| Case Report | Peer reviewed |
Cite as: Tousignant SJP, Henry SC, Rovira A, et al. Effect of oral vitamin D3 supplementation on growth and serum 25-hydroxy vitamin D levels of pigs up to 7 weeks of age. J Swine Health Prod. 2013;21(2):94–98.
Also available as a PDF.
SummaryBeginning in the spring of 2011, investigations on farms across the United States led to the anecdotal observation that most weaned pigs tested were vitamin D deficient. Work began on developing an oral supplement to be given to piglets early in life, and a few farms immediately adopted its use. Reports from the field by veterinarians and farm staff described encouraging improvement in piglet performance. This case report describes the effects of oral vitamin D3 on one farm during the summer of 2011, where 387 piglets from 36 gilt litters were assigned to either a group supplemented with vitamin D3 at 2 days of age or a control group. Pigs in the supplemented group were heavier at weaning and 7 days post weaning, fewer lost weight during the first 7 days post weaning, and their serum 25-hydroxy vitamin D (25-OHD) levels were higher 26 days post weaning. | ResumenIniciando en la primavera de 2011, investigaciones de granjas a lo largo de los Estados Unidos, llevó a la observación anecdótica de que la mayoría de los cerdos analizados tenían una deficiencia de vitamina D. El trabajo inició desarrollando un suplemento oral que se administraría a lechones a temprana edad, unas pocas granjas adoptaron su uso inmediatamente. Los reportes de campo de los veterinarios y empleados de estas granja describieron mejoras alentadoras en el desempeño de los lechones. Este reporte de caso describe los efectos de la vitamina D3 oral en una granja durante el verano de 2011, donde 387 lechones de 36 camadas de hembras primerizas fueron asignados a un grupo suplementado con vitamina D3 a los 2 días de edad ó a un grupo control. Los cerdos en el grupo suplementado fueron más pesados al destete y a los 7 días post destete, menos cerdos perdieron peso durante los primeros 7 días post destete, y sus niveles de 25-hidroxivitamina D de suero fueron más altos a los 26 días post destete. | ResuméAu début du printemps 2011, des enquêtes sur des fermes à travers les États-Unis ont conduit à l’observation anecdotique que la plupart des porcs sevrés testés avaient une déficience en vitamine D. Le travail débuta à développer un supplément oral pouvant être donné aux porcelets tôt dans leur vie, et quelques fermes adoptèrent immédiatement son utilisation. Des rapports en provenance de vétérinaires en pratique et du personnel de ferme ont décrit une amélioration encourageante dans les performances des porcelets. Ce rapport de cas décrit les effets de la vitamine D3 orale sur une ferme durant l’été 2011, où 387 porcelets provenant de 36 cochettes ont été répartis soit à un groupe recevant un supplément de vitamine D3 à l’âge de deux jours ou à un groupe témoin. Les porcelets dans le groupe recevant un supplément étaient plus pesants au sevrage et 7 jours post-sevrage, ont perdu moins de poids durant les 7 premiers jours post-sevrage, et leur taux sérique de vitamine D-25 hydroxy étaient plus élevés 26 jours post-sevrage. |
Keywords: swine, vitamin D, vitamin D3, cholecalciferol
Search the AASV web site
for pages with similar keywords.
