Case report |
Peer reviewed |
Reproductive failure associated with porcine circovirus
type 2 in gilts
Falla reproductiva asociada con el circovirus porcino tipo 2 en primerizas
Échec reproducteur chez des cochettes associé au circovirus porcin type 2
Jeremy S. Pittman, DVM
Murphy-Brown, LLC, Waverly Division, Waverly, Virginia. Corresponding author: Dr Jeremy S. Pittman, Murphy-Brown, LLC, Waverly Division, PO Box 1240, Waverly, VA 23890; Tel: 804-834-2109; Fax: 804-834-8926; E-mail: jeremypittman@murphybrownllc.com.
Cite as: Pittman JS. Reproductive failure associated with porcine circovirus type 2 in gilts. J Swine Health Prod. 2008;16(3):144–148..
Also available as a PDF.
SummaryA 4800-sow farrow-to-wean farm with a new on-site gilt-development facility experienced an 8-week period of increased abortions and mummified pigs in gilt litters. Replacement gilts previously supplied from an external source at 24 weeks of age were not affected. Fetuses from 10 aborted litters were necropsied. Placenta, umbilical cord, stomach contents, thoracic fluid, heart, lung, liver, spleen, kidney, and thymus, and whole blood from dams, were collected. Histopathological evaluation demonstrated nonsuppurative myocardial mononuclear inflammation with extensive multifocal myocardial mineralization. In all litters submitted, porcine circovirus type 2 (PCV2) antigen was identified by immunohistochemistry in fetal heart lesions. Pooled fetal lung tissue was positive for PCV2 by polymerase chain reaction (PCR). Fetal lung tissue and thoracic fluid were PCR-negative for porcine reproductive and respiratory syndrome virus (PRRSV). Fetal lung tissue was negative for parvovirus by fluorescent antibody testing. Fetal kidneys were culture-negative for Leptospira and other pathogenic bacteria. Gilt sera were negative for PRRSV by PCR and negative for antibodies to PRRSV by ELISA. Serological testing of gilts demonstrated low antibody titers to parvovirus and six serovars of Leptospira interrogans. In addition, PCV2-associated enteritis, pneumonia, and systemic infections were diagnosed in five 20-week-old gilts during the period of reproductive failure. | ResumenUna granja de 4800 hembras de parto a destete con una nueva unidad de desarrollo de primerizas experimentó un aumento de abortos y cerdos momificados en camadas de primerizas por un periodo de 8 semanas. Las primerizas de reemplazo previamente abastecidas por una fuente externa a las 24 semanas de edad no fueron afectadas. Se realizó la necropsia a los fetos de 10 camadas abortadas. Se recolectaron placenta, cordón umbilical, contenidos del estómago, fluido torácico, corazón, pulmón, hígado, bazo, riñón, y timo, y sangre de las madres. La evaluación histopatológica demostró inflamación no supurativa mononuclear del miocardio con mineralización multifocal extensiva del miocardio. En todas las camadas analizadas, se identificó el antígeno del circovirus tipo 2 (PCV2 por sus siglas en inglés) porcino por inmunohistoquímica en lesiones de corazón fetal. El tejido pulmonar fetal analizado en grupos resultó positivo al PCV2 por la reacción en cadena de polimerasa (PCR por sus siglas en inglés). El tejido pulmonar y el fluido torácico fetales resultaron PCR negativos al virus del síndrome reproductivo y respiratorio porcino (PRRSV por sus siglas en inglés). El tejido pulmonar fetal resultó negativo a parvovirus mediante la prueba de anticuerpos fluorescentes. Los riñones fetales resultaron negativos al cultivo de Leptospira y otras bacterias patógenas. El suero de las primerizas fue negativo a PRRSV por PCR y negativo a los anticuerpos de PRRSV por ELISA. Las pruebas serológicas de las primerizas demostraron bajos títulos de anticuerpos a parvovirus y a seis serovariedades de Leptospira interrogans. Además, se diagnosticó enteritis asociada al PCV2, neumonía, e infecciones sistémicas en cinco primerizas de 20 semanas de edad durante el periodo de falla reproductiva.
