| Case report | Peer reviewed |
Cite as: Hughes PE, Smits RJ, Xie Y, et al. Relationships among gilt and sow live weight, P2 backfat depth, and culling rates. J Swine Health Prod. 2010;18(6):301–305..
Also available as a PDF.
SummaryTo examine a high culling rate problem, data were collected on 2154 sows culled from a commercial 5400-sow farrow-to-finish unit in New South Wales, Australia, during a 12-month period. Data recorded were parity, body weight, P2 backfat depth, and reason for culling. Additionally, body weights and backfat depths were recorded for a cross-section of the herd (3378 sows) in order to provide control sows for comparison. Reasons for culling were 63.7% for reproductive inefficiency or failure, while a further 12% were due to locomotor problems. Of females culled for reproductive reasons, 42.2% were gilts, indicating gilt management to be a major contributor to the high culling rate. When adjusted for parity, season, and day relative to weaning, culls had lower body weights (P < .05) and less backfat depth (P < .001) than controls (209.7 versus 211.4 kg and 14.8 versus 15.3 mm for culls and controls, respectively). These data suggest that inadequate sow nutrition was a contributor to the high culling rates, although the main problem was gilt management. Studies are ongoing to determine effects on retention of increased backfat and body mass of gilts at breeding and of providing increased dietary protein to younger sows. | ResumenPara examinar un problema de alto índice de desecho, se recaudó información de 2154 hembras desechadas de una unidad comercial de parto a finalización de 5400 hembras en New South Wales, Australia, durante un periodo de 12 meses. La información recaudada fue parto, peso corporal, profundidad de grasa P2, y la razón del desecho. Adicionalmente, se registraron los pesos corporales y la profundidad de grasa de un grupo de hembras del mismo hato (3378 hembras) con la finalidad de proveer hembras control para la comparación. Las razones para el desecho, fueron 63.7% por ineficiencia reproductiva ó falla, mientras que un 12% adicional se debió a problemas locomotores. De las hembras rechazadas por razones reproductivas, 42.2% fueron primerizas, señalando al manejo de primerizas como el mayor contribuidor del alto índice de desecho. Cuando se ajustaron por paridad, estación, y día relacionado al destete, las hembras desechadas tuvieron menor peso corporal (P < .05) y menos profundidad de grasa (P < .001) que los controles (209.7 contra 211.4 kg y 14.8 contra 15.3 mm para las hembras rechazadas y controles, respectivamente). Esta información sugiere que la nutrición inadecuada de la hembra fue un factor que contribuyó a los altos índices de desecho, aunque el problema principal fue el manejo de las primerizas. Los estudios continúan para determinar los efectos sobre la retención de hembras al aumentar la grasa y la masa corporal de primerizas al momento de la inseminación y de incrementar la proteína dietética a hembras jóvenes. | ResuméAfin d’investiguer un problème de taux de réforme élevé, des données ont été amassées sur 2154 truies d’une unité naisseur-finisseur de 5400 truies dans le New South Wales, Australie, pendant une période de 12 mois. Les données recueillies étaient la parité, le poids corporel, l’épaisseur du gras dorsal P2, et la raison pour la réforme. De plus, le poids corporel et l’épaisseur du gras dorsal ont été enregistrés pour un échantillonnage du troupeau (3378 truies) afin d’avoir des truies témoins aux fins de comparaison. Les causes de réforme des animaux étaient dans 63.7% des cas associées à des problèmes reproducteurs, avec un 12% supplémentaire associés à des problèmes locomoteurs. Chez les femelles réformées pour problèmes reproducteurs, 42.2% étaient des cochettes, montrant ainsi que la gestion des cochettes est un contributeur important au haut taux de réforme. Après ajustement pour la parité, la saison, et le jour relatif au sevrage, les animaux réformés avaient un poids corporel plus faible (P < .05) et une profondeur de gras dorsal moindre (P < .001) que les animaux témoins (209.7 versus 211.4 kg et 14.8 versus 15.3 mm pour les animaux réformés et les animaux témoins, respectivement). Ces données suggèrent qu’une alimentation inadéquate des truies était un contributeur majeur aux hauts taux de réforme, bien que le problème majeur soit la gestion des truies. Des études sont en cours pour déterminer les effets sur la rétention d’une augmentation de l’épaisseur de gras dorsal et de la masse corporelle des cochettes au moment de la saillie ainsi que de fournir une alimentation avec une augmentation des protéines alimentaires aux truies plus jeunes. |
Keywords: swine, sows, gilts, body condition, culling
Search the AASV web site
for pages with similar keywords.
