| Original research | Peer reviewed |
Cite as: Lizer JT, Madson DM, Schwartz KJ, et al. Experimental infection of conventional neonatal pigs with Clostridium difficile: A new model. J Swine Health Prod. 2013;21(1):22–29.
Also available as a PDF.
SummaryObjective: To reproduce Clostridium difficile-associated disease (CDAD) using conventional pigs as an animal model. Materials and methods: Nineteen conventional piglets, removed from the sow immediately as they were being born, were enrolled in the study. Neonatal pigs were given pooled colostrum and then transported to a research facility. At approximately 4 hours of age, 13 pigs randomly assigned to treatment groups were each orogastrically inoculated with one of two different swine-origin Clostridium difficile field isolates, and six control pigs were sham-inoculated. All pigs were individually housed and randomly assigned to necropsy at 24, 48, or 72 hours post infection. Results: Commonly observed lesions and indications of CDAD, including mesocolonic edema, toxin detection, diarrhea, neutrophilic infiltration of the colonic and cecal lamina propria, and mucosal ulceration or erosion of the colon and cecum, were observed in challenged pigs. Implications: Data generated in this study provides evidence that this has potential to be an effective challenge model for CDAD. Possible uses for this model include studies of disease pathogenesis and intervention strategies. The described model can be adapted for in vivo studies of human-origin C difficile strains and therapies intended for use in human medicine. | ResumenObjetivo: Reproducir la enfermedad asociada a Clostridium difficile (CDAD por sus siglas en inglés) utilizando cerdos convencionales como un modelo animal. Materiales y métodos: Diecinueve lechones convencionales fueron retirados de la hembra inmediatamente después del nacimiento y fueron asignados a un estudio. Se les administró calostro mezclado a los cerdos recién nacidos y fueron transportaron a un centro de investigación. Aproximadamente a las 4 horas de edad, 13 cerdos, asignados al azar a grupos de tratamiento fueron inoculados de manera orogástrica con uno de dos diferentes aislados de campo de Clostridium difficile de origen porcino, y seis cerdos fueron asignados a un grupo control. Todos los cerdos fueron alojados individualmente y asignados al azar a necropsia a las 24, 48, o 72 horas post infección. Resultados: Se observaron lesiones comúnmente observadas en lesiones e indicaciones de CDAD, incluyendo edema mesocolónico, detección de toxina, diarrea, infiltración neutrofílica de la lámina propia cecal y colónica, y erosión ó ulceración de mucosa del colon y cecal, se observó en cerdos puestos a prueba. Implicaciones: La información generada en este estudio provee evidencia de que este modelo tiene potencial para CDAD. Los usos posibles para este modelo incluyen estudios de estrategias de intervención y la patogénesis de enfermedad. El modelo descrito puede adaptarse a estudios in vivo de cepas de C difficile de origen humano y terapias propuestas para uso en medicina humana. | ResuméObjectif: Reproduire la maladie associée à Clostridium difficile (CDAD) en utilisant des porcs conventionnels comme modèle animal. Matériels et méthodes: Dix-neuf porcelets conventionnels, retirés de leur mère immédiatement après la naissance, ont été incorporés à cette étude. Les porcs nouveau-nés ont reçu du colostrum combiné et par la suite ont été transportés à l’unité de recherche. À l’âge approximatif de 4 heures, 13 porcelets ont été assignés à des groupes de traitement et inoculés par voie orogastrique avec un des deux isolats porcins de champs, et six porcs témoins ont été faussement inoculés. Tous les porcs ont été logés individuellement et assignés de manière aléatoire à être soumis à une nécropsie 24, 48, ou 72 heures post-infection. Résultats: Les lésions observées couramment et les indications de CDAD, incluant l’œdème du mésocôlon, la détection de toxine, de la diarrhée, une infiltration neutrophilique de la lamina propria du colon et du cæcum, ainsi que des ulcérations et des érosions du côlon et du cæcum furent notés chez les porcs infectés. Implications: Les données obtenues dans la présente étude montrent le potentiel de ce modèle à être utilisé pour investiguer le CDAD. Les utilisations possibles de ce modèle incluent des études sur la pathogénèse de la maladie et des stratégies d’intervention. Le modèle décrit peut être adapté pour des études in vivo des souches de C difficile d’origine humaine ainsi que pour des thérapies destinées à utilisation en médecine humaine. |
Keywords: swine, Clostridium difficile, Clostridium difficile-associated disease, challenge model
Search the AASV web site
for pages with similar keywords.
