| Diagnostic notes | Peer reviewed |
Cite as: Arnold J, Madson DM, Ensley SM, et al. Survey of serum vitamin D status across stages of swine production and evaluation of supplemental bulk vitamin D premixes used in swine diets. J Swine Health Prod. 2015;23(1):28–34.
Also available as a PDF.
SummaryThe objectives of this investigation were to evaluate the serum 25-hydroxyvitamin D [25(OH)D] concentrations in pigs of different age groups, to determine if 25(OH)D concentrations varied with season, and to assess the quality of vitamin D supplements used in swine diets from multiple commercial suppliers. Serum samples (n = 1200) submitted to a diagnostic laboratory for routine surveillance were assayed for serum 25(OH)D concentrations. Vitamin D premix samples were obtained from suppliers and analyzed at two laboratories over a 9-month period. In all age categories, 25(OH)D concentrations in numerous serum samples were lower than reference values. In the nursery, finisher, and boar age categories, there was a difference between the months of January and June (P < .05), with June samples containing higher quantities of circulating 25(OH)D. Serum samples from outdoor herds had higher 25(OH)D concentrations than samples from confined pigs (P < .01). Among the supplement samples evaluated, no individual supplement had a concentration of 25(OH)D significantly lower than 500,000 IU per g. These results revealed that commercial swine may be deficient in serum vitamin D at varying times of the year, and feed-supplement concentrations may vary. | ResumenLos objetivos de esta investigación fueron evaluar las concentraciones en el suero de vitamina 25-D hydroxy [25(OH)D] en cerdos de diferentes grupos de edad, para determinar si las concentraciones de 25(OH)D variaron con la estación, y valorar la calidad de los suplementos de la vitamina D utilizados en las dietas porcinas de varios proveedores comerciales. Se analizaron las muestras de suero (n = 1200) enviadas al laboratorio de diagnóstico para el monitoreo de rutina en busca de concentraciones de suero 25(OH)D. Se obtuvieron muestras de premezclas de vitamina D de los proveedores y se analizaron en dos laboratorios durante un periodo de 9 meses. En todas las categorías de edad, las concentraciones de 25(OH)D en numerosas muestras de suero fueron más bajas que los valores de referencia. Hubo una diferencia entre los meses de enero y junio (P < .05) en las categorías de destete, finalización, y machos, las muestras de suero de junio tuvieron un contenido más alto de concentraciones de 25(OH)D. Las muestras de suero de hatos de pastoreo tuvieron concentraciones más altas de 25(OH)D que las muestras de cerdos confinados (P < .01). Entre las muestras de suplementos evaluados, ningún suplemento individual tuvo una concentración de 25(OH)D significativamente más baja a 500,000 IU por gr. Estos resultados revelaron que los cerdos comerciales pueden ser deficientes en suero a vitamina D en diferentes épocas del año, y que las concentraciones de suplemento de alimento pueden variar. | ResuméLes objectifs de cette étude étaient de déterminer les concentrations sériques de la 25-hydroxyvitamine D [25(OH)D] chez les porcs de différents groupes d’âge, de déterminer si les concentrations de 25(OH)D variaient avec les saisons, et d’évaluer la qualité des suppléments de vitamine D utilisés dans l’alimentation porcine et provenant de plusieurs fournisseurs commerciaux. Des échantillons de sérum (n = 1200) soumis à un laboratoire de diagnostic dans le cadre de programme de surveillance de routine furent testés pour déterminer les concentrations de 25(OH)D. Des échantillons de pré-mélange de vitamine D furent obtenus de fournisseurs et analysés par deux laboratoires sur une période de 9 mois. Dans toutes les catégories d’âge, les concentrations de 25(OH)D dans de nombreux échantillons de sérum étaient inférieures aux valeurs de référence. Dans les catégories d’âge des animaux en pouponnière, des animaux en finition et chez les verrats, il y avait une différence entre les mois de janvier et de juin (P < 0,05), avec les échantillons sériques de juin contenant des concentrations de 25(OH)D plus élevées. Les échantillons sériques provenant des animaux logés à l’extérieur avaient des concentrations plus élevées de 25(OH)D que les animaux en confinement (P < 0,01). Parmi les échantillons de supplément évalués, aucun supplément individuel n’avait une concentration de 25(OH)D significativement inférieure à 500,000 UI par g. Ces résultats révèlent que les porcs commerciaux pourraient être déficients en vitamine D sérique à différents moments dans l’année, et que les concentrations de suppléments alimentaires peuvent varier.
|
Keywords: swine, vitamin D, serum, feed
Search the AASV web site
for pages with similar keywords.