Received: January 20, 2010
Accepted: May 28, 2010
The average sow replacement rate and parity at removal for Australian herds are 61% and 4.1, respectively.1 It has been suggested that high replacement rates are primarily a result of inadequate management, particularly inadequate nutrition of the gilt prior to herd entry and throughout her breeding life.2 Further, it has been documented that reproductive failure is the largest single cause for culling sows, and that this failure is most evident in younger sows, particularly primiparous, and to a lesser extent, second-parity sows.3 A major cause of sow infertility that, in turn, may result in early culling, is inadequate lactation nutrient intake. This will necessitate that sows mobilize their body reserves of fat and protein (lean tissue) in order to fulfill the nutrient requirements for maintenance, growth, and milk yield.4
It has been extensively documented that excessive losses during lactation of body weight and backfat, and in particular of lean tissue, will adversely affect fertility after weaning.5-7 The underlying link between lactation nutrient intake and subsequent fertility is the metabolic status of the sow, with sows having a relatively poor metabolic status exhibiting delayed estrus or anestrus after weaning.6,8 Sows having wean-to-estrus intervals > 5 days exhibit poorer fertility9,10 and are at higher risk for early culling.11 Loss of body reserves is most evident in young sows because of their innately smaller appetites. This has led to the suggestion that the breeding of gilts should be delayed in order to ensure that they have a greater lean mass and backfat depth at farrowing to buffer tissue mobilization during lactation, and so reduce post-weaning infertility. If a greater body weight and backfat depth at breeding does positively impact subsequent performance and survival, the corollary is that culled sows likely will be of lower body weight and backfat depth.
Over recent years, we have seen the introduction of increasingly leaner gilts with very high growth rates. At a given chronological age, these gilts will be considerably heavier than their counterparts of 10 or 20 years ago, but will have lower levels of body fat, resulting in their being mated at a time when their tissue reserves are marginal (eg, 12 to 16 mm P2 backfat).12 It has been suggested that when these gilts are subsequently fed employing conventional strategies, they struggle to maintain body condition from parity to parity and thus have difficulty sustaining a long breeding life (> six litters).2 It is probable that relatively low protein and fat reserves will not be a problem to a gilt or sow if she is not required to mobilize these reserves to meet nutrient demands above her daily nutrient intake. However, in practice, most sows will need to draw on their tissue reserves during lactation, although there is much between-sow variation in the degree to which this occurs. Hence, it is not surprising that there is considerable between-sow variation in body condition at weaning.
Taken together, these facts imply that sows in poor body condition, as measured by body weight and P2 backfat depth, are more likely to be culled. Further, due to the relatively hot climate, Australian sow lactation-feed intakes are relatively low, which could increase the risk of being culled.13 The objective of this study was to determine whether the high culling rate in an Australian herd was due to inadequate lactation nutrient intake as reflected in differences in body condition status of gilts and sows being culled from the breeding herd and those that remained.
Case description
Animals and reasons for culling
This study was undertaken during a 12-month period at a mature commercial 5400-sow (Large White × Landrace) farrow-to-finish facility in New South Wales, Australia, that was suffering a very high sow replacement rate (75% to 85%). Maximum daily temperatures varied from 12°C in winter to 36°C in summer. Body weights and P2 backfat depths (measured 6.5 cm off the midline at the level of the last rib; Renco Leanmeater; Renco, Minnesota) were recorded for culled animals at the time of culling (n = 2154) and for 3378 non-culled sows in order to provide herd average control measurements to which the cull-sow data could be compared.
Sows and gilts were culled due to reproductive problems (fertility or performance), locomotor problems, age (> six parities), poor body condition, and “other.” The supervisory stockpersons at the farm were responsible for culling decisions based on management decisions and adherence to welfare requirements as part of a national quality assurance program, APIQ (Australian Pork Limited, Canberra). Culling reasons were due to a single reason, the one described in the data set collected. Reproductive (fertility) reasons for sow and gilt culling (and the day relative to weaning) included anestrus (30 to 40 days), a second return to estrus (45 to 50 days), pregnancy test negative (40 to 47 days), not-in-pig (100 to 110 days), vulval discharge (0 to 10 days), and abortion (arbitrarily set at 41 to 56 days). Anestrus gilts were culled at 55 days after expected breed date. Reproductive (performance) reasons were high stillbirth rate (0 days), repeated small litter size (0 days), and low milk yield (0 days). The “other” category included udder problems (0 days) and subjectively assessed low body condition (0 days). The control animals were allocated to the study and their body weights and backfat determined on the basis of their stage of gestation, to allow comparison with nonpregnant sows culled at comparable stages, eg, if a cull sow had two returns to service after weaning, the wean-to-cull interval would be about 42 days. Weight and backfat comparisons to these cull sows would employ gestating control sows at 40 to 45 days after breeding.