Received: May 7, 2012
Accepted: June 21, 2012
Clostridium difficile is a spore-forming, gram-positive bacillus associated with diarrhea in neonatal pigs and other species. The disease caused by this bacterium has been described as possibly the most important uncontrolled cause of neonatal diarrhea in pigs.1 Clostridium difficile is a principal infectious cause of antibiotic-associated diarrhea in humans, in which antibiotic-mediated disruption of the normal gut flora results in unimpeded growth of C difficile upon its introduction to the gut.2
Over the last decade, the incidence of Clostridium difficile-associated disease (CDAD) has significantly increased, and the disease is now considered a major cause of neonatal diarrhea within the swine industry.1 Observations from multiple studies have documented C difficile toxin (Tcd) detection in 29% to 50% of tested piglets.1,3-5 Morbidity and mortality both average 20%, but mortality rates as high as 50% have been documented.6 A recent study by Hopman et al7 demonstrated that 100% of 71 piglets born to six sows were positive for C difficile within 48 hours after birth. Other studies have shown a significant decrease in C difficile colonization with age of pigs, with the greatest decline occurring over the first 2 months of life.8
Clostridium difficile-associated disease is most commonly observed in 1- to 7-day-old piglets. Described clinical signs may include dyspnea, mild abdominal distention, scrotal edema, and occasional diarrhea.6 Clinical diarrhea, however, is an inconsistent finding.3,4,9 Weaning weights in affected pigs are approximately 450 to 900 grams below average.1 Clostridium difficile may also be an important subclinical issue, as toxin can be detected in apparently healthy piglets.4 Microscopic lesions are limited to the cecal and colonic mucosae and consist of multifocal exudation of mucus, fibrin, and neutrophilic aggregates in the lamina propria, with additional accumulation of neutrophils in the colonic mesentery.2,4-6 The disease in pigs may be spontaneous, exclusive to neonates, and unassociated with antibiotic usage.4-6,9 Currently, treatment with antibiotics or probiotics has been unsatisfactory,6 and no licensed immunoprophylactic products are available for CDAD.10
The primary known virulence factors implicated in CDAD are two exotoxins, toxin A (TcdA) and toxin B (TcdB). Toxin A has mild cytotoxic activity, but is primarily an enterotoxin, causing fluid accumulation in the intestine, while TcdB is a potent cytotoxin.4,11-13 Both toxins induce inflammatory effects, including increased vascular permeability and production of proinflammatory cytokines, chemokines, and tumor necrosis factor-α.13,14
Experimentally induced disease has been described in hamsters, mice, rats, and rabbits,2,15 but the importance of this disease in pigs necessitates a species-specific model. A recent study published during the preparation of this manuscript described a challenge model using gnotobiotic pigs.16 While one benefit of using gnotobiotic pigs is the ability to maintain a controlled flora and prevent colonization with potentially pathogenic and confounding ubiquitous organisms, gnotobiotic pigs are more costly, less available, and more labor intensive than standard crossbred conventional pigs and are less representative of a typical pig from a production setting. Working from the hypothesis that CDAD can be experimentally induced in conventional neonatal pigs, the objective of this study was to create a reliable and cost-effective model to study C difficile in relation to its clinical implication in swine-production medicine. Information gained can be used to further investigate intervention strategies and increase understanding of CDAD in swine.
Materials and methods
Animals and housing
The experimental protocol was approved by the Iowa State University (ISU) Institutional Animal Care and Use Committee.
Nineteen conventional neonatal Duroc × (Landrace × Large White) pigs were enrolled in the study. Neonatal pigs were from a commercial Iowa sow farm. Sows were seropositive for porcine reproductive and respiratory syndrome virus (PRRSV), but were not known to be shedding virus. All pigs in this study were PRRSV-negative by polymerase chain reaction (PCR) testing of sera.17
At the farm of origin, three sows in farrowing crates were cleaned and monitored for signs of parturition. Sows were cleaned with ammonium chloride disposable wipes (Clorox Disinfecting Wipes; The Clorox Company, Oakland, California). The entire rump area of each sow was wiped, with extra scrubbing around the vulvar and perineal regions. Once parturition began, as indicated by active abdominal straining, a surgical drape was placed in the farrowing crate behind the sow. Sows were allowed to farrow naturally, and immediately post delivery, individual piglets were removed from the sow (snatch-farrowed), dried, and placed in a clean plastic tote with supplemental heat. When there was a break in piglets being born, umbilical cords were clamped, cut, and then sprayed with iodine. Colostrum was collected from the farrowing sows, and equal volumes from each sow were pooled. Approximately 1 hour after birth, each neonatal pig received 10 mL of pooled colostrum mixed in the same 60-mL syringe with 20 mL of commercial puppy milk replacer (Esbilac liquid puppy formula; Pet-Ag, Hampshire, Illinois). The 30-mL mixture of colostrum and milk replacer was delivered via orogastric tube (Sovereign 8-gauge rubber French catheter; Tyco Healthcare, Mansfield, Massachusetts). Samples of colostrum were saved and later tested for neutralization of purified TcdA or TcdB in a tissue culture assay using methods previously described.18,19 Pigs were then transported to a Biosafety Level 2 animal facility at ISU. At the research facility, pigs were housed in two identical raised plastic decks partitioned into individual pens (approximately 0.7 × 0.7 m) with solid dividing walls (Figure 1) and individual feeding bowls. All pigs were housed in the same airspace. Room temperature was maintained at 29°C, and heat lamps were suspended over the pigs to keep the immediate environment at 35°C. Pigs were bowl-fed 60 mL commercial puppy milk replacer three times daily, with feedings spaced at approximately 7- to 9-hour intervals.