| ResuméUne ferme de type naisseur-sevreur comptant 4800 truies et possédant sur le site de nouvelles installations de développement des cochettes a été confrontée pendant une période de 8 semaines avec une augmentation des avortements et des porcelets momifiés dans les portées des cochettes. Les cochettes de remplacement fournies préalablement par une source externe à 24 semaines d’âge n’étaient pas affectées. Des nécropsies ont été effectuées sur les fÅ“tus provenant de 10 portées avortées. Le placenta, le cordon ombilical, le contenu stomacal, le liquide thoracique, le cÅ“ur, le poumon, le foie, la rate, le rein, et le thymus ont été récoltés, de même que du sang entier prélevé des mères. L’évaluation histopathologique a montré une inflammation non-suppurée mononuclée du myocarde avec une minéralisation multifocale extensive du myocarde. Dans toutes les portées soumises, l’antigène du circovirus porcin de type 2 (PCV2) a été identifié par immunohistochimie dans les lésions cardiaques fÅ“tales. Le pool de tissu pulmonaire était positif pour PCV2 par réaction d’amplification en chaîne par la polymérase (PCR). Les échantillons de tissu pulmonaire fÅ“tal et de fluide thoracique étaient négatifs par PCR pour le virus du syndrome reproducteur et respiratoire porcin (PRRSV). Le tissu pulmonaire fÅ“tal était négatif pour le parvorirus par immunofluorescence. Les tissus rénaux fÅ“taux étaient négatifs en culture pour Leptospira et autres bactéries pathogènes. Les cochettes étaient négatives pour le PRRSV par PCR et négative par ELISA pour la présence d’anticorps dirigés contre PRRSV. La conduite de tests sérologiques chez les cochettes a démontré la présence de faibles taux d’anticorps dirigés envers le parvovirus et six sérovars de Leptospira interrogans. De plus, des lésions d’entérite, de pneumonie, et d’infections systémiques associées à PCV2 ont été diagnostiquées chez cinq cochettes âgés de 20 semaines durant la période des échecs reproducteurs. |
Keywords: swine, porcine circovirus type 2,
reproductive failure, gilts, mummified fetuses, PCV2
Search the AASV web site
for pages with similar keywords.
Received: September 13, 2007
Accepted: December 21, 2007
Porcine circovirus type 2 (PCV2) disease is commonly associated with poor growth performance and mortality in the grow-finish phase of swine production. Porcine circovirus associated disease (PCVAD) includes pneumonia, systemic infections, lymphadenopathy with lymphoid depletion, enteritis, and nephritis. Other descriptions of PCVAD pathology include post-weaning multisystemic wasting syndrome and porcine dermatitis and nephropathy syndrome.1,2 Less commonly reported are reproductive losses attributed to PCV2 infection. Clinical signs are described as increases in mid- to late-term abortions, mummified fetuses, stillborn pigs, and weak, non-viable piglets at birth. Cases of reproductive disease associated with PCV2 have been described in newly populated herds and in replacement breeding stock from new sources and are almost exclusively reported in gilts.
The first reported case of PCV2-associated reproductive failure in swine in 1999 by West et al3 described PCV2 as the causative agent of abortion in a single litter in a herd experiencing late-term abortions as well as increased incidence of stillborn and mummified piglets in a new farm that had been stocked with unbred gilts. No other clinical signs of disease were reported, and there was no demonstrated association with other abortigenic agents, such as porcine reproductive and respiratory syndrome virus (PRRSV), parvovirus, or Leptospira interrogans serovars. West et al3 described the fetal pathology as diffuse myocarditis with extensive staining of PCV2 antigen by immunohistochemistry, with PCV2 antigen also present in other fetal tissues.