Received: May 1, 2014
Accepted: July 14, 2014
Vitamin D is a fat-soluble hormone essential for calcium homeostasis, with bodily stores in adipose tissue, muscle, and liver.1 Mammals maintain serum calcium concentrations within a narrow range for normal muscle contractions, nerve activity, and release of various other hormones. Calcium homeostasis is maintained by mobilization of calcium from bone reserves, conservation of calcium in the kidney, and absorption of calcium from the diet.2 Vitamin D is involved in regulation of active calcium absorption from the intestine. Sustained hypovitaminosis D can result in metabolic bone disease, a general term used to describe multiple nutritional diseases related to bone growth, bone modeling, or both. In growing pigs with hypovitaminosis D, the open growth plates become widened due to failure of endochondral ossification. This disease process is known as rickets. In mature animals with hypovitaminosis D, the disease is classified as osteomalacia, as the growth plates have closed and the primary lesion is defective bone remodeling.3
Not only does vitamin D have a crucial role in calcium absorption, homeostasis, and bone formation, other body systems also utilize vitamin D. Vitamin D receptors on the nuclei of activated T-lymphocytes and antigen-presenting cells are consistent with a role for vitamin D in control of immune responses. The enzyme 25-hydroxyvitamin D3-1-hydroxylase, which converts 25(OH)D to the active hormone, is found in locations other than the kidney, suggesting that it has other functions, eg, autocrine and immune system functions.4
Over the past 3 years, cases of vitamin D deficiency resulting in hypocalcemia have drawn interest in the swine industry. In 2010, the Iowa State University Veterinary Diagnostic Laboratory (ISU-VDL) investigated several cases of sudden death in nursery and finishing pigs that were ultimately attributed to vitamin D deficiency and hypocalcemia. Several cases were associated with feed errors; however, other cases were not associated with mixing errors nor improper amounts of other ingredients, despite diagnosis of hypovitaminosis D.5 The objectives of this study were to further investigate vitamin D-related issues in swine by determining the range of serum 25(OH)D concentrations in pigs of different ages and from different stages of production, comparing these values to published reference values and determining if 25(OH)D concentrations are affected by season, with the additional objective of assessing the vitamin D concentrations in feed premixes from multiple commercial suppliers.
Materials and methods
All biological samples were either obtained under a valid client-patient relationship or submitted to the ISU-VDL for primary purposes other than vitamin D surveillance. Institutional Animal Care and Use Committee approval was not necessary for this evaluation.
Serum vitamin D assessment
In January and June of 2011, swine serum samples submitted to the ISU-VDL for routine disease surveillance were screened to meet study requirements, which included no reported clinical history of lameness or metabolic disease, at least eight serum samples from different pigs per herd submission, and submissions from pigs within a defined age range. Age categories analyzed are summarized in Table 1 and included nursery, grower, and finisher pigs, sows, and mature boars. Fifteen case submissions in each age category, from pigs raised predominantly in the upper Midwest United States, were selected for study inclusion from a 3-week period in January and then again in June 2011. A total of 1200 serum samples were selected, 600 for each month. Serum samples were stored at -80°C and then submitted by month of collection to Heartland Assays LLC (Ames, Iowa) for 25(OH)D assay. The serum 25(OH)D assay has a detection range of 2.5 to 100 ng per mL, with an assay coefficient of variation of 8.0 to10.0.6
Table 1: Age categories of confined swine (nursery, grower, finisher, sow, and boar) assessed for serum 25-hydroxyvitamin D concentration*
| Category | Age |
|---|---|
| Nursery | 2-4 weeks |
| Grower | 10-14 weeks |
| Finisher | 6-8 months |
| Sow | Mature |
| Boar | Mature |
* A total of 1200 serum samples were assayed; 120 samples were collected per group in January and June 2011.
In June 2011, an additional set of serum samples from pigs raised outdoors or that had access to open lots were obtained. The additional pigs included nursery, grower, and finisher pigs and sows. Ten serum samples were collected from each age group to compare 25(OH)D concentrations in the samples from confined and outdoor pigs.
Vitamin D supplement assessment
Through collaboration with swine feed companies, samples of vitamin D premixes were collected monthly from November 2011 through July 2012. Samples were submitted to the ISU-VDL from five independent swine nutrition companies during this time. Each received sample was assigned a unique identification number, and then all identifying information was removed. Information recorded for each received sample included the following: date of collection, date of manufacturing, expected concentration of 25(OH)D, vitamin D manufacturer name, manufacturer country of origin, lot number, and supplier name (the swine nutrition company providing the sample).