Received: February 6, 2012
Accepted: August 16, 2012
Vitamin D has been at the center of a resurgence of research activity during the last two decades, with particular attention to the role it plays in development and function of the immune system. Vitamin D (used to describe either vitamin D2 or D3 unless otherwise specified) enters the body in two ways: exposure to the sun or consumption of fortified foods and feeds, oily fish such as salmon, or yeasts and mushrooms exposed to ultra-violet light B (UVB light). The primary endogenous source of vitamin D is production by the epidermis exposed to UVB sunlight, which produces the vitamin D precursor known as cholecalciferol. Cholecalciferol is transported to the liver where it is converted to 25-hydroxy vitamin D (25-OHD).1 Commonly regarded as the storage form of vitamin D, 25-OHD is most useful in determining vitamin D status of the animal.2 It is converted into the active form, 1,25(OH)2D by the enzyme 1α-hydroxylase. Previously thought to be only in the kidney and bone where it is involved in mineral homeostasis, this enzyme is now known to exist in various tissues of the body, including those of the immune system.3
Classically, vitamin D is known primarily for its role in bone and muscle metabolism. More recently, there is a growing body of evidence documenting the importance of vitamin D in many other systems of the body, including the development and function of the immune system. One of the earliest studies reporting that alveolar macrophages can convert the storage form of vitamin D into the active form was done using porcine alveolar macrophages.4 It is also reported5 that antigen-presenting cells, such as dendritic cells, possess the enzyme 1α-hydroxylase, needed to convert storage 25-OHD into active 1,25(OH)2D. The same study5 reported that this process is up-regulated when stimulated by lipopolysaccharides, suggesting production of active vitamin D in this way may serve as a localized signal during infection to improve immune response. Another study6 showed activation of toll-like receptors (TLRs) on human macrophages increased expression of vitamin D receptors and the enzyme 1α-hydroxylase. Likewise, it has been shown7 that respiratory epithelial cells have the similar ability to up-regulate production of active vitamin D during viral infections. This ultimately leads to production of important antimicrobial peptides such as cathelicidin, as well as cluster of differentiation 14 (CD14; one of a group of cell-surface marker proteins), which are likely to be part of the host defense response. A notable review paper8 suggests a relationship between low levels of serum vitamin D and influenza epidemics in people. In a recent randomized placebo-controlled study,9 the authors noted a lower incidence of influenza virus infection in school children who received vitamin D supplementation.
The collection of studies outlined above begins to build the case that vitamin D is likely to play a role in proper development and function of the immune system. Modern swine production is dominated by confinement operations where pigs have no direct exposure to natural sunlight. It is becoming increasingly well established that in most, if not all, weaned piglets tested for 25-OHD, levels are extremely low. One study10 reports reference values for 25-OHD of 25 to 30 ng per mL. The same study reported values of 8.42 to 13.75 ng per mL, depending on the season, for nursery-aged pigs raised indoors, and 58.54 ng per mL for 10 nursery age pigs reared outdoors in 2011. The objective of this case report is to describe findings from one farm where oral vitamin D3 supplementation was provided to piglets at 2 days of age.
Case description
Herd description
The design of this study was approved by the University of Minnesota Institutional Animal Care and Use Committee.
Animals were from a privately owned, 1500-head sow farm that was willing to participate and sign a consent form. The herd was representative of generally healthy, well-managed, modern swine production. Monthly blood samples collected from gilts and piglets and submitted to the University of Minnesota Veterinary Diagnostic Laboratory tested negative by polymerase chain reaction for porcine reproductive and respiratory syndrome virus and positive by enzyme-linked immunosorbent assay for swine influenza virus and Mycoplasma hyopneumoniae. Piglets were weaned twice weekly at approximately 21 days of age to an off-site continuous-flow nursery managed all-in, all-out by room. After a 26-day nursery period, pigs were moved to on-site finishing barns. None of the facilities employed bio-aerosol filtration, but all were shower-in, shower-out.
Establishment of baseline 25-OHD
A power calculation indicated baseline 25-OHD levels for piglets in the case herd could be established from 11 piglets with a precision of 2.5 ng per mL, standard deviation of 4 ng per mL, and a confidence level of 95%. A convenience sample of one piglet from 11 randomly chosen litters was selected the day prior to weaning and blood was collected. Mean serum 25-OHD values were 14.4 ng per mL with a standard deviation of 2.7 ng per mL. This supported the authors’ hypothesis that vitamin D levels in this herd were below commonly used reference ranges.10
Selection of test litters
A second power calculation showed that including approximately 180 piglets in each treatment group would detect a difference of 0.3 kg in mean body weight at 26 days post weaning with α = .05 and β of 20%. To reduce the potential for confounded results, only gilt litters were enrolled. During a 3-week period in late summer of 2011, a group of 387 piglets from the first 36 gilt litters that farrowed were enrolled into the trial.