Other case reports and retrospective analyses of reproductive failure have implicated PCV2 either as the sole agent or in conjunction with other reproductive disease agents.4-13 Others have demonstrated that experimental infection with PCV2 can result in reproductive disease with associated fetal pathology.14-18 Subsequent experimental work and field evaluation suggest that the fetal heart is the main target organ of PCV2-associated reproductive failure, but that this myocardial tropism changes as gestational age increases.19-21 A more complete review of PCV2 pathogenesis of reproductive losses has been documented.22
Diagnosis of PCV2-associated reproductive disease is based on three criteria: increased late-term abortions and stillborns or mummies or both; microscopic nonsuppurative necrotizing to fibrosing myocarditis in fetal heart tissue; and PCV2 antigen in affected fetal tissues.23
Diagnosis of PCV2-associated reproductive failure is infrequent, according to cases submitted to the following diagnostic laboratories: the Iowa State University Veterinary Diagnostic Laboratory (47 cases, January 2000 to May 2007; L. Karriker, written communication, 2007)2, the University of Minnesota Veterinary Diagnostic Laboratory (19 cases, October 2006 to May 2007; J. Torrison, written communication, 2007), the Kansas State University Diagnostic Laboratory (four cases, November 2005 to May 2007; R. Hesse, personal communication, 2007), the South Dakota State University Animal Disease Research and Diagnostic Laboratory (no cases, June 2006 to May 2007; L. Holler, personal communication, 2007), and the University of Guelph Animal Health Laboratory (four cases, January 1998 to December 2004).24
This report describes reproductive failure associated with PCV2 in gilts in a 4800-sow farrow-to-wean facility.
Herd description
The case herd included 4800 sows in a farrow-to-wean production system with a newly added on-site gilt-development facility. Prior to construction of the gilt facility, the on-site isolation building had received, at 8-week intervals, 390 twenty-four to 26-week-old PRRS-negative gilts from an external source. In the fall of 2005, the owner had built a new 500-head nursery and 500-head grower barn. The existing isolation building was used as a 500-head developer building. At 8- to 9-week intervals, females were bred with a maternal line of semen to produce replacement gilts. Selected gilts were moved into a breeding barn used primarily for gilt breeding, growers were moved into the developer, nursery pigs were moved to the grower, and internally multiplied gilts were weaned into the nursery as a cohort of candidates. For the year prior to appearance of clinical signs, monthly monitoring of serum samples from 20 piglets at weaning tested PCR-negative for PRRSV. Results of monthly monitoring of 20 gilts 2 weeks before their due dates, and bimonthly monitoring of 20 gilts at the end of each 8-week grow phase in the on-site gilt facilities, remained seronegative by Idexx PRRS ELISA (Idexx Laboratories, Westbrook, Maine).
Candidate replacement gilts were vaccinated with a one-dose Mycoplasma hyopneumoniae vaccine in the gilt nursery and two doses of an autogenous trivalent swine influenza virus (SIV) vaccine in the gilt grower. Gilts received two doses of a parvovirus-Leptospira-erysipelas vaccine, and one additional dose of both M hyopneumoniae and SIV vaccines before entering the breeding herd.
Clinical signs
During an 8-week period beginning mid-March 2007, after the first internally multiplied gilts had farrowed, the herd experienced an increase in incidence of gilt abortions and mummified fetuses in gilt litters. Abortions included mummified or stillborn fetuses or both, of varying crown-rump lengths. Replacement gilts that had entered the herd from the external source were not affected.
Production data combining all parities from PigCHAMP records (PigCHAMP, Inc, Ames, Iowa) indicated an increase in mummies during the 8-week period of the outbreak. In the 52 weeks prior to the outbreak, average mummified fetuses per litter was < 0.1. During the outbreak, average mummified fetuses per litter increased to 0.4, ranging from 0.1 to 0.7 per litter per week. In the 10-week period following the outbreak, average mummified fetuses per litter returned to the normal level of < 0.1.
Percentage of piglets born alive was lower and percentage of stillborn and mummified fetuses was higher in the internally sourced gilts than in the externally sourced gilts that had farrowed just before them. This resulted in 1237 fewer piglets born alive to internal gilts than expected from historical gilt performance (Table 1). The increase in mummified fetuses was observed only in gilt litters.