Housing, feeding, and management
Gilts entered the study at the start of puberty stimulation, which commenced at 23 weeks of age using boars > 12 months of age. Each week, the stimulation procedure alternated between introducing four vasectomised boars into the pen (33 gilts per pen; 0.94 m2 per gilt) for 20 minutes with full contact and gilts being moved into a detection mating area with fenceline contact with six crated intact boars for 20 minutes. All gilts were transferred to the mating unit at 29 weeks of age regardless of recorded pubertal status.
Gilts were offered a gilt developer diet (13.8 MJ digestible energy [DE] per kg, 13% crude protein, 0.8% lysine, 3.8% crude fiber) ad libitum from 23 to 29 weeks of age and then provided with ad libitum access to a sow lactation diet (13.9 MJ DE per kg; 16% crude protein, 0.8% lysine, 3.6% crude fiber) until mating. Weaned sows were allowed 3 to 4 kg per day of the lactation diet until mating. After mating, gilts were fed 2.0 kg per day and sows fed 2.5 to 2.7 kg per day, depending on an assessment of body condition, of a gestation diet (13.0 MJ DE per kg; 13.5% crude protein, 0.6% lysine, 5.0% crude fibre). The body scoring system was based on the score described by Close and Cole.14 The exception was that there was no score 0, as these sows were identified earlier as required by welfare rules when observed as a score 1 and culled or euthanized for body condition. Sows observed as score 2 or score 1 to 2 were fed an extra 0.5 kg per day. Where possible, these sows were also removed from gestation stalls to recovery pens. This was a subjective assessment of the individual sow’s condition. At 2 weeks prior to farrowing, feed intake was increased to 2.7 kg per day for all gilts and sows. Sows were fed 3 kg of the lactation diet on the day of farrowing and the next day. Thereafter, they were fed to appetite three times a day. Sow lactation feed intakes were not recorded.
From 29 weeks of age until mating, gilts were housed in pens in groups of three to five (1.4 m2 per gilt). Weaned sows were housed in pairs. Primiparous sows were bred at their second estrus after weaning (skip-a-heat breeding). Following mating, all gilts and sows were housed in stalls (0.65 × 1.8 m). The gestation building was not insulated, and gilts and sows were housed on concrete slats. Ventilation was provided through side blinds and a central ridge vent. Cooling in summer was provided by spray cooling (5 minutes on and 20 minutes off), with temperature set at 27°C. There was no heating in winter. At 112 days of gestation, gilts and sows were transferred to farrowing crates. While in farrowing crates, sows were cooled by drip coolers set at 25°C, room ventilation occurred through slide blinds, and creep heat was supplied by heat lamps.
Gilts and sows were mated by a combination of boar service at detection of estrus and thereafter artificial insemination at 24-hour intervals while still exhibiting standing estrus. Semen was collected and processed on-site and supplied 4 × 109 sperm in 80 mL extender. Gilts were mated from approximately 30 weeks of age. The average weaning age was 25 days, with weaning occurring three times a week.
Data analysis
Statistical Analysis System software (SAS, Cary, North Carolina) version 9.1.3 (2002) was used for data analysis. Descriptive statistics, such as means and standard deviations for continuous variables and frequencies and relative frequency for categorical variables, were obtained. The 12-month period was divided into four seasons, corresponding approximately to summer (January through March), autumn (April through June), winter (July through September), and spring (October through December). General linear model (GLM) was used to compare the difference between the culled and control sows in lower body weights and lesser backfat depths after adjustment for parity, season, and day relative to weaning. Logistic regression was used to determine if weight and backfat after adjustment for season and parity were associated with the probability of being culled. The GLM and logistic regression were implemented by procedures Proc GLM and Proc logistic procedure in SAS, respectively. A P value of < . 05 was considered significant.