Trial design
To minimize the potential for confounding by litter, both treatments (control and treatment with vitamin D3) were represented within each litter. At approximately 2 days of age, all piglets from each gilt litter enrolled into the trial during the 3-week period were placed into a cart for processing and administration of the oral vitamin D3 supplement. Farm staff were instructed to visually allocate pigs into the two treatment groups by size; birth weights were not recorded. They began with the two visually largest piglets. One was given 1.0 mL vitamin D3 (40,000 IU per mL) (Wean-D; GlycoMyr, Ames, Iowa) according to label directions and the other was left unsupplemented. A sham dose was not administered to the control group. Ear-tag numbers and treatment group were recorded, and pigs were returned to the farrowing crate. Staff then selected the next two visually largest pigs, treating and recording as before until all piglets were processed. If a litter had an odd number of piglets, the largest piglet in the next litter was selected and either treated or left unsupplemented so that numbers of pigs in each group remained approximately equal. All other treatments, including vaccinations, iron injections, and fostering, were performed according to farm protocols regardless of vitamin D treatment group.
Piglets were weaned at an average of 21 days of age. On the day prior to weaning, all pigs in the study were individually weighed on a scale capable of reading to 0.01 kg. One supplemented and one control pig were selected from each litter by a random number generator (72 pigs total), and blood was collected for 25-OHD assay.
Upon arrival at the nursery, piglets enrolled in the trial were haphazardly allocated to pens containing both supplemented and control pigs. Weights were recorded for every pig at 7 and 26 days post weaning. Additionally, blood samples were collected at 7 and 26 days post weaning from the same 72 pigs identified at weaning. All treatments and care in the nursery were performed according to farm protocol. The trial ended at 26 days post weaning.
Blood collection and assay
Blood was collected by jugular venipuncture using a 20-gauge, 1-inch multi-use needle (BD, Franklin Lakes New Jersey) into a 3.0-mL red-topped Vacutainer tube (BD). Blood was refrigerated for 12 to 18 hours before centrifugation. Serum was separated and stored frozen in 1.5-mL microcentrifuge tubes (Eppendorf North America, Inc, Westbury, New York) at -80ºC. All serum 25-OHD assays were performed by Heartland Assays Inc (Ames, Iowa). Of the 36 matched pairs of serum samples collected during the trial, 18 pairs had a complete set of samples from weaning and 26 days post weaning without hemolysis. Because hemolysis interferes with the 25-OHD assay, only these samples were submitted for analysis.
Statistical analysis
Means for body weight, average daily gain (ADG), and serum 25-OHD concentrations were compared between the control and vitamin D-supplemented groups using a t test, and the F test was used to compare the homogeneity of variances of mean body weight. A chi-square test was used to compare the numbers of pigs that lost weight in each group during the first 7 days post weaning. All statistical analyses were performed with the statistical software Statistix version 9 (Analytical Software, Tallahassee, Florida). A P value < .05 was considered significant.
Body weight, ADG, and 25-OHD results
Table 1 summarizes weight and serum 25-OHD data. At weaning and 7 days post weaning, pigs supplemented with vitamin D were significantly heavier than controls. At 26 days post weaning, weights did not differ between treatments. Average daily gain did not differ between groups during the study. While birth weights were not recorded or included in the analysis, significantly fewer pigs in the supplemented group lost weight during the first 7 days post weaning. Serum 25-OHD levels did not differ at weaning, but were significantly higher in supplemented pigs at 26 days post weaning.