Table 1: Gilt farrowing performance data by gilt source in a herd experiencing an outbreak of reproductive failure
* Data for the externally sourced gilts was considered the performance standard. Difference in numbers of pigs born to externally and internally sourced gilts was calculated by multiplying the difference between gilt sources for each performance mean by the number of litters born to gilts from the internal source. NA = not applicable. |
|||||||||||||||||||||||||||||||||||||||
Between March 1 and May 31, 2007, records showed that 17 of a total of 37 abortions (46%) occurred in gilts. In April, at the peak of clinical disease, nine of the total of 13 abortions (69%) occurred in gilts. In contrast, during March 2006 to January 2007, when replacement gilts came from the external source, only 27 of the total of 222 recorded abortions (12%) occurred in gilts. Gilt abortions occurred at 79 to 109 days of gestation.
Laboratory testing
Mummified and stillborn fetuses and whole blood were collected from gilts that aborted, and fresh and formalin-fixed tissues were collected from two to three fetuses per litter of 10 aborted gilt litters. Placenta, umbilical cord, stomach contents, thoracic fluid, heart, lung, liver, spleen, kidney, and thymus were collected from each fetus. Tissue samples from a litter were pooled in the same container and submitted to the Iowa State University Veterinary Diagnostic Laboratory (ISU-VDL) in Ames, Iowa.
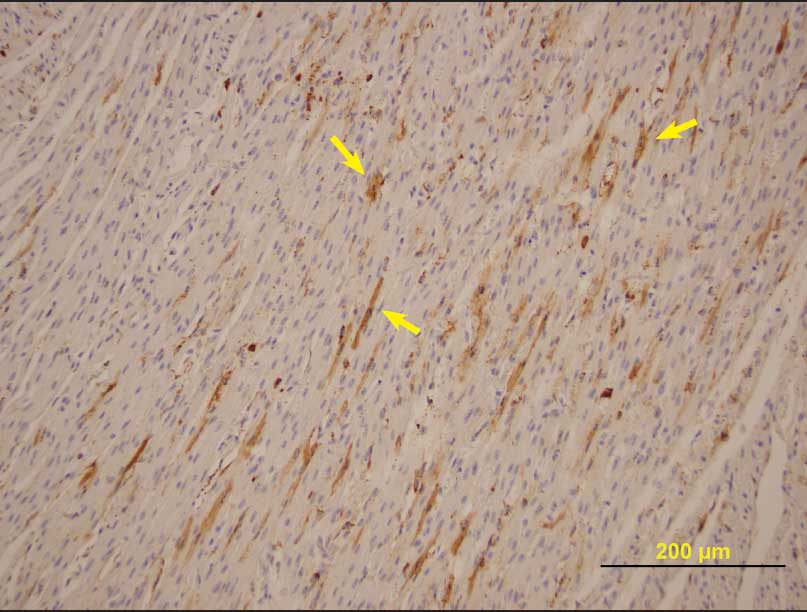
Histopathological evaluation of fetal tissue demonstrated nonsuppurative myocardial mononuclear inflammation, extensive multifocal myocardial mineralization, scattered foci of shrunken or poorly developed cardiomyofibers, and scattered irregular foci of myofiber atrophy or lack of development. Immunohistochemistry for PCV2 was positive in all litters from which samples were submitted, with moderate amounts of antigen in fetal heart tissues (Figure 1) and other organs. Pooled lung tissue was PCR-positive for PCV2, and lung tissue and thoracic fluid were PCR-negative for PRRSV. Fetal lung tissue was negative for parvovirus by fluorescent antibody testing. Leptospira organisms were not cultured from fetal kidneys, and other pathogenic bacteria were not isolated on routine bacteriological culture.
| Figure 1: Porcine circovirus type 2 antigen (arrows)
demonstrated by immunohistochemical staining of nonautolyzed
myofibers in a aborted fetus from a herd experiencing an outbreak
of reproductive failure in internally sourced gilts (magnification
× 200).
|
Gilt sera were PCR-positive for PCV2, PCR-negative for PRRSV, and negative for antibody to PRRSV by ELISA. Gilt antibody titers to parvovirus and six L interrogans serovars were low, suggesting a lack of exposure to these pathogens.