Changes in body condition
Sow body weights increased progressively (P < .05) in sows of increasing parity with the largest gain being between parities 0 and 1 (Figure 1). These body-weight changes were not mirrored by changes in P2 backfat thickness, with little change occurring between herd entry and removal from the herd (Figure 1). Although differences were not great, after correction for parity, season, and stage of gestation, culled sows had lower body weights (P < .05) and lesser backfat depths (P < .001) than control sows (209.7 kg versus 211.4 kg and 14.8 mm versus 15.3 mm for culls and controls, respectively). Backfat depth was ≤ 12mm in 35.7% of cull sows, but in only 25.5% of control sows. On the basis of the logistic regression, as sow weight increased, sows became less likely to be culled (P < .001), but backfat depth was not related to probability of being culled, likely because differences were small. Interestingly, there was no apparent seasonal effect on sow body weight or backfat depth, and the odds ratio for being removed during January to March (the presumed period of seasonal infertility) was not significantly different from that in the period July to September. This suggests that seasonal infertility did not impact culling rates.
Figure 1: Changes in body weight and P2 backfat depth (± SD) in relation to parity of gilts and sows culled or not culled (controls) from a 5400-sow commercial farrow-to-finish facility in New South Wales, Australia, during a 12-month period. Numbers of non-culled (control) females were 492, 847, 644, 1132, and 263 in parities 0, 1, 2, 3-5, and ≥ 6, respectively. Numbers of culled sows were 528, 243, 161, 586, and 636, in parities 0, 1, 2, 3-5, and ≥ 6, respectively. Body weight increased with increasing parity (P < .05; ANOVA), but backfat did not (P > .05). 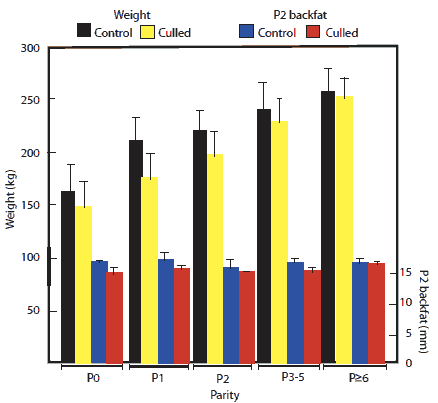 |
Reason for culling
Of the 2154 gilts and sows removed from the herd, the reason for removal and parity at removal are shown in Table 1. The single largest cause for culling was poor fertility associated with estrus or failure to become pregnant or maintain pregnancy. Of the 924 culls in this category, 395 (42.7%) were gilts, and 147 (15.9%), 83 (9.0%), 233 (25.2%), and 66 (7.1%) were from parities 1, 2, 3 to 5, and ≥ 6, respectively. Within the gilt subpopulation of reproductive culls, 166 (42.0%) failed to exhibit pubertal estrus, 44 (11.1%) were for second returns (presumed failure to conceive), 97 (24.6%) were for pregnancy test negative, 44 (11.1%) aborted, 22 (5.6%) were not-in-pig, and 22 (5.6%) had vulval discharges.
Table 1: Reasons for culling of gilts and sows during a 12-month period from a 5400-sow commercial farrow-to-finish facility in New South Wales, Australia
* Means ± SD. † Anestrus, vulva discharge, second return to estrus, pregnancy test negative, not-in-pig, abortion. ‡ High stillbirths, repeated small litter size, low milk yield. § Poor body condition, udder problems. |
For the 1710 non-age-related culls, 528 (29.8%) were gilts, 242 (14.2%) were primiparous sows, and 159 (9.3%) were second-parity sows. Within the gilt subpopulation of these females, 395 (74.8%) were for reproductive reasons, 115 (21.8%) were due to locomotor problems, and 18 were culled for other reasons. Of the 263 females culled for locomotor problems, 116 (44.1%) were gilts, and 53 (20.2%), 27 (10.3%), 49 (18.6%), and 18 (6.8%) were from parities 1, 2, 3 to 5, and ≥ 6, respectively.
Discussion
The basic hypothesis underlying the current study was that the body condition of gilts and sows being culled from the breeding herd would be poorer than that of contemporary gilts and sows remaining in the herd. This hypothesis is supported, since across all parities, sow body weights and backfat depths were lower for females culled from the herd than for sows that remained in the herd. The mean P2 backfat depths for the cull and control sows herd were 14.4 and 15.4 mm, respectively. The mean backfat depths for sows on this farm indicate that sows were very lean, suggesting that all parities were in a constant marginal state with respect to body condition. This is supported by the difference in mean backfat depths between culls and controls being small and thus the difference between those that fell over the culling “tipping point” and those that remained was minimal. Interestingly, while 35.7% of cull sows had P2 depths of ≤ 12 mm, only 25.5% of control sows were that thin. These data further support an association between low lactation feed intake and higher culling rates, as suggested by a retrospective records analysis by previous authors.13 Indeed, it is well established that inadequate nutrient intake during lactation will adversely affect sow fertility after weaning,4,5 and a weaning-sow backfat depth of ≤ 12 mm has previously been associated with inferior sow longevity.15 Together, these data suggest that we need to re-evaluate feeding strategies for modern breeding herds, particularly the present herd, as current strategies clearly fail to maintain the herd in reasonable body-condition status. The variation in sow backfat depths within the herd also indicates that the problem is not solely one of general feeding levels, but also reflects a failure to adequately account for individual sow needs.