Figure 1: Neonatal piglets inoculated with Clostridium difficile were housed on two raised plastic decks, each partitioned into eight individual pens with solid dividing walls on a common grate.  |
A number of measures were taken to prevent accidental contamination of all pigs. New and disinfected or sterilized totes, surgical drapes, and towels were used for handling the pigs on the sow farm. Designated personnel attended the farrowings, changing disposable gloves after delivery of each piglet, and within the farrowing room, totes were maintained away from crates and other litters. Instruments used for umbilical cord clipping were disinfected, and the rooms used at the research facility were also extensively disinfected. All personnel showered before entering the room in the research facility, gloves were changed before handling each different group of pigs, and dedicated feeding instruments and tubes were used for each pig.
Experimental design
Due to space constraints, the study was executed in two identical trials 1 week apart, with pigs randomly assigned, using a random number generator (Excel; Microsoft, Redmond, Washington), to a challenge group (n = 6 or 7) or the control group (n = 3) per trial. In the event that one isolate did not elicit disease, two different C difficile isolates were used. Approximately 4 hours after birth, pigs were orogastrically either sham-inoculated (Control) or inoculated with C difficile using an 8-gauge rubber French catheter (Sovereign;Tyco Healthcare). At 24, 48, and 72 hours post inoculation (hpi), one Control pig and two or three pigs per challenged group were randomly selected (using a random number generator; Excel) and euthanized by an intravenous overdose of pentobarbital (Fatal-Plus; Vortech Phamaceutical, Ltd, Dearborn, Michigan). Experimental design is summarized in Table 1.
Inoculum
Two different field isolates of C difficile were selected for experimental infection (Table 1). Both were isolated at the ISU Veterinary Diagnostic Laboratory from clinical field cases of diarrhea in neonatal pigs from different farms. In both cases, high levels of toxin (4+) in colon content were detected by enzyme-linked immunosorbent assay (ELISA) (Clostridium difficile tox A/B II kit; TechLab, Blacksburg, Virginia). Isolates were stored at -80°C until culture preparation.
Table 1: Experimental design for conventional pigs sham-inoculated (Controls) or challenged with two different Clostridium difficile field isolates*
* Two trials were conducted, with necropsies performed at 24, 48, and 72 hour post inoculation. † C difficile isolates originated from two different animals from different farms in Missouri. Both isolates were ribotype 078, toxinotype V. Hpi = hours post inoculation |
|||||||||||||||||||||||||||||||
Lawns of isolates were cultivated on anaerobic brain-heart infusion agar with 0.5% yeast extract and 0.05% L-cysteine and incubated in an anaerobic glovebox (Bugbox; Ruskinn Life Sciences Ltd, Pencoed, Bridgend, Mid Glamorgan, United Kingdom) with an atmosphere of 10% carbon dioxide, 10% hydrogen, and 80% nitrogen at 37°C for 7 days. Lawns were harvested with 5 mL phosphate-buffered saline (PBS; 0.01 M, pH 7.2) and centrifuged at 10,000g for 20 minutes at 4°C. The pellet was washed with 1M KCl:0.5M NaCl, centrifuged as before, and resuspended in 25 mL 50 mM Tris-HCl (pH 7.2) with 10 mg lysozyme per mL. After 1 hour incubation at 37°C, the material was washed three times with 25 mL sterile nanopure water, using the same centrifugation conditions as before. Before centrifugation of the third wash, the spores were counted on a hemacytometer. The pellet was adjusted to a final concentration of approximately 2 × 109 spores per mL in sterile nanopure water and stored at 4°C. Immediately prior to challenge, spores were heat-shocked in a water bath at 80°C for 10 minutes. Brain-heart infusion broth with 0.1% taurocholic acid and 5% fetal bovine serum was added to the heated spore suspension at a concentration of 25% v/v and incubated 1 hour at 37°C. Brain-heart infusion broth was used in place of spores in the sham (Control) inoculum. Toxinotype, ribotype, genus and species identity, and presence of tcdA and tcdB genes were determined or confirmed by PCR as previously described.20-23
Inoculation
Orogastric inoculation was performed for all groups. The sham inoculum for the control group in each trial was 1.25 mL of sterile medium immediately followed by 15 mL of milk replacer. For the treatment groups, 1.25 mL of challenge preparation containing 2 × 109 per mL heat-shocked C difficile spores was administered, followed by 15 mL of milk replacer. Challenge material or sham inoculum was administered approximately 3 hours after colostrum. Pigs were allowed access to milk replacer (via bowls) immediately after administration of challenge.