Based on the clinical findings and results of diagnostic tests, the diagnosis was PCV2-associated fetal death and mummification with myocardial necrosis or dysplasia.
Sequencing of a PCV2 isolate from one set of mummified fetuses characterized it as a PCV2b strain, with a predicted restriction fragment length polymorphism pattern of 3-2-1.
Supporting the diagnosis of PCV2-associated disease, concurrent clinical signs of PCVAD were observed in the on-site gilt grower and developer populations during the period of reproductive losses. Approximately 2% to 3% of the developing gilts lost weight and body condition, developed persistent diarrhea, appeared jaundiced, demonstrated respiratory distress, and responded poorly to antibiotic treatment. Five 70-kg to 80-kg clinically affected gilts were euthanized. Samples of heart, lung, liver, spleen, kidney, ileum, jejunum, cecum, colon, tonsil, and lymph nodes (thoracic, bronchial, internal iliac, mesenteric, and inguinal) were collected fresh and fixed in formalin. Samples were submitted to the ISU-VDL. Immunohistochemical staining demonstrated moderate to severe lymphoid depletion with abundant amounts of PCV2 antigen in all five animals, and bronchointerstitial pneumonia and PCV2-associated enteritis were evident in four of the five animals. All pooled lung samples were PCR-negative for PRRSV, SIV, and M hyopneumoniae. Streptococcus suis was isolated from lung tissue of one pig with fibrinosuppurative epicarditis lesions.
Treatment and outcome
Due to an incomplete understanding of the disease, the relative infrequency of the syndrome, the short duration of clinical signs, and inability of treatment programs to control other PCV2-associated diseases, there are no recommended treatments for reproductive failure associated with PCV2. Previously reported management strategies have been limited to changing the gilt source, planned exposure to PCV2-infected tissues, or allowing the population to become immune through natural exposure.4,5
In February of 2007, this herd began a new program for vaccination of replacement gilts against PCV2. Weaned pigs placed in the on-site gilt nursery were vaccinated intramuscularly at 3 weeks of age with Inglevac CircoFLEX (Boehringer Ingelheim Vetmedica, Inc, St Joseph, Missouri). When PCV2 was diagnosed, pigs in the nursery phase had already been vaccinated. With the goal of eliminating remaining nonimmune gilts, all internally multiplied gilts in the developing and breeding herds were vaccinated intramuscularly with Inglevac CircoFlex in April of 2007.
In an attempt to reduce the impact of PCVAD in the grower and developer populations, affected animals were segregated and either euthanized on-farm or shipped to a cull market, depending on size and body condition. Three weeks after the developing gilt herd was vaccinated for PCV2, a manure-feedback protocol was initiated, ie, fecal material from sows and gilts in the breeding and farrowing barns was collected and fed to gilts in the development barn.
Discussion
This case of reproductive failure caused by PCV2 involved a change in gilt source, as in previously reported cases.3-6,9,10 Increases in late-term abortions and mummified fetuses were observed in gilts born and raised in the new on-site gilt-development facilities, but not in sows or gilts from a previous external source. Prior to this outbreak, there was no clinical or diagnostic evidence of PCVAD in the herd. It is not clear if clinical disease developed with introduction of nonimmune gilts into the herd or with introduction of a new PCV2 strain.
Externally sourced sows in this herd did not experience an outbreak of reproductive failure, suggesting that these animals had developed protective immunity prior to breeding. Gilts born and raised on the farm were weaned at 17 to 21 days of age, and colostral immunity might have produced a population of weaned gilts with passive protection against PCV2. The first groups of replacement gilts were placed in newly built facilities where there would be little or no environmental exposure to PCV2, which may have predisposed groups of gilts to enter the breeding herd with no active immunity to PCV2. Exposure and infection during gestation may have resulted in PCV2 viremia, subsequent fetal pathology, and fetal death, manifested as an increase in mummified fetuses and abortions.