Each sample was homogenized, then divided into two equal aliquots and stored at 4°C until submitted for vitamin D analysis to two separate laboratories, DSM Nutritional Products North America (Parsippany, New Jersey) and Heartland Assays, LLC (Ames, Iowa). Vitamin D analysis at both laboratories was accomplished by high-performance liquid chromatography with ultraviolet detection. Vitamin D concentrations in the supplements were determined by DSM Nutritional Products and Heartland Assays according to their standard operating procedures.
Statistical analysis
Microsoft Excel (Microsoft Corporation, Redmond, Washington) and JMP (JMP software version 8.0.2; SAS Institute, Cary, North Carolina) were used to generate one-way analyses of variance (ANOVAs) for the serum data. SAS (SAS Institute) was used to analyze vitamin D concentrations in vitamin D premixes. Measured concentrations were evaluated in a linear mixed model with distributing company, laboratory, company of manufacturer, and month as fixed-effect variables, sample as a random effect, and their interactions. Differences were considered significant at P < .05 for both the serum and vitamin D premix samples.
Results
Serum 25(OH)D analysis
There was considerable variation in all age groups, with individual pigs within each group having samples deemed deficient compared to historical reference values (Table 2). Mean serum 25(OH)D results from January and June collections were lower or near the low side of the reference intervals for nursery pigs, growers, sows, and boars (Table 3). Figure 1 illustrates that mature animals had greater 25(OH)D concentrations than younger animals (in both January and June). When January submissions were compared to June submissions, serum 25(OH)D concentrations in the June samples were higher in nursery, finisher, and boar age categories (Table 3). Grower pig values were, however, significantly higher in January than in June. When June submissions from confined herds were compared to submissions from pigs with access to the outdoors, in all age categories (excluding boars, which were not tested), serum 25(OH)D concentrations were significantly greater in outdoor pigs than in confined animals (P < .05) (Table 4).
Table 2: Previously reported swine reference intervals by age for serum 25-hydroxyvitamin D [25(OH)D] compared to overall mean and range of samples assayed in this study
| Serum 25(OH)D | |||
|---|---|---|---|
| Age | Reference intervals (ng/mL)* | Current study (ng/mL) | |
| Combined overall mean (SEM)† | Combined overall range‡ | ||
| Neonate | 5-15 | NA | NA |
| 10 days | 8-23 | NA | NA |
| 2-4 weeks | 18-30 | 11.4 (0.71) | 2.5-62.4 |
| 10-14 weeks | 18-30 | 19.5 (0.61) | 3.4-54.1 |
| 6-8 months | 18-30 | 26.4 (0.90) | 3.7-77.9 |
| Mature sow | 35-70 | 36.0 (1.19) | 4.7-94.5 |
| Mature boar | 35-70 | 38.5 (1.04) | 8.9-93.8 |
* Serum 25-hydroxyvitamin D reference intervals.5
† Mean of all 240 serum samples collected in each age range (SEM): overall values derived from both the January and June 2011 serum samples, collected as described in Table 1.
‡ Range of all 240 serum samples collected in each age category.
SEM = standard error of the mean; NA = not applicable; no animals sampled in this age category for this study.
Table 3: Comparison of mean serum 25-hydroxyvitamin D concentrations in confined swine in January and June 2011*
| Serum 25(OH)D (ng/mL) (SEM) | |||
|---|---|---|---|
| Age category | January 2011 | June 2011 | P† |
| Nursery | 8.72 (1.02) | 13.75 (1.12) | < .001 |
| Grower | 21.02 (0.94) | 18.05 (0.75) | .014 |
| Finisher | 24.61 (1.21) | 28.18 (1.32) | .048 |
| Sow | 35.70 (1.45) | 36.33 (1.89) | .792 |
| Boar | 31.56 (1.22) | 45.42 (1.43) | < .001 |
* Samples collected as described in Table 1.
† ANOVA; P < .05 considered statistically significant.
25(OH)D = 25-hydroxyvitamin D; SEM = standard error of the mean.