Sample collection
Rectal swabs for aerobic and anaerobic culture (Culturette; BD, Franklin Lakes, New Jersey) and C difficile toxin detection (Stool-Prep; TechLab, Blacksburg, Virginia) were collected from each pig immediately prior to inoculation. A rectal swab for toxin assay was collected from each pig each day post inoculation until necropsy and tested within 1 hour post sampling.
Necropsy
Gross observations at necropsy, including body condition, hydration status, perineal fecal staining, consistency of colonic contents, mesocolonic edema, and visible luminal necrosis, were assessed as previously described.4 Body condition was scored 0 to 2 (0 = normal, 1 = thin, 2 = emaciated). All other assessments were scored 0 to 3 (0 = normal, 1 = mild, 2 = moderate, 3 = severe). Fresh and formalin-fixed tissues were collected with sterilized instruments and included ileum, jejunum, descending colon, cecum, and a cross-section through the spiral colon containing four to five loops. Colonic contents were collected in a sterile plastic cup.
Culture and toxin detection
Colonic content and mucosa swabbed from two sections of the spiral colon, approximately 3 to 4 cm long, were combined and cultured on C difficile selective agar (CDSA; Remel, Lenexa, Kansas) both directly and after a 30-minute room-temperature incubation of the swab in 0.5 mL 100% ethanol (e-mail communication, 2009, J. G. Songer, Research Professor, ISU; and written communication, 2009, J. M. Kinyon, Assistant Scientist, Clinical Microbiology, ISU). All plates were incubated at 37°C for 48 hours in an anaerobic chamber. Plates were positioned under a Wood’s lamp, and two fluorescing colonies with morphology typical of C difficile were subcultured to CDSA and incubated at 37°C for 48 hours in an anaerobic chamber. Two fluorescing colonies with typical C difficile morphology from these plates were again subcultured on blood agar and incubated at 37°C for 48 hours in an anaerobic chamber. Blood agar plates were examined for pure cultures with typical colony morphology, a characteristic “horse stable-like” odor, and fluorescence under a Wood’s lamp. Identity was confirmed by Gram stain revealing long, thin, straight, gram-positive rods, a positive L-proline aminopeptidase disk test (PRO-disc; Remel, Lenexa, Kansas),24 and PCR as previously described.21 Polymerase chain reaction was used for determination of ribotype, toxinotype, and detection of toxin genes in isolates and challenge material using methods described previously.20,22 Pigs received a score of 0 or 1 for negative or positive C difficile culture, respectively.
Pooled colon and cecum contents were assayed for C difficile toxins with the Clostridium difficile tox A/B II kit according to the manufacturer’s instructions. This ELISA detects both TcdA and TcdB, but does not differentiate between them. Optical densities of the tests were measured on a microplate ELISA reader and toxin levels were graded on a scale from 0 (no toxin detected) to 4+ (marked toxin detection). This value was used as the toxin score.
Small intestinal contents were tested by routine aerobic, anaerobic, and enrichment culture methods to test for Salmonella species, hemolytic or mucoid Escherichia coli, and Clostridium perfringens. Two samples of cultured hemolytic or mucoid E coli and three samples of C perfringens were arbitrarily selected for PCR genotyping.25,26
Histopathology
Tissue sections were collected in 10% neutral buffered formalin, routinely processed, and stained with hematoxylin and eosin. Tissues were examined by a veterinary pathologist blinded to group designation. Cecum and spiral colon were assessed using 400× magnification for the percentage of goblet cells lining glands, number of neutrophils in the lamina propria, and mucosal alterations (erosions or ulcerations). Additionally, mesenteritis was scored in the spiral colon. Microscopic lesions were evaluated and scored as described previously4 with the following modifications: addition of a third category for neutrophil numbers in the lamina propria, more detailed (quantifiable) description of mucosal epithelial erosions with addition of an ulceration score, and addition of a mesenteritis category. Scores were reported for the entire examined section of cecum, the four or five loops of spiral colon, and the mesentery. Table 2 summarizes the scoring system for histopathology used in this study. Maximum cecal and colonic scores were 9 and 12, respectively.
