| Case report | Peer reviewed |
SummaryThis study demonstrated a clear benefit in vaccinating pigs subclinically infected with porcine circovirus type 2 (PCV2) with a one-dose PCV2 vaccine. At weaning (3 weeks of age), 1427 pigs were vaccinated and 1431 were sham-inoculated with a placebo (0.9% physiological saline). After 33 days in the nursery, 528 pigs from each treatment group were moved into 48 pens in a commercial finisher barn, ensuring equal age of pigs across both treatments. Each pig was weighed on day 2 in the finisher barn and on day 88 (end of the study period). The vaccinates had a 36-g-per-day advantage in daily gain and 4.6% less mortality than the unvaccinated pigs. More than twice as many vaccinates as unvaccinated pigs (40 versus 16, respectively) were marketed at the first shipment. Furthermore, carcass weight (95.0 kg versus 94.0 kg; P < .05), lean percent (60.52% versus 60.26%; P < .05) and carcass index (111.6 versus 111.1; P < .05) were all greater for the vaccinates. The mean loin depth was 65.1 mm for vaccinates and 63.3 mm for unvaccinated pigs (P < .05). The vaccinated pigs delivered a return on investment of $5.90 per pig over the unvaccinated pigs. | ResumenEste estudio demuestra un beneficio claro al vacunar cerdos infectados subclínicamente con circovirus porcino tipo 2 (PCV2 por sus siglas en inglés) con una vacuna de dosis única de PCV2. Al destete (3 semanas de edad), se vacunaron 1427 cerdos y se simuló la vacunación con un placebo (solución salina al 0.9%) a 1431 cerdos. Después de 33 días en el destete, 528 cerdos de cada tratamiento se movieron a 48 corrales de un sitio de finalización comercial, manteniéndose la misma edad en los cerdos en ambos tratamientos. Se pesó cada cerdo en el día 2 en la finalización y en el día 88 (fin del estudio). Los cerdos vacunados tuvieron una ventaja de 36 g por día en la ganancia diaria y 4.6% menos mortalidad que los cerdos no vacunados. Más del doble de los cerdos vacunados que de los no vacunados (40 contra 16, respectivamente) fueron vendidos en el primer embarque. Además, el peso de la canal (95.0 kg contra 94.0 kg; P < .05), el porcentaje de magro (60.52% contra 60.26%; P < .05), y el índice de canal (111.6 contra 111.1; P < .05) fueron mayores en los cerdos vacunados. La profundidad media de lomo fue de 65.1 mm para los cerdos vacunados y de 63.3 mm en los cerdos no vacunados (P < .05). Los cerdos vacunados produjeron un retorno a la inversión de $5.90 por cerdo comparados con los cerdos no vacunados . | ResuméCette étude a permis de démontrer clairement le bénéfice de vacciner les porcs infectés de manière sub-clinique avec le circovirus porcin de type 2 (PCV2) à l’aide d’un vaccin anti-PCV2 à dose unique. Au sevrage (3 semaines d’âge), 1427 porcs ont été vaccinés et 1431 ont été inoculés avec un placebo (saline physiologique à 0.9%). Après 33 jours dans la pouponnière, 528 porcs de chaque groupe de traitement ont été déménagés dans 48 enclos dans une ferme de finition commerciale, en s’assurant d’avoir une uniformité de l’âge des porcs parmi les deux groupes de traitement. Chaque porc a été pesé au jour 2 dans la ferme de finition et au jour 88 (à la fin de la période d’étude). Comparativement aux animaux non-vaccinés, les animaux vaccinés ont eu un gain de poids quotidien supérieur de 36 g par jour et 4.6% moins de mortalité. Plus du double des animaux vaccinés que de non-vaccinés (40 versus 16, respectivement) ont été mis en marché au moment de la première expédition d’animaux. De plus, le poids carcasse (95.0 kg vs 94.0 kg; P < .05), le pourcentage de maigre (60.52% vs 60.26%; P < .05), et l’indice de carcasse (111.6 vs 111.1; P < .05) étaient tous supérieurs pour les animaux vaccinés. La profondeur moyenne de la longe était de 65.1 mm pour les vaccinés et de 63.3 mm pour les porcs non-vaccinés (P < .05). Les porcs vaccinés ont permis un retour sur l’investissement supplémentaire de $5.90 par porc comparativement aux porcs non-vaccinés. |
Keywords: swine, porcine circovirus type 2 vaccine, subclinical infection, carcass benefits, return on investment, PCV2, ROI
Search the AASV web site
for pages with similar keywords.
