroughly between batches grew 39 g per day faster, and had significantly less lung damage and pleurisy at slaughter, than pigs housed in adjacent AIAO sections that were not cleaned.21 In this experiment, pigs were housed on farms that were managed AIAO by barn and by site, with thorough cleaning between batches, and were likely to have been healthier than pigs grown on a single site using continuous-flow management. The positive health and growth performance outcomes that could be predicted from the AIAO management of these pigs were most likely offset to some degree by the air quality in the nursery barns. High concentrations of atmospheric contaminants, particularly dust and ammonia, have been recorded in bedded barns housing pigs.22,23 Although no air quality parameters were measured during this study, previous air quality measurements in barns on Site Two demonstrated ammonia concentrations as high as 28 mg per kg22 (target for pigs is < 11 mg per kg)24 and total dust concentrations as high as 19 mg per m3 (target for pigs is < 2.4 mg per m3).24 This is likely due to the amount of dust associated with rice-hull bedding and failure to remove manure and urine from the environment in these bedded systems.23 The combination of MH infection pressure and poor air quality most likely contributed to the pneumonia in these pigs.25-27
Implications
- Under the conditions of this study, vaccinating pigs for MH at 4 and 13 to 17 days of age (off-label use) increases profitability by approximately AU$5.13 per pig.
- Factors affecting the profitability of this early MH-vaccination strategy include price received for pig meat, vaccine cost, extra feed costs for growing vaccinated pigs, and impact of increased carcass backfat on percentage of lean meat or price received.
References
1. Betts AO. Respiratory diseases of pigs. V. Some clinical and epidemiological aspects of virus pneumonia of pigs. Vet Rec. 1952;64:283-288.
2. Amass S, Clark L, Alstine WV. Interaction of Mycoplasma hyopneumoniae with Pasteurella multocida infections in swine. JAVMA. 1994;204:102-107.
3. Pointon AM, Byrt D, Heap P. Effect of enzootic pneumonia of pigs on growth performance. Aust Vet J. 1985;62:13-18.
4. Straw BE, Tuovinen VK, Bigras-Poulin M. Estimation of the cost of pneumonia in swine herds. JAVMA. 1989;195:1702-1706.
5. Straw BE, Shin S, Yeager A. Effect of pneumonia on growth rate and feed efficiency of minimal disease pigs exposed to Actinobacillus pleuropneumoniae and Mycoplasma hyopneumoniae. Prev Vet Med. 1990;9:287-294.
6. Maes D, Deluyker H, Verdonck M, Castryck F, Miry C, Vrijens B, Verbeke W, Viaene J, Kruif A. Effect of vaccination against Mycoplasma hyopneumoniae in pig herds with an all-in/all-out production system. Vaccine. 1999;17:1024-1034.
*7. Lium B, Lund A, Skomsov A. A field study on vaccination against Mycoplasma hyopneumoniae infection of pigs. Proc IPVS. Bangkok, Thailand. 1994;191.
*8. Vraa-Andersen L, Christensen G, Kuiper R. Vaccine efficacy trial with Suvaxyn™ M. hyo in Denmark. Proc IPVS. Bangkok, Thailand. 1994;192.
*9. Technical Fact Sheet TFS-04-00 (1). Suvaxyn™ M. hyo - How it works. Baulkham Hills NSW 2153, Australia: Fort Dodge Australia Pty Ltd; 2000.
*10. RespisureOne™ Technical Manual. West Ryde NSW 2114, Australia: Pfizer Animal Health; 2002.
*11. Cargill C, Banhazi T, Connaughton I. The influence of air quality on production increases associated with all-in/all-out management. Proc IPVS. Birmingham, UK. 1998;248.
*12. Cargill C, Skirrow S, Banhazi T. The relationship between pig population size, stocking density, air quality parameters and pleurisy in pig herds. Proc IPVS. Bologna, Italy. 1996;521.
13. Straw BE, Backstrom L, Lehman AD. Examination of swine at slaughter. Part II. Findings at slaughter and their significance. Comp Contin Educ Pract Vet. 1986;8:S106-S112.
14. Frey B. Eradication of Mycoplasma pneumonia by Swiss depopulation. In: Cutler R, ed. Eradicating Diseases of Pigs. Kingston, Australian Capital Territory: The Pig Research and Development Corporation; 2001:47-59.
*15. Cleary G. PigStats. Deakin West, Australian Capital Territory 2600, Australia: Australian Pork Ltd (formally Pig Research and Development Corporation); 2001.
16. Smith S, Pommier P, Keita A, Pagot E, Thevasagayam S, Salt J, Peters A. Efficacy of Stellamune™ one, a single dose Mycoplasma hyopneu-moniae vaccine, administered in pigs at one week of age under field conditions. Tierarztliche Umschau. 2003;58:316-324.
17. Jorsal S, Thomsen B. A Cox regression analysis of risk factors related to Mycoplasma suipneumoniae reinfection in Danish SPF herds. Acta Vet Scand Suppl. 1988;29:436-438.
18. Goodwin R. Apparent reinfection of enzootic pneumonia in pig herds. Search for possible causes. Vet Rec. 1985;116:690-694.
*19. Baeko P, Pedersen P, Thomsen L. Impact of air quality on respiratory diseases and productivity. Proc IPVS. Bologna, Italy. 1996;522.
*20. Cargill C, Madec F, Banhazi T. Hygiene, health and production. Proc Aust Assoc Pig Vet. Perth, Australia. 2000;77-84.
*21. Cargill C, Banhazi T. The importance of cleaning in all-in/all-out management systems. Proc IPVS. Birmingham, England. 1998;15.
*22. Holyoake P. Developing a safe environment for people working in pig production systems. Final Report for Australian Pork Limited. Deakin West, Australian Capital Territory 2600, Australia. 2002.
*23. Banhazi T, Cargill C, Marr G, Kefford A, Moore K, Koch S, Payne H, Nicholls R. Relating airborne pollution to management and housing factors. Final report to Australian Pork Limited (formally Pig Research and Development Corporation), PO Box 148, Deakin West, Australian Capital Territory 2600, Australia. 2000;39-41.
24. Donham K. Association of environmental air contaminants with disease and productivity in swine. Am J Vet Res. 1991;52:1723-1730.
25. Crowe C, Harris DH, Elliot L, Wilson E, Wiseman B. A possible relationship between facility dust and endotoxin levels and improved growth rates in pigs reared by Isowean. Swine Health Prod. 1996;4:231-236.
26. Jolie R, Backstrom L, Olson L, Chase C. Respiratory and systemic health parameters in pigs raised in a conventional farm or in isolation. Swine Health Prod. 1999;7:269-275.
27. Lee C, Giles L, Bryden W, Downing J, Owens P, Kirby A, Wynn P. Performance and endocrine responses of group housed weaner pigs exposed to the air quality of a commercial environment. Livest Prod Sci. 2005:933;255-262.
* Non-refereed references.