Table 2: Scoring system for intestinal histopathology in neonatal pigs inoculated with Clostridium difficile or sham-inoculated and necropsied 24, 48, or 72 hours later
* PMNs counted in the entire section using 400× magnification. † Erosions or ulcerations counted in the entire section using 400× magnification. |
||||||||||||||||||||||||||||||||||||||||||
Scoring
Three categories of scores were compared: clinical signs, gross lesions, and microscopic lesions. Clinical signs scores were created by summing scores for body condition, hydration status, and perineum staining (maximum total score of 8).4 A pig was considered to be diarrheic if it received a perineum staining score of 1 or greater. Gross lesions scores were created by summing scores for necrotizing lesions, mesocolonic edema, culture, and toxin (maximum total score 13), and the microscopic lesion score was the sum of scores for all histopathology categories in the cecum and colon (maximum total score 21).
Results
Toxin neutralization was not observed in any of the colostrum samples collected at parturition and fed to piglets.
Bacterial culture
Clostridium difficile was isolated from the colon content of all pigs at necropsy, including controls. All isolates recovered from challenged pigs were ribotype 078, toxinotype V, the same as their respective challenge isolates. Toxinotypes 0, V, and IIIb and ribotypes 078 and 027 were recovered from control pigs. Day 0 rectal culture for C difficile was negative for all but one pig in challenge isolate group 15454-1. In that pig, a single colony was cultured after ethanol incubation, but there was no growth from the direct culture. No Salmonella species was isolated from any pig. Hemolytic E coli was isolated from the intestinal contents of one of the 19 pigs (5.2%). Upon genotyping, this isolate was nontoxigenic. Clostridium perfringens was isolated from the intestinal contents of 14 of the 19 pigs (73.6%). Three C perfringens isolates were randomly selected for genotyping and were positive for alpha and beta-2 toxins.
Clinical signs
Clinical signs data are listed in Tables 3 and 4. At 24 hpi, all pigs had normal to thin body condition, and all were normally hydrated with no diarrhea. Half of the control pigs had diarrhea, mild to moderate dehydration, and a thin body condition at the later time points, whereas most of the challenge pigs had severe diarrhea (six of nine; 66.7%), mild to moderate dehydration (five of nine; 55.6%), and thin or emaciated body condition (four of nine; 44.4%).
Table 3: Mean scores* at necropsy for clinical signs, gross lesions, and microscopic lesions in the colonic lamina propria in neonatal pigs at three time points post orogastric inoculation of a Clostridium difficile isolate or sham-inoculation (Controls)
* Experimental design described in Table 1. Scoring system for microscopic lesions defined in Table 2 (maximum score 21). Gross lesions scores were summed for necrotizing lesions, mesocolonic edema, culture for C difficile, and detection of C difficile TcdA and TcdB toxins by enzyme-linked immunosorbent assay (maximum total score 13). Hpi = hours post inoculation; SD = standard deviation; Hist = histologic (microscopic) lesions. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 4: Numbers of neonatal pigs showing clinical signs, positive for Clostridium difficile toxin,* or with microscopic intestinal lesions at necropsy at 24, 48, or 72 hours after orogastric inoculation of C difficile or sham-inoculation (Controls)†
* Colon contents tested by enzyme-linked immunosorbent assay for C difficile TcdA and TcdB toxins. † Study design described in Table 1. ‡ Erosions or ulceration. § Group challenged with isolate 13912-1 (n = 7) plus group challenged with isolate 15454-1 (n = 6). |
||||||||||||||||||||||||||||
Gross lesions
Gross-lesion data are summarized in Tables 3 and 4. Mesocolonic edema was observed in two of the six control pigs (33.3%) across all time points, but colon contents in most (three of four; 75.0%) became pasty or watery with time. Mesocolonic edema (12 of 13; 92.3%) (Figure 2) and loose colon contents (10 of 13; 76.9%) were observed in most of the challenged pigs, increasing in severity with time. Grossly visible necrotizing mucosal lesions were not observed in any of the pigs.
Figure 2: Marked mesocolonic edema in the spiral colon of a neonatal pig inoculated with Clostridium difficile (isolate 13912-1) 48 hours previously.  |
Microscopic lesions
Microscopic lesions (Tables 3 and 4) were observed in half of the controls. While most were minor, one pig did have severe lesions, scoring 3 for neutrophilic infiltration in the colonic mucosa. Nearly all of the challenged pigs (11 of 13; 84.6%) had microscopic lesions, and seven of these 13 (53.8%) pigs had moderate to severe lesions. Three severely affected pigs had characteristic fibrinonecrotic “volcano lesions” (Figure 3). Microscopic lesion scores for challenged pigs were highest at 48 hpi.