Received: May 15, 2009
Accepted: January 20, 2010
Porcine circovirus type 2 (PCV2) is a small, non-enveloped, single-stranded DNA virus with a circular genome.1,2 It is the necessary agent in causing postweaning multisystemic wasting syndrome (PMWS) and may be associated with several other disease syndromes in pigs, including proliferative and necrotizing pneumonia, porcine dermatitis and nephropathy syndrome, and reproductive failure.1-3 Diseases linked to PCV2 are now called porcine circovirus diseases in Europe or porcine circovirus associated disease (PCVAD) in North America.2
Since its first description as PMWS in the mid-1990s in Western Canada,4,5 the disease was seen sporadically until the fall of 2004, when epidemic outbreaks erupted in Quebec and Ontario, and subsequently across most of North America. As familiarity with the disease grew, it became clear that infection with PCV2 is ubiquitous in herds and pigs worldwide, with almost every pig becoming infected with the virus some time during its growing period. Yet, although virtually all pigs become infected with PCV2, in many infected herds there is no clinical evidence of PCVAD. During 2006, as the PCVAD epidemic escalated across North America, PCV2 vaccines became available for growing pigs in Canada and the United States. These vaccines were not only immediately successful in curbing the severe mortality associated with PCVAD, but it soon became evident from field observations that vaccinating pigs against PCVAD often resulted in productivity improvements over and above the levels that were present before PCVAD appeared clinically in the herd, clearly suggesting that PCV2 vaccination was neutralizing or overcoming some hitherto unrecognized, potentially deleterious effects of PCV2 infection.
The purpose of this study was to determine the biological and economic impact of a one-dose PCV2 vaccine in growing pigs in a herd subclinically infected with PCV2.
Case description
Statement of care of study animals
The system was managed with regard to the Canadian code of practice for the care and handling of pigs.6 This study was conducted in a research barn that conducts research trials for the University of Alberta and the Government of Alberta, and the study conformed to the standard operating protocol approved by the university. The facilities were inspected and approved by the university personnel.
Production system
Crossbred pigs ([Landrace × Large White dams] × Duroc boars) used in this study came from a single-source, high-health, 2850-sow multi-site system without clinical signs of PCVAD.
Vaccination and pen allocation
Each week, prior to placement in the farrowing room, sows due to farrow that week were sorted by parity, and each was then allotted to one of two treatment groups, Vaccine or Placebo, ensuring that numbers were equal for the two groups. Each sow’s identification card was marked yellow or white to indicate the tag color with which her pigs would be tagged. When each sow finished farrowing, and prior to cross fostering, all live pigs born in the litter were uniquely identified with a yellow or white tag depending on the designated color on the sow card.
Each treatment included equal numbers of barrows and gilts. Pigs to be vaccinated were notched in the left ear. Pigs were allotted to treatment by sow parity, which ranged from 2nd to 10th, to remove the impact of parity. In the nursery and research finisher barns, pigs were blocked by treatment within room to remove the impact of barn or environment. On entry into the research barn, pigs were blocked by treatment, gender, and weight to keep each treatment group within block (two pens) as alike as possible, ensuring that if there were differences in biological performance, they would be only treatment related. Feed and genetic background were the same for both treatments.
A total of 2858 farrowed pigs born within 2 weeks of each other were weaned into the nursery at approximately 3 weeks of age. Weaning occurred twice a week, with half of the sows in a farrowing group weaned on Tuesday and the remainder on Thursday. On entry into the nursery, pigs weaned on each day were housed in a single room, with pigs penned within room by tag color and gender. At weaning, pigs were injected with 1 mL of either Ingelvac CircoFlex vaccine (Boehringer Ingelheim Canada Ltd, Burlington, Ontario; Vaccinates) or a placebo (0.9% physiological saline; Vétoquinol Canada Ltd, Lavaltrie, Québec; Controls). Pigs were weighed on the day of weaning and 33 days later. All study pigs were fed the same diet.