The PCV2 isolate from this case was characterized as a PCV2b strain, which might represent a novel strain introduced into the herd. Isolates characterized as PCV2b have been associated with a higher prevalence of PCVAD outbreaks in Canadian25 and Kansas26 swine herds than have isolates characterized as PCV2a. However, a relationship between PCV2b and reproductive disease has not been documented. Meehan et al7 characterized two isolates from cases of reproductive failure as PCV2b, and Farnham et al27 identified two similar isolates in stillborn pigs. In contrast, the isolate that Yoon et al17 used to reproduce reproductive disease was characterized as a PCV2a isolate (KJ Yoon, written communication, 2007). There are no other published reports of strain identification in cases of PCV2-associated reproductive failure. No historical data concerning PCV2 status of this herd was available, thus no temporal relationship may be made between identification of the isolate and clinical disease. The impact of strain variation as it pertains to clinical disease and epidemiology in this case is unclear.
Implications
- Differential diagnosis of abortions, mummified fetuses, and stillborn piglets should include PCV2, particularly when gilts are predominantly affected.
- Confirmatory diagnosis of PCV2-associated reproductive failure requires tissue pathology and detection of PCV2 antigen in fetal tissues, especially fetal heart tissue.
- Immunity of replacement gilts to PCV2 should be considered when changing gilt sources and in low-immunity, high-risk herds, such as all-gilt or low-parity herds, recently populated herds, and herds with high replacement rates.
Acknowledgements
The author would like to thank Dr Alan Loynachan (ISU-VDL) for assistance with the histopathology image.
References
1. Allan GM, Ellis JA. Porcine circoviruses: a review. J Vet Diagn Invest. 2000;12:3–14.
*2. Opriessnig T, Halbur P. Current status of PCVAD: diagnostic and research update. Proc Iowa State Univ Swine Disease Conf Swine Pract. Ames, Iowa. 2006;105–128.
3. West KH, Bystrom JM, Wojnarowicz C, Shantz N, Jacobson M, Allan GM, Haines DM, Clark EG, Krakowka S, McNeilly F, Konoby C, Martin K, Ellis JA. Myocarditis and abortion associated with intrauterine infection of sows with porcine circovirus 2. J Vet Diagn Invest. 1999;11:530–532.
*4. Janke B. Case report: porcine circovirus as a cause of reproductive problems. Proc Iowa Vet Med Assoc. Ames, Iowa. 2000;101.
5. Josephson G, Charbonneau G. Case report of reproductive problems in a new startup operation. J Swine Health Prod. 2001;9:258–259.
6. Ladekjær-Mikkelsen AS, Nielsen J, Storgaard T, Břtner A, Allan G, McNeilly F. Transplacental infection with PCV-2 associated with reproductive failure in a gilt. Vet Rec. 2001;148:759–760.
7. Meehan BM, McNeilly F, McNair I, Walker I, Ellis JA, Krakowka S, Allan GM. Isolation and characterization of porcine circovirus 2 from cases of sow abortion and porcine dermatitis and nephropathy syndrome. Arch Virol. 2001;146:835–842.
8. O’Connor B, Gauvreau H, West K, Bogdan J, Ayroud M, Clark EG, Konoby C, Allan G, Ellis JA. Multiple porcine circovirus 2-associated abortions and reproductive failure in a multisite swine production unit. Can Vet J. 2001;42:551–553.
*9. Sanford SE. PCV2 related reproductive failure in startup herds. Proc 17th IPVS. Ames, Iowa. 2002;1:171.
10. Kim J, Jung K, Chae C. Prevalence of porcine circovirus type 2 in aborted fetuses and stillborn piglets. Vet Rec. 2004;155:489–492.
11. Mauch C, Bilkei G. Porcine circovirus (PCV) associated losses in pregnant gilts. Pig J. 2004;53:69–74.
12. Mikami O, Nakajima H, Kawashima K, Yoshii M, Nakajima Y. Nonsuppurative myocarditis caused by porcine circovirus type 2 in a weak-born piglet. J Vet Med Sci. 2005;67:735–738.