Figure 3: Spiral colon of a neonatal pig inoculated with Clostridium difficile (isolate 13912-1) 48 hrs previously. The colonic glands are lined with hyperplastic enterocytes with decreased goblet cells; numerous neutrophils infiltrate the lamina propria. Focally there is a mucosal ulceration associated with necrotic cellular debris (volcanic lesion; arrow) (hematoxylin and eosin stain, original magnification × 200). Bar = 100 μm. 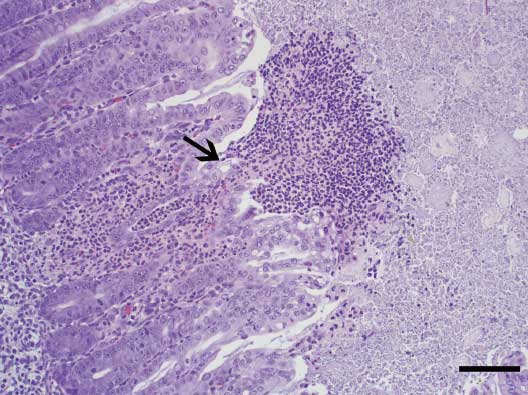 |
Toxin
Prior to sham-inoculation or challenge with C difficile, rectal swabs from all pigs were negative for toxins A and B. Toxin data at necropsy are summarized in Tables 4 and 5. Toxin was detected in half of the control pigs (three of six; 50.0%) and in most of the challenged pigs (10 of 13; 76.9%). As with microscopic lesions, toxin scores in challenged pigs were highest at 48 hpi. Two challenged pigs (15.4%) had moderate fecal toxin scores (2 or 3) before the day of necropsy, but toxin was not detected in these pigs at necropsy.
All challenge and recovered isolates possessed genes for TcdA and TcdB as determined by PCR. Fecal toxin was detected in four of 13 challenged pigs (30.8%) at least once during the study, but toxin was not detected in colonic content at necropsy in these pigs. In a smaller number of challenged pigs (two of 13; 15.4%), toxin was detected in colon content but never in fecal swabs.
Table 5: Mean colon and fecal Clostridium difficile toxin scores* in neonatal pigs orogastrically inoculated with C difficile or sham-inoculated (Controls) and necropsied at 24, 48, or 72 hpi
* Experimental design described in Table 1. Colon content or rectal swab samples tested by enzyme-linked immunosorbent assay (ELISA) (Clostridium difficile tox A/B II kit; TechLab, Blacksburg, Virginia), which detects both TcdA and TcdB, but does not differentiate between them. Optical densities were measured on a microplate ELISA reader and toxin levels were graded on a scale from 0 (no toxin detected) to 4+ (marked toxin detection). Hpi = hours post inoculation; SD = standard deviation. |
||||||||||||||||||||||||||||||||||||||||||||||||||
Discussion
In this study, disease was reproduced by both challenge isolates that had been initially recovered from field cases. Commonly observed lesions and indications of CDAD, including mesocolonic edema, toxin, diarrhea, and microscopic lesions, were observed in challenged pigs. The hallmark volcano lesions resulting from excessive neutrophil exudation into the gut lumen were observed in 23.1% of the pigs. These observations are similar to those described by others4-6 in naturally occurring field cases.
Additionally, the current study was able to reproduce signs of disease after administration of colostrum and without starvation or antibiotic pretreatment, which may be required in other animal models of CDAD.16 The data from this study demonstrated that clinical signs and both gross and microscopic lesions were generally more severe at 48 and 72 hpi than at 24 hpi. This is of use to future studies, as some time is required for the disease to fully develop. Though it could not be statistically demonstrated due to the small sample size, there is a slight observable trend in diminishing disease severity from 48 to 72 hpi that suggests lesions may decrease if the period between inoculation and necropsy is longer.
Though the scores of the controls were generally lower than those of the challenged pigs, the number of controls per necropsy is a limiting factor, as a reliable statistical analysis is not possible. The small sample size, as well as the study design, allowed for only two or three challenged pigs to be compared to a single control pig at any given necropsy. Because of the small number of comparisons, care must be taken in interpreting the data. For instance, mean scores for clinical signs, gross and microscopic lesions, and fecal and colon content toxin for each necropsy period may be misleading, as the means are based on observations of only two or three pigs. Thus, an outlier value may have considerable influence and effect on the overall outcome and perception of differences in disease.