After 33 days in the nursery, 1056 pigs, 528 from each treatment group, were moved into a commercial finisher research barn, ensuring age of pigs was equal across both treatments. Pigs selected at the nursery were the oldest pigs to be shipped to the finisher barn. Each nursery room had a capacity for 750 pigs, thus one and a half nursery rooms were used to fill the trial room at the grow-finish barn. Equal numbers of pigs from both treatments were selected from the second room to ensure pig age was constant across treatment.
The research barn consisted of two rooms containing 52 pens per room. One room was used for this study, with study pigs placed in 48 pens and the four corner pens left vacant for pigs removed from the study. Each room was divided by a center alley. The rectangular pens (6.1 m × 2.4 m) allowed 0.7 m2 space per pig, with 22 pigs per pen. Each pen was equipped with a nose-to-nose wet-dry stainless steel feeder (Crystal Springs Colony, Magrath, Manitoba) with two feeding spaces. An additional water source was available from a bowl drinker located at the side wall of each pen.
Prior to the pigs’ entry into the finishing barn, contiguous pairs of pens were assigned to Vaccinate and Control groups. On arrival at the barn, pigs were initially placed in pens by tag color and gender without sorting by weight. Pens of pigs were then weighed on the same day of loading on a pen scale accurate to 1 kg. Pigs were also weighed individually on an individual pig scale accurate to 0.1 kg on days 2 and 88 in the finisher barn. Feed intake was measured bi-weekly in the finishing barn until marketing, and after the pigs had been weighed on the day of entry into the finishing barn, pens of pigs were blocked by weight. All pigs were fed the same diet from day of entry until marketing, consisting of Phase 1 (50 kg per pig), Phase 2 (55 kg per pig), Phase 3 (70 kg per pig), and Phase 4 (90 kg per pig). Feed was delivered using a computerized robotic feed delivery and weighing system (FeedLogic Corporation Inc, Willmar, Minnesota), which allowed compilation of feed data by pen.
Measurements and statistical analysis
The pen was the experimental unit and the basis for all measurements. Measurements included average daily gain (ADG), average daily feed intake (ADFI), total weight gain, feed conversion, mortality, and total removals for each 2-week period. The ADG was calculated as (pen weight at end of the study period) - (pen weight at the beginning of the study period) ÷ (pig days). Pig days were calculated as (end day in the study period) - (start day in the study period) × (pig inventory in the pen). If a pig or pigs were removed during the period, then the days they were in the pen were added back into the pig days. Feed cost per kg of gain and income over feed cost (IOFC) were calculated for the overall study period, with IOFC defined as the value of the weight gained less the feed cost incurred during the study period. The value of the gain was calculated as weight gain during the study × dressing (79%) × index × $1.00 per kg (all currency in $CAN). Feed cost per pig was calculated as (weight gain during the study period) × (feed cost per kg of gain). The return on investment (ROI) was calculated as ROI = IOFC ([ADG × 97 days × 79% × Index × $1.00 per kg] - [ADG × 97 days × Feed cost per kg gain]) + $1.00 per pig per 1% mortality - vaccine cost (market value per dose at study initiation + cost of injection).
Data were analyzed using analysis of variance (ANOVA) with the MIXED procedure of SAS (SAS Institute, Inc, Cary, North Carolina), with pen the experimental unit for all data. Count data, including the number of dead and number removed from each pen, were analyzed using generalized estimating equations with a poisson distribution, log link function, a repeated term to account for clustering of outcomes within block, and an exchangeable correlation structure (PROC GENMOD, SAS version 9.2, SAS for Windows; SAS Institute, Inc). Results were reported as the expected counts in each group with 95% CI, and relative difference in expected counts between treatment groups with 95% CI. Differences were considered significant at P < .05. Carcass data were collected from the slaughter plant. Carcass weight was used as a covariate in the analysis of carcass data. A standard lean yield equation is used across Canada, consisting of lean yield = 68.1863 - (0.7833 × fat mm) + (0.0689 × muscle mm) + (0.0080 × (fat mm × fat mm)) - (0.0002 × (muscle mm × muscle mm)) + (0.0006 × (fat mm × muscle mm)). Carcass index is based on an average of 1.125 for 61% lean yield average and adjusted up and down depending on the lean yield percentage.