The overall pattern of sow culling observed in this study was similar to those in previous reports16-21 and demonstrates that premature culling is a widespread problem, with only about 20% of sows reaching a parity at which a conscious decision was made to cull on the basis of likely reduction in performance if the sow was maintained in the herd. As expected, and consistent with earlier reports, aspects of reproductive performance constituted the primary reason for early culling of sows. Data reviewed by others demonstrated that culling for reproductive reasons occurred primarily in the younger parities (mean parity at removal of 2.7).3 The data presented from this study also indicate a relatively low parity at removal for reproductive (fertility) reasons (1.8), but relatively few of the culls were from parities 1 and 2. Indeed, on our farm, the female most likely to be removed for reproductive reasons was the gilt. Furthermore, gilts represented the major parity culled for locomotor problems (43.7% of locomotor culls) and gilts represented 31.8% of all non-age-related culls.
Overall, what the present data suggest is that on this farm, body condition may be one of several contributing factors to early sow culling. The major over-representation of gilts within the culled population strongly indicates a need for a renewed focus on gilt management and welfare. Such a focus would likely be acknowledged with increased sow longevity. In particular, given the association between high growth rates and locomotor problems,22,23 and that in the present study locomotor problems were largely associated with gilts, a restrictive growth curve may be more appropriate. However, when growth restriction should be imposed and for how long remains to be determined. Current commercial advice suggests that the breeding of gilts should be delayed in order to allow them to acquire sufficient body reserves to buffer the demands of their first lactation and so promote longevity in the herd. In contrast, there is a wealth of information suggesting that sow longevity is promoted by earlier breeding,24-26 with the optimal breeding age being approximately 210 days.27,28 In common with these latter authors, we conclude from the present data that in order to promote a longer productive herd life, the focus needs to be on gilt management, in particular the stimulation of puberty onset and breeding management to improve pregnancy and farrowing rates. The conflicting requirements for a period of feed restriction to minimize locomotor problems, earlier breeding, and the achievement of an adequate lean tissue reserve at first farrowing could be addressed by changes in pregnancy feeding management. The applicability of our conclusions regarding this farm to the wider swine industry is not established. However, for this farm, further studies are ongoing to determine effects on retention of increased levels of backfat and body mass of gilts at breeding and of providing increased dietary protein to younger sows.
Implications
- The present data indicate that for this farm, the greatest risk for culling was for gilts followed by younger sows.
- Where a case of high culling exists, the management of these populations is likely the priority.
Acknowledgements
The authors thank Australian Pork Limited for their financial support during this study.
References
1. Australian Pig Annual. PO Box 148, Deakin West, ACT 2600 Canberra, Australia: Australian Pork Ltd; 2005
2. Hughes PE, Varley MA. Lifetime performance of the sow. In: Wiseman J, Varley MA, Kemp B, eds. Perspectives in Pig Science. Nottingham, UK: Nottingham University Press; 2003:333–355.
3. D’Allaire S, Drolet R. Culling and mortality in breeding animals. In: Straw BE, D’Allaire S, Mengeling WL, Taylor DJ, eds. Diseases of Swine. 8th ed. Ames, Iowa: Iowa State University Press; 1999:1003–1016.
4. Aherne FX, Foxcroft GR, Pettigrew JE. Nutrition of the sow. In: Straw BE, D’Allaire S, Mengeling WL, Taylor DJ, eds. Diseases of Swine. 8th ed. Ames, Iowa: Iowa State University Press; 1999:1029–1043.
5. Aherne FX, Kirkwood RN. Nutrition and sow prolificacy. J Reprod Fert. 1985;(Suppl)33:169–183.
6. Kirkwood RN, Baidoo SK, Aherne FX, Sather AP. The influence of feeding level during lactation on the occurrence and endocrinology of the post weaning estrus in sows. Can J Anim Sci. 1987;67:405–415.