The fact that the controls were not completely free of C difficile or other potentially pathogenic organisms at necropsy does reveal one disadvantage of the method of snatch-farrowing conventional pigs and underscores the fact that a full understanding of C difficile transmission in swine is lacking.16 Although extensive precautions were taken to avoid contaminating the pigs, contamination still occurred. Some controls may have been inadvertently contaminated, as they shared the same room and raised grate flooring as the challenged pigs. It is also possible that the signs and isolation of C difficile resulted from exposure to the organism at the farm of origin, as supported by the toxinotypes and ribotypes recovered from these pigs, which did not match those of the isolates used in the challenge. The overwhelming dose of spores administered to the challenged pigs may have masked recovery of the ribotypes and toxinotypes isolated from the controls.
A recent study published after the completion of this work described spores isolated from the air around infected human patients.27 While there is still potential for unintended exposure at the farm, this risk can be mitigated in future studies by housing challenged and control piglets in separate rooms. Regardless of precautions to prevent accidental cross-contamination, transmission of disease occurred much more easily than is the current belief. Hopman et al7 recently showed that in a production setting, piglets can become positive for C difficile within 1 hour post partum, while caesarian-derived piglets remained negative for the 21 days they were kept in sterile incubators, information that was not available at the time of this study.
Clostridium perfringens type A was isolated from most of the pigs (73.6%). This was expected, as this organism is commonly found in most commercial farms and is considered normal microbiota of the neonatal intestinal tract. This does potentially confound some of the data, but it is impossible to determine how the overall outcome may have been affected by the presence of C difficile of farm origin. Reports have shown that commensal and pathogenic C perfringens type A strains cannot be differentiated. While it is possible that farm strains may have contributed to diarrhea or development of lesions, little is known about the pathogenesis of C perfringens A in pigs, and its isolation from a diarrheic piglet may not indicate that it is the cause of disease.28,29 Hemolytic E coli was isolated from a single pig, which was less likely to be problematic, as genotyping revealed that the isolate was nontoxigenic.
This study has raised additional points of interest that may require further investigation and could employ the use of this model of CDAD. If disease is dose dependent, determining the minimum infectious dose could aid in the study of disease transmission. Studying the roles of different toxinotypes in disease can benefit understanding of existing epidemic strains and allow for identification of other strains with epidemic potential. The model can also be used to determine if the presence of potentially pathogenic secondary bacteria, such as E coli or C perfringens, have an influence on development or severity of CDAD. Such information could add to existing knowledge of disease pathogenesis, as well as facilitate disease control strategies in swine-production centers. Perhaps the most promising use for the model and pressing need for control of CDAD in swine and humans is evaluation of intervention strategies.
Implications
• CDAD can be successfully reproduced in a conventional neonatal piglet model.
• Possible uses for this model include studies of disease pathogenesis and intervention strategies.
• The model can be adapted for in vivo studies of human-origin C difficile strains and therapies intended for use in human medicine.
Acknowledgments
Funding for this project was supported by the National Pork Board. The authors would like to thank Hien Trinh at the University of Arizona and Dr Maja Rupnik at the Institute of Public Health Maribor, Maribor, Slovenia, for toxinotyping the Clostridium difficile isolates; Dr Scott Weese at the University of Guelph, Ontario, for ribotyping and additional toxinotyping; and Stephen Gaul in the Harris Lab for assistance with spore preparation and PCR.
References
1. Songer JG, Anderson MA. Clostridium difficile: an important pathogen of food animals. Anaerobe. 2006;12:1–4.
2. Keel MK, Songer JG. The comparative pathology of Clostridium difficile-associated disease. Vet Pathol. 2006;43:225–240.
3. Songer JG. The emergence of Clostridium difficile as a pathogen of food animals. Anim Health Res Rev. 2004;5:321–326.
4. Yaeger MJ, Kinyon JM, Songer JG. A prospective, case control study evaluating the association between Clostridium difficile toxins in the colon of neonatal swine and gross and microscopic lesions. J Vet Diagn Invest. 2007;19:52–59.
5. Yaeger M, Funk N, Hoffman L. A survey of agents associated with neonatal diarrhea in Iowa swine including Clostridium difficile and porcine reproductive and respiratory syndrome virus. J Vet Diagn Invest. 2002;14:281–287.
*6. Songer J, Post K, Larson D, Jost B, Glock R. Infection of neonatal swine with Clostridium difficile. J Swine Health Prod. 2000;8:185–189.
7. Hopman NE, Keessen EC, Harmanus C, Sanders IM, van Leengoed LA, Kuijper EJ, Lipman LJ. Acquisition of Clostridium difficile by piglets. Vet Microbiol. 2011;149:186–192.
8. Weese JS, Wakeford T, Reid-Smith R, Rousseau J, Friendship R. Longitudinal investigation of Clostridium difficile shedding in piglets. Anaerobe. 2010;16:501–504.