Virological and serological investigation
Blood samples were collected from a convenience sample of 50 pigs, 25 of each tag color, on the day of vaccination at 3 weeks of age. Blood-sampled pigs were tagged with a second ear tag of a different color for future identification, and the same pigs were re-sampled at 7, 13, and 17 weeks of age. Serum samples were frozen and stored until completion of the study. Sera from 14 Controls (randomly selected by a technician blinded to treatment) were collected 4.5 weeks post placement in the finisher, when the pigs were 13 weeks of age. Samples were tested at the Iowa State University Veterinary Diagnostic Laboratory (ISU VDL)7 by quantitative polymerase chain reaction (qPCR) to determine log-transformed titers to PCV2, in order to confirm natural challenge. The qPCR results are reported as log10 PCV2 genomic copies per mL of serum. ELISA testing was conducted on all sera at the ISU VDL to detect antibody to PCV2. ELISA results were analyzed at each time point using one-way analysis of variance (SPSS 15; SPSS Inc, Chicago, Illinois), and average sample-to-positive ratio (S:P) at each time point was reported for Vaccinates and Controls.
Response to vaccination
At the end of the nursery phase, there were no significant differences in body weight (20.9 kg versus 20.7 kg; P > .05), ADG (467.8 g per day versus 456 g per day; P > .05), or mortality (0.49% versus 0.21%; P > .05) for Vaccinates and Controls, respectively.
Table 1 shows the ADG, ADFI, mortality, and feed-to-gain ratios for Vaccinates and Controls in 27- to 28-day intervals over the first 84 days in the finishing barn.
Table 1: Impact of PCV2 vaccination on biological and economic live-animal performance of finisher pigs*
* A total of 1056 animals housed 22 per pen with 24 treatment replications and 528 pigs per treatment. Pigs were injected at weaning (approximately 3 weeks of age) either with a porcine circovirus type 2 vaccine (Vaccinates) or with physiological saline (Controls). An outbreak of ileitis affecting both treatment groups occurred between days 57 and 70; all pigs were treated with in-feed tylosin (220 mg/kg feed) for 14 days. † IOFC = ([ADG × 97 days × 79% × Index × $1/kg] – [ADG × 97 days × Feed cost/kg gain]). ‡ Removals and death loss: pen level expected count data and 95% confidence interval (CI) converted to a percentage. Removal reasons included sudden death, Streptococcus suis infection, chronic lameness, and poor doer unrelated to porcine circovirus associated disease. PCV2 = porcine circovirus type 2; SED = standard error of difference; ADG = average daily gain; ADFI = average daily feed intake; IOFC = income over feed cost |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
On day 88 in the finishing barn, just before the first batch of heaviest pigs were marketed and the last time all pigs were weighed individually, average weight was 107.3 kg for the Vaccinates and 104.8 kg for the Controls (P < .01) (Figure 1). Moreover, the Vaccinates had numerically greater ADGs than the Controls in each 2-week period measured in the finishing barn (Figure 2). During the period between days 57 and 70, an outbreak of clinical ileitis occurred and all pigs were treated for 14 days with in-feed tylosin at 220 mg per kg of feed (Tylosin phosphate; Bio Agri Mix, Mitchell, Ontario). Ileitis was confirmed by histological examination of tissues submitted to the Prairie Diagnostic Centre Laboratory at the University of Saskatchewan, Saskatoon, Saskatchewan, Canada, and by a positive PCR test for Lawsonia intracellularis on intestinal sections. Growth rate slowed for both groups over this 2-week period and returned to earlier levels in the subsequent 2 weeks (Figure 2). During the ileitis outbreak, 12 of the original 528 Controls (2.27%) and two of the 528 Vaccinates (0.38%) died (P < .01). Over the entire study period, total pig removals were greater for the Controls (11%) than for Vaccinates (7%), with a significant relative difference in expected counts of 1.57 (P < .05). Mortality was also greater in the Controls (7.2%) than the Vaccinates (2.6%), with a significant relative difference in expected counts of 2.79 (P < .01; Table 1). Most of the difference in mortality occurred between days 15 and 70, when 1% to 2% of Controls died for each 2-week period, whereas for most of this period there were few or no deaths among the Vaccinates. Reasons for pig removals were sudden death, clinical illness consistent with Streptococcus suis infection, chronic lameness, and nonspecific ill-thrift.