Table 1: Effect of oral vitamin D3 supplementation (40,000 IU) given at 2 days of age on nursery pig growth and serum 25-hydroxy vitamin D (25-OHD) levels from weaning to 26 days post weaning (dpw)*
* Pigs weaned at approximately 21 days of age were housed in an off-site modern nursery of a 1500-sow farm. Pigs in the supplemented group (Vitamin D3) received 1.0 mL (40,000 IU) Wean-D (GlycoMyr, Ames, Iowa) orally at 2 days of age. Controls were not treated. During each sampling event, body weights and sera were collected from the same pigs. Mean serum 25-OHD was calculated using 18 samples from the same pigs within each treatment group at each sampling event. ab Values within a row with different superscripts are statistically different (P < .05). † P values calculated using a two-sample t test. ‡ P values calculated using an F test for homogeneity of variances. § P value calculated using a chi-square test. ND = not done. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Figure 1 is a box-and-whisker plot showing the distribution of weights by treatment at weaning and at days 7 and 26 post weaning. In general, weights were normally distributed between treatments and across time with the exception of control pigs on day 26, where more pigs were in the upper 25th percentile when compared to the vitamin D-supplemented group.
Figure 1: Groups of pigs were either orally supplemented with vitamin D3 at 2 days of age or not treated. Pigs were weighed at weaning (approximately 21 days of age) and 7 and 26 days post weaning (dpw). Box-and-whisker plot displays distribution of body weights by treatment and sampling event. The whiskers represent the highest and lowest observation at each sampling event. The lower box represents the 25th percentile of the mean and the upper box represents the mean of the 75th percentile at each sampling event. Numbers of pigs at each sampling event are shown in Table 1. 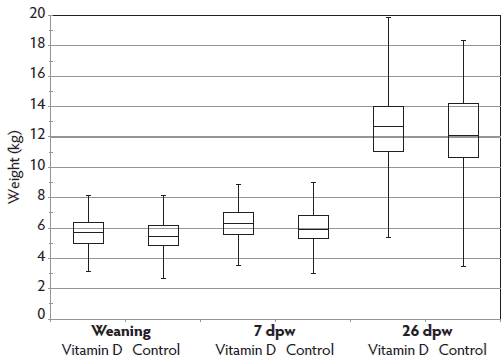 |
Discussion
To the best of our knowledge, this is the first report of the effect of oral vitamin D3 supplementation in suckling pigs. At this farm, supplemented pigs were significantly heavier at weaning and 7 days post weaning, and fewer pigs lost weight during the first 7 days post weaning, but birth weight could not be controlled for in the analysis. The difference in weight was not evident at 26 days post weaning. One might speculate that supplemented pigs began consuming pelleted nursery feed more quickly than controls during the first 7 days post weaning. This is, however, purely speculative, since feed intake was not measured during this trial. If it were true that supplemented pigs ate better than the controls during the first 7 days post weaning, it could be argued that vitamin D3-supplemented pigs might have been easier for farm staff to manage during this critical first week post weaning.
Serum 25-OHD levels did not differ at weaning, yet were significantly different 26 days post weaning. It seems plausible that the one-time dose of oral vitamin D3 increased serum 25-OHD to a peak level a few days after treatment at 2 days of age. Since no other sources of vitamin D are available to the nursing piglet, 25-OHD levels appear to have declined to basal level before weaning. Explaining the significant difference at 26 days post weaning is difficult, keeping in mind the two treatment groups were commingled and eating the same nursery diets.
Limitations
The herd in this trial had relatively high health with excellent management. It is possible that the effects of supplementation would be more pronounced in herds with higher disease pressure. Also, it is important to note the piglets were visually allocated into treatment groups by farm staff at the sow farm, and no birth weights were recorded. The staff may have imparted selection bias in this trial by unknowingly favoring the allocation of oral vitamin D3 supplement to the largest pigs. Additionally, researchers and farm staff at the sow farm were not blinded to treatment.
No other samples, such as nasal swabs, fecal samples, or histopathology samples were collected during the trial, and therefore it was not possible to examine differences in shedding or pathogen concentration.