13. Brunborg IM, Jonassen CM, Moldal T, Bratberg B, Lium B, Koenen F, Schönheit J. Association of myocarditis with high viral load of porcine circovirus type 2 in several tissues in cases of fetal death and high mortality in piglets. A case study. J Vet Diagn Invest. 2007;19:368–375.
14. Johnson CS, Joo HS, Direksin K, Yoon KJ, Choi YK. Experimental in utero inoculation of late-term swine fetuses with porcine circovirus type 2. J Vet Diagn Invest. 2002;14:507–512.
*15. Nielson J, Ladekjær-Hansen AS, Bille-Hansen V, Lohse L, Břtner A. PCV2-associated disease following intrauterine infection. Proc 18th IPVS. Hamburg, Germany. 2004;1:14.
16. Pensaert MB, Sanchez RE, Ladekær-Mikkelsen AS, Allan GM, Nauwynck HJ. Viremia and effect of fetal infection with porcine viruses with special reference to porcine circovirus 2 infection. Vet Microbiol. 2004;98:175–183.
17. Yoon KJ, Jepsen RJ, Pogranichniy RM, Sorden S, Stammer R, Evans LE. A novel approach to intrauterine viral inoculation of swine using PCV type 2 as a model. Theriogenology. 2004;61:1025–1037.
18. Park JS, Kim J, Ha Y, Jung K, Choi C, Lim JK, Kim SH, Chae C. Birth abnormalities in pregnant sows infected intranasally with porcine circovirus 2. J Comp Pathol. 2005;132:139–144.
19. Sanchez RE, Nauwynck HJ, McNeilly F, Allan GM, Pensaert MB. Porcine circovirus 2 infection in swine foetuses inoculated at different stages of gestation. Vet Microbiol. 2001;83:169–176.
20. Sanchez RE, Meerts P, Nauwynck HJ, Pensaert MB. Change of porcine circovirus 2 target cells in pigs during development from fetal to early postnatal life. Vet Microbiol. 2003;95:15–25.
21. Rose N, Blanchard P, Cariolet R, Grasland B, Amenna N, Oger A, Durand B, Balasch M, Jestin A, Madec F. Vaccination of porcine circovirus type 2 (PCV2)-infected sows against porcine parvovirus (PPV) and erysipelas: effect on post-weaning multisystemic wasting syndrome (PMWS) and on PCV2 genome load in the offspring. J Comp Pathol. 2007;136:133–144.
*22. Nauwynck H, Lefebvre D, Misinzo G, Meerts P, Mateusen B, Sanchez R, Delputte P. Pathogenesis of porcine circovirus 2 infections. Proc AASV. Orlando, Florida. 2007;489–495.
23. Segales J, Allan GM, Domingo M. Porcine circovirus diseases. In: Straw BE, Zimmerman JJ, D’Allaire S, Taylor DJ, eds. Diseases of Swine. 9th ed. Ames, Iowa: Blackwell Publishing; 2006:299–307.
*24. Van Dreumel T, McEwen B, Hordyk J, DeLay J. Abortions associated with porcine circovirus type 2. AHL Newsletter. 2004;8:38–39. Available at: http://www.labservices.uoguelph.ca/labserv/units/ahl/files/ANwsl8–4.pdf. Accessed 7 February 2008.
*25. DeLay J, McEwen B, Carman S, Van Dreumel T, Fairles J. Porcine circovirus type 2-associated disease is increasing. AHL Newsletter. 2005;9:22. Available at: http://www.labservices.uoguelph.ca/labserv/units/ahl/files/ANwsl9–3.pdf. Accessed 7 February 2008.
26. Horlen KP, Schneider P, Anderson J, Nietfeld JC, Henry SC, Tokach LM, Rowland RRR. A cluster of farms experiencing severe porcine circovirus associated disease: clinical features and association with the PCV2b genotype. J Swine Health Prod. 2007;15:270–278.
27. Farnham MW, Choi YK, Goyal SM, Joo HS. Isolation and characterization of porcine circovirus type-2 from sera of stillborn fetuses. 2003;67:108–113.
* Non-refereed references.