Figure 1: Distribution of weights in pigs individually weighed on day 88 in a commercial finisher barn. Pigs had been injected at weaning (approximately 3 weeks of age) with either a porcine circovirus type 2 vaccine (Vaccinates) or with physiological saline (Controls). After 33 days in the nursery, 528 pigs from each treatment group were moved into the finisher. On day 88 the average weight of the Vaccinates was greater than that of the Controls (P < .01). Data were analyzed using ANOVA with the MIXED procedure of SAS (SAS Institute, Inc, Cary, North Carolina). 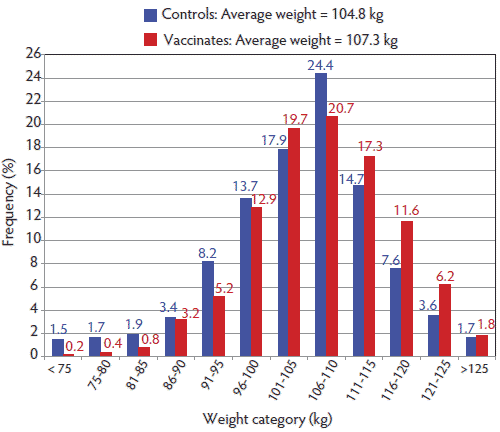 |
Figure 2: Bi-weekly growth rate of finisher pigs injected at weaning (approximately 3 weeks of age) with either a porcine circovirus type 2 vaccine (Vaccinates; 528 pigs) or physiological saline (Controls; 528 pigs). An outbreak of clinical ileitis occurred between days 57 and 70 in the finisher; all pigs were treated with tylosin at 220 g/kg of feed for 14 days. Growth rate was greater (P < .01) for the Vaccinates than the Controls for days 0-14 and 15-28, showed a trend (P < .10) in favor of the Vaccinates during days 29-56, and was not significant (P > .05) for the final 28 days. ADG was greater (P < .01) for the Vaccinates than the Controls over the entire study period. Data were analyzed using ANOVA with the MIXED procedure of SAS (SAS Institute, Inc., Cary, North Carolina). 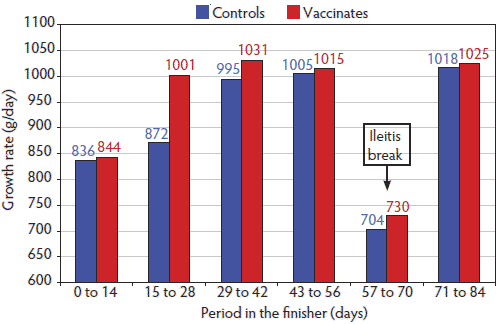 |
More Vaccinates (40; 7.5% of the population) than Controls (16; 3% of the population) had reached market weight and were shipped on the first day of marketing after the last weighing on day 88. As the Vaccinates were significantly heavier than the Controls (P < .01), by day 88 there were numerically fewer Vaccinates (9.8%) than Controls (16.7%) weighing < 95 kg and numerically more Vaccinates (19.6%) than Controls (12.9%) weighing > 115 kg, the target market weight (Figure 1). The coefficient of variation in individual pig weights decreased for Controls between day 2 and day 88 in the finisher from 11.6% to 9.2% and for Vaccinates from 12.0% to 8.4%, a difference that was not statistically significant (P > .05).
Carcass weight (95.0 kg versus 94.0 kg;
P < .05), lean percent (60.52% versus 60.26%; P < .05), and carcass index (111.6 versus 111.1; P < .05) were all greater for the Vaccinates. Mean loin depth was 65.1 mm for Vaccinates and 63.3 mm for Controls (P < .05).