9. Waters EH, Orr JP, Clark EG, Schaufele CM. Typhlocolitis caused by Clostridium difficile in suckling piglets. J Vet Diagn Invest. 1998;10:104–108.
10. Songer JG, Jones R, Anderson MA, Barbara AJ, Post KW, Trinh HT. Prevention of porcine Clostridium difficile-associated disease by competitive exclusion with nontoxigenic organisms. Vet Microbiol. 2007;124:358–361.
11. Weese JS, Staempfli HR, Prescott JF. A prospective study of the roles of Clostridium difficile and enterotoxigenic Clostridium perfringens in equine diarrhoea. Equine Vet J. 2001;33:403–409.
12. Bartlett JG. Clostridium difficile: history of its role as an enteric pathogen and the current state of knowledge about the organism. Clin Infect Dis. 1994;18(suppl 4):S265-S272.
13. Poutanen SM, Simor AE. Clostridium difficile-associated diarrhea in adults. Can Med Assoc J. 2004;171:51–58.
14. Voth DE, Ballard JD. Clostridium difficile toxins: mechanism of action and role in disease. Clin Microbiol Rev. 2005;18:247–263.
15. Lyerly DM, Saum KE, MacDonald DK, Wilkins TD. Effects of Clostridium difficile toxins given intragastrically to animals. Infect Immun. 1985;47:349–352.
16. Steele J, Feng H, Parry N, Tzipori S. Piglet models of acute or chronic Clostridium difficile illness. J Infect Dis. 2010;201:428–434.
17. Hermann JR, Hoff SJ, Yoon KJ, Burkhardt AC, Evans RB, Zimmerman JJ. Optimization of a sampling system for recovery and detection of airborne porcine reproductive and respiratory syndrome virus and swine influenza virus. Appl Environ Microbiol. 2006;72:4811–4818.
18. Ehrich M, Van Tassell RL, Libby JM, Wilkins TD. Production of Clostridium difficile antitoxin. Infect Immun. 1980;28:1041–1043.
19. Sullivan NM, Pellett S, Wilkins TD. Purification and characterization of toxins A and B of Clostridium difficile. Infect Immun. 1982;35:1032–1040.
20. Bidet P, Barbut F, Lalande V, Burghoffer B, Petit JC. Development of a new PCR-ribotyping method for Clostridium difficile based on ribosomal RNA gene sequencing. FEMS Microbiol Lett. 1999;175:261–266.
21. Braun V, Hundsberger T, Leukel P, Sauerborn M, von Eichel-Streiber C. Definition of the single integration site of the pathogenicity locus in Clostridium difficile. Gene. 1996;181:29–38.
22. Kato H, Kato N, Watanabe K, Iwai N, Nakamura H, Yamamoto T, Suzuki K, Kim SM, Chong Y, Wasito EB. Identification of toxin A-negative, toxin B-positive Clostridium difficile by PCR. J Clin Microbiol. 1998;36:2178–2182.
23. Rupnik M, Avesani V, Janc M, von Eichel-Streiber C, Delmee M. A novel toxinotyping scheme and correlation of toxinotypes with serogroups of Clostridium difficile isolates. J Clin Microbiol. 1998;36:2240–2247.
24. Fedorko DP, Williams EC. Use of cycloserine-cefoxitin-fructose agar and L-proline-aminopeptidase (PRO Discs) in the rapid identification of Clostridium difficile. J Clin Microbiol. 1997;35:1258–1259.
25. Casey TA, Bosworth BT. Design and evaluation of a multiplex polymerase chain reaction assay for the simultaneous identification of genes for nine different virulence factors associated with Escherichia coli that cause diarrhea and edema disease in swine. J Vet Diagn Invest. 2009;21:25–30.
26. Meer RR, Songer JG. Multiplex polymerase chain reaction assay for genotyping Clostridium perfringens. Am J Vet Res. 1997;58:702–705.
27. Best EL, Fawley WN, Parnell P, Wilcox MH. The potential for airborne dispersal of Clostridium difficile from symptomatic patients. Clin Infect Dis. 2010;50:1450–1457.
28. Songer JG. Clostridiosis. In: Zimmerman JJ, Karriker LA, Ramirez A, Schwartz KJ, Stevenson GW, eds. Diseases of Swine. 10th ed. Ames, Iowa: Blackwell Publishing; 2012:712–714.
29. Baker AA, Davis E, Rehberger T, Rosener D. Prevalence and diversity of toxigenic Clostridium perfringens and Clostridium difficile among swine herds in the Midwest. Appl Environ Microbiol. 2010;769:2961–2967.
* Non-refereed reference.