Future study
This case report only begins to understand the role of vitamin D in growing pigs. More work understanding pig responses to supplemental vitamin D3 is needed. Since little vitamin D is transferred through the placenta or sow milk, and most piglets have no exposure to UV light, a one-time dose of 40,000 IU of vitamin D3 might not be sufficient in rapidly growing piglets that have a high demand due to bone and muscle growth, along with demand from an immune system challenged by farm endemic pathogens.
Supplemented pigs had higher 25-OHD levels than controls at 26 days post weaning. Trials could be designed to understand the mechanisms underlying this difference. Histopathology and more specialized techniques demonstrating intestinal epithelium function could help investigate the role of vitamin D on intestinal absorption.
Additionally, a trial with more rigorous allocation could be performed to determine if these results are repeatable. More frequent serum collection and collection of nasal and rectal swabs would allow for additional measurements of immune function and diagnostic investigations of differences in subclinical disease to help elucidate the mechanism of improved weight gain.
Implications
• Under the conditions and limitations of this case report, suckling piglets supplemented at 2 days of age with oral vitamin D3 are heavier than untreated piglets at weaning and 7 days post weaning, fewer pigs lose weight during the first 7 days post weaning, and serum 25-OHD is higher at 26 days post weaning.
• More studies need to be conducted to understand whether these results are repeatable, in addition to exploring the appropriate dose, frequency, and method of vitamin D3 supplementation in suckling and postweaning pigs.
Acknowledgement
The authors would like to thank the Minnesota Rapid Response Fund for providing the funding for this study.
Conflict of interest
Dr Steven Henry reported interests in GlycoMyr regarding product development, testing, and distribution.
References
1. Holick M. Vitamin D: A d-lightful solution for health. J Invest Med. 2011;59:872–880.
2. Adams J, Hewison M. Update in vitamin D. J Clin Endocrinol Metab. 2010;95:471–478.
3. Beard J, Bearden A, Striker R. Vitamin D and the anti-viral state. J Clin Virol. 2011;50:194–200.
4. Reichel H, Bishop JE, Koeffler HP, Norman AW. Evidence for 1,25-dihydroxyvitamin D3 production by cultured porcine alveolar macrophages. Mol Cell Endocrinol. 1991;75:163–167.
5. Fritsche J, Mondal K, Ehrnsperger A, Andreesen R, Kreutz M. Regulation of 25-hydroxyvitamin D3–1α-hydroxylase and production of 1α, 25-dihydroxyvitamin D3 by human dendritic cells. Blood. 2003;102:3314–3316.
6. Liu P, Stenger S, Li H, Wenzel L, Tan B, Krutzik S, Ochoa M, Schauber J, Wu K, Meinken C, Kamen D, Wagner M, Bals R, Steinmeyer A, Zgel U, Gallo R, Eisenberg D, Hewison M, Hollis B, Adams J, Bloom B, Modlin R. Toll-like receptor triggering of a vitamin D-mediated human antimicrobial response. Science. 2006;311:1770–1773.
7. Hansdottir S, Monick M, Hinde S, Lovan N, Look D, Hunninghake G. Respiratory epithelial cells convert inactive vitamin D to its active form: Potential effects on host defense. J Immunol. 2008;181:7090–7099.
8. Cannell JJ, Vieth R, Umhau JC, Holick MF, Grant WB, Madronich S, Garland CF, Giovannucci E. Epidemic influenza and vitamin D. Epidemiol Infect. 2006;134:1129–1140.
9. Urashima M, Segawa T, Okazaki M, Kurihara M, Wada Y, Ida H. Randomized trial of vitamin D supplementation to prevent seasonal influenza A in schoolchildren. Am J Clin Nutr. 2010;91:1255–1260.
*10. Abbott J, Madson D. Survey of vitamin D levels in swine serum across different stages of production. Proc AASV. 2012;113–115.
* Non-refereed reference.