Virological and serological results
In 11 of the 14 sera collected from the Control pigs 4.5 weeks post placement, qPCR titers for PCV2 ranged from 1.72 × log10 PCV2 genomic copies per mL of serum to 3.50 × log10 PCV2 genomic copies per mL of serum, thereby confirming low levels of PCV2 infection in the Controls.8 Titers of the remaining three sera were 4.05, 4.44, and 6.65 × log10 PCV2 genomic copies per mL of serum. The ELISA S:P ratios at 3, 7, 13, and 17 weeks post vaccination were 0.81 versus 0.82, 0.44 versus 0.46, 0.70 versus 0.80, and 0.90 versus 0.87 for Controls and Vaccinates, respectively, showing a trend (P < .10) towards slightly earlier increases in S:P ratios and numerically slightly higher S:P ratios for Vaccinates 4.5 weeks post placement in the finisher barn, when some Control pigs were already PCR-positive.
The IOFC was greater (P < .01) per pig marketed by $1.20 for the Vaccinates. Each 1% death loss was calculated as equating to a net loss of $1.00 per pig. Combining mortality and IOFC (growth and grading advantage), the additional ROI for the PCV2 Vaccinates over the Controls in this study was $5.90 per pig.
Discussion
The purpose of this study was to determine the biological and economic impact of vaccinating growing pigs with a one-dose PCV2 vaccine in a herd subclinically infected with PCV2. This high-health commercial production herd had a baseline finisher mortality of approximately 2% and no clinical signs of PCVAD, but historically experienced surges in mortality that approached 6% due to sporadic S suis and Haemophilus parasuis outbreaks. Over the years, clinical signs and necropsy results did not include a diagnosis of PCVAD. Nevertheless, in this study, pigs vaccinated with a single dose of PCV2 vaccine had a rate of growth 36 g per day greater than that of controls, and 4.6% lower mortality.
The better production parameters for the vaccinated pigs clearly indicate that, although there was no obvious clinical evidence of PCVAD in the herd, PCV2 infection did have a negative impact on the biological functioning of these apparently normal growing pigs. The daily gain of 904 g per day by the unvaccinated controls makes their growth rate comparable to the expected growth rate of normal, uncompromised, fast-growing pigs on diets fed in Western Canada.9
An unexpected clinical ileitis outbreak occurred during days 57 to 70 in the finishing barn. Immediate antibiotic treatment instituted by the herd veterinarian probably mitigated more severe losses. Nevertheless, there was a 30% decrease in growth rate observed for both the Vaccinates and Controls during this 14-day time span. Mortality among the Controls during this period was more than five times that of the Vaccinates. The reason for the Lawsonia-associated mortality being higher in the Controls is unknown, but it is likely not happenstance that the PCV2-vaccinated pigs were less severely affected by the ileitis outbreak. It would seem worthwhile to determine if this phenomenon is repeatable, and if so, to determine what causes PCV2-vaccinated, subclinically infected pigs to be less severely impacted by L intracellularis infection.
Mortality and total pig removals were greater for the unvaccinated pigs, even when the surge in deaths during the ileitis break is taken out of the calculations. At each 2-week observation period, mortality and total removals were numerically greater for unvaccinated pigs. Reasons for removals were again due to signs indicative of S suis infection, as was historically the case for this herd. Nevertheless, the vaccinated pigs fared better than their unvaccinated cohorts, again raising the question about previously unrecognized compromise that might result in the pigs subclinically infected with PCV2 having less ability to combat endemic bacterial infections. Removing or reducing the impact of the PCV2 infection by vaccination may have left the pigs better prepared to ward off these endemic infections.
Testing of sera by qPCR confirmed very low levels of PCV2 infection in 11 of 14 Controls. That notwithstanding, the Vaccinates housed in the same barn as the Controls were expected to have similar temporal exposure to PCV2, but the S:P ratios showed a trend towards earlier development in the Vaccinates than in the Controls, indicating that even under conditions of very low PCV2 challenge, the vaccinated pigs tended to respond faster to the PCV2 challenge.
Better overall carcass characteristics, including lean percent, carcass index, and loin depth, were achieved by the vaccinated pigs than by their unvaccinated cohorts. Similar findings have since been reported in at least two other studies conducted in pigs subclinically infected with PCV2 in the United States.10,11 These superior carcass characteristics were unexpected. It is clear, however, from results being reported from a growing number of independent studies,10-14 that superior carcass characteristics in pigs vaccinated for PCV2 are repeatable across different types of herds, under different management conditions, and in different countries.