7. Clowes EJ, Aherne FX, Foxcroft GR, Baracos VE. Selective protein loss in lactating sows is associated with reduced litter growth and ovarian function. J Anim Sci. 2003;81:753–764.
8. Hughes PE. The effect of food level during lactation and early gestation on the reproductive performance of mature sows. Anim Prod. 1993;57:437–445.
9. Vesseur PC, Kemp B, Den Hartog LA. The effect of the weaning
to oestrus interval on litter size, live born piglets and farrowing
rate in sows.
J Anim Physiol Anim Nutr. 1994;71:30–38.
10. Steverink DW, Soede NM, Groenland GJ, van Schie FW, Noordhuizen JP, Kemp B. Duration of estrus in relation to reproductive results in pigs on commercial farms. J Anim Sci. 1999;77:801–809.
11. Tantasuparuk W, Lundeheim N, Dalin AM, Kunavongkrit A, Einarsson S. Weaning-to-service interval in primiparous sows and its relationship with longevity and piglet production. Livest Prod Sci. 2001;69:155–162.
12. Edwards SA. Nutrition of the rearing gilt and sow. In: Wiseman J, Varley MA, Chadwick JP, eds. Perspectives in Pig Science. Nottingham, UK: Nottingham University Press; 1998:361–382.
13. Koketsu Y, Dial GD, King VL. Returns to service after mating and removal of sows for reproductive reasons from commercial swine farms. Theriogenology. 1997;47:1347–1363.
14. Close WH, Cole DJA. Nutrition of Sows and Boars. Nottingham, UK: Nottingham University Press; 2000:365.
15. Young LG, King GJ, Shaw J, Quinton M, Walton JS, McMillan I. Interrelationships among age, body weight, backfat and lactation feed intake with reproductive performance and longevity of sows. Can J Anim Sci. 1991;71:567–575.
16. Dagorn J, Aumaitre A. Sow culling: reasons for and effect on productivity. Livest Prod Sci. 1979;6:167–177.
*17. Stone MW. Sow culling survey in Alberta [letter]. Can Vet J. 1981;22:365.
18. Friendship RM, Wilson MR, Almond GW, McMillan I, Hacker RR, Pieper R, Swaminathan SS. Sow wastage: reasons for and effect on productivity. Can J Vet Res. 1986;50:205–208.
19. D’Allaire S, Stein TE, Leman AD. Culling patterns in selected Minnesota swine breeding herds. Can J Vet Res. 1987;51:506–512.
20. Dijkhuizen AA, Krabbenborg EMM, Huirne RBM. Sow replacement: A comparison of farmers’ actual decisions and model recommendations. Livest Prod Sci. 1989;23:207–218.
21. Stein TE, Dijkhuizen AA, D’Allaire S, Morris RS. Sow culling and mortality in commercial swine breeding herds. Prev Vet Med. 1990;9:85–94.
22. Jorgensen B. Effect of different energy and protein levels on leg weakness and osteochondrosis in pigs. Livest Prod Sci. 1995;41:171–181.
23. Jorgensen B, Sorensen MT. Different rearing intensities of gilts: II. Effects on subsequent leg weakness and longevity. Livest Prod Sci. 1998;54:167–171.
24. Young LG, King GJ, Walton JS, McMillan I, Klevorick M. Reproductive performance over four parities of gilts stimulated to early estrus and mated at first, second, or third observed estrus. Can J Anim Sci. 1990;70:483–492.
25. Rozeboom DW, Pettigrew JE, Moser RL, Cornelius SG, el Kandelgy SM. Influence of gilt age and body composition at first breeding on sow reproductive performance and longevity. J Anim Sci. 1996;74:138–150.
26. Le Cozler Y, Dagorn J, Lindberg JE, Aumaitre A, Dourmad JY. Effect of age at first farrowing and herd management on long-term productivity of sows. Livest Prod Sci. 1998;53:135–142.
27. Schukken YH, Buurman J, Huirne RBM, Willemse AH, Vernooy JCM, van den Broek J, Verheijden JHM. Evaluation of optimal age at first conception in gilts from data collected in commercial swine herds. J Anim Sci. 1994;72:1387–1392.
28. Serenius T, Stalder KJ. Length of productive life of crossbred sows is affected by farm management, leg conformation, sow’s own prolificacy, sow’s origin parity and genetics. Animal. 2007;1:745–750.
*Non-referred reference.