Finally, heavier weight at marketing and lower mortality, combined with the several carcass characteristics, all in favor of the vaccinated pigs, calculated out to be a return of $5.90 per pig advantage over their unvaccinated cohorts.
Implications
• There are possible economic benefits to vaccinating pigs with PCV2 vaccines in the absence of clinical signs of PCVAD.
• Since virtually all pigs worldwide are infected with PCV2 at some time during their growing period, it is not unreasonable to expect that most would benefit from PCV2 vaccination.
• The benefits of PCV2 vaccination in subclinically infected herds include heavier carcass weight and larger loin depth, lean percentage, and carcass index.
Acknowledgements
We would like to express thanks to the Lewisville Pork Farm management and staff, the Drumloche research barn management team for their expertise in animal care and husbandry and pig weighing, the Lewisville Pork farm for their use of the animals, Sunhaven Farms Milling for feed supply, and Britco Pork for providing the carcass grading data. This study was funded by Boehringer Ingelheim.
References
1. Allan G, Ellis J. Porcine circoviruses: a review. J Vet Diagn Invest. 2005;12:3–14.
2. Opriessnig T, Meng XJ, Halbur PG. Porcine circovirus type 2-associated disease: Update on current terminology, clinical manifestations, pathogenesis, diagnosis and intervention strategies. J Vet Diagn Invest. 2007;19:591–615.
3. Segalés J, Domingo M. Post-weaning multisystemic wasting syndrome (PMWS) in pigs. A review. Vet Q. 2002;24:109–124.
*4. Harding J. Post weaning multisystemic wasting syndrome (PMWS): Preliminary epidemiology and clinical findings. Proc West Can Assoc Swine Pract. Saskatoon, Saskatchewan. 1996;21.
*5. Clark E. Pathology of post-weaning multisystemic wasting syndrome (PMWS) of pigs. Proc West Can Assoc Swine Pract. Saskatoon, Saskatchewan. 1996;22–25.
6. Agriculture Canada. Recommended code of practice for care and handling of pigs. Communications Branch, Agriculture Canada, Ottawa, Ontario, Canada. 1984. Publication 1771/E.
7. Fenaux M, Halbur PG, Haqshenes G, Royer R, Thomas P, Nawagitgul P, Gill M, Toth TE, Meng XJ. Cloned genomic DNA porcine circovirus infections when injected directly into the liver and lymph nodes of pigs: characterization of clinical disease, virus distribution and pathological distribution. J Virol. 2002;76:541–551.
8. Brumborg IM, Modal T, Jonassen CM. Quantification of porcine circovirus type 2 isolated from serum/plasma and tissue samples of healthy pigs and pigs with postweaning multisystemic wasting syndrome using a TaqMan-based real-time PCR. J Virol Methods. 2004;122:171–172.
*9. Zijlstra RT, Beltranena E. Regaining competitiveness: alternative feedstuffs for swine. Adv Pork Prod. 2009;20:237–243.
*10. King D, Dubois P, Painter T, Holck T, Edler R, Johnson C, Diaz E. Biologic and economic benefits of controlling subclinical PCVAD with PCV2 vaccination. Proc IPVS. Durban, South Africa. 2008;2:36.
*11. Bretey K, Diaz E. Efficacy of Ingelvac CircoFLEX in a herd with subclinical PCVAD. Leman Swine Conf. 2008;(suppl):11.
*12. Orveillon FX, Fachinger V, Denotte JP. Effect of PCV2 vaccination on growth performance in a subclinically affected herd. Leman Swine Conf. 2008;(suppl):14.
*13. Verbeek J, Waddell J, Baysinger A. Field evaluation of Ingelvac CircoFLEX. Proc AASV. Dallas, Texas. 2009;40:353–356.
*14. Gillespie T. Comparative performance of barns of pigs vaccinated or not vaccinated with a one-dose PCV2 vaccine. Proc IPVS. Durban, South Africa. 2008;2:35.
* Non-refereed references.