Figure 1: Mean, standard deviation, and range of serum 25-hydroxyvitamin D [25(OH)D] concentrations in blood samples from commercial swine herds submitted to the Iowa State University-Veterinary Diagnostic Laboratory (Ames, Iowa) for routine disease surveillance in January and June 2011, and concentrations in known outdoor swine. There was a significant increase (P < .05) in the nursery, grower, finisher, and boar serum 25(OH)D concentrations from January to June. There were also significantly higher serum 25(OH)D concentrations (P < .05) in the outdoor nursery, grower, finisher, and sow samples than in the June samples from confined animals.
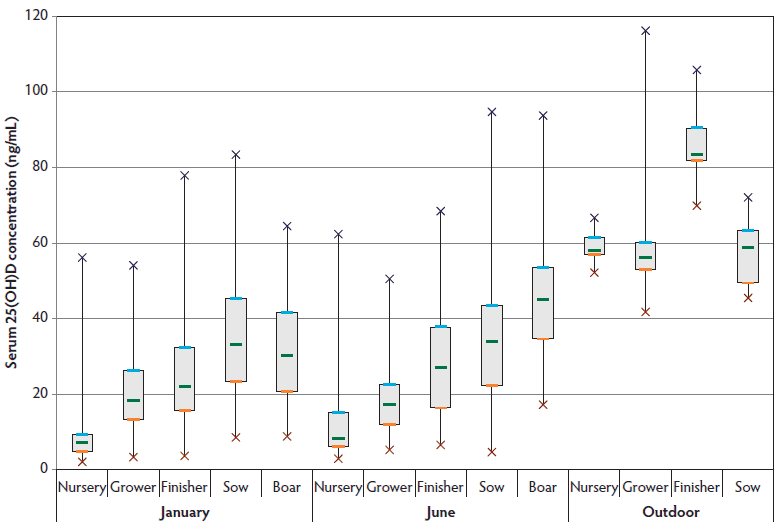
Table 4: June 2011 mean 25(OH)D serum concentrations (SEM) of confined herds compared to outdoor swine herds
| Serum 25(OH)D (ng/L) | P† | ||
|---|---|---|---|
| Age category | Confined pigs | Outdoor pigs* | |
| Nursery | 13.75 (1.12) | 58.64 (1.41) | < .001 |
| Grower | 18.05 (0.75) | 61.05 (6.56) | < .001 |
| Finisher | 28.18 (1.32) | 85.98 (3.31) | < .001 |
| Sow | 36.33 (1.89) | 57.17 (2.80) | .002 |
| Boar | 45.42 (1.43) | NA | NA |
* The outdoor sows had black skin, while the nursery, grower, and finisher pigs were white breeds.
† ANOVA; P < .05 considered statistically significant.
25(OH)D = 25-hydroxyvitamin D; SEM = standard error of the mean; NA = not applicable, ie, no samples were collected from outdoor boars.
Vitamin D premixes
A total of 45 vitamin D premix samples were collected, resulting in 90 assays completed. Of the 45 samples received, 23 were manufactured outside the United States and 22 samples originated from two US manufacturers. Two nutrition companies provided vitamin D samples sourced strictly from foreign manufacturers during the study period, one supplier provided samples from a US source only, and the remaining two suppliers provided a mix of US and foreign vitamin D sources for analysis.
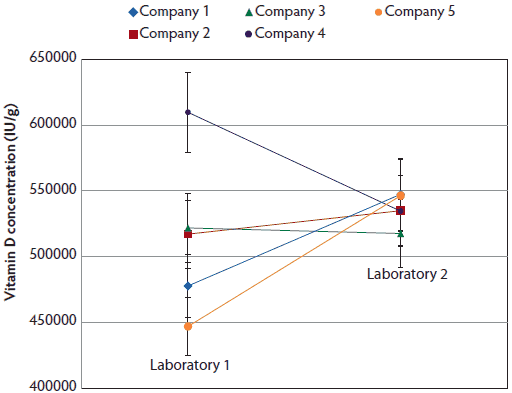
Although the vitamin D concentration varied in the supplement samples evaluated (Figure 2), no samples had vitamin D concentrations statistically lower than the labelled concentration of 500,000 IU per g. No differences between the country of origin or laboratory utilized for testing were detected. Depending on the month of sampling, differences were detected (Figure 3). Premixes collected had lower concentrations in the spring (February, March, and April), than those collected during summer months. Thus, vitamin D premixes varied by sampling date. In addition to these results, an interaction of vitamin D premix supplier and testing laboratory was detected with 95% confidence limits (Figure 4).
Figure 2: Comparison of bulk vitamin D sample concentrations (IU/g) for inclusion in swine diets over a 9-month period, assayed by two different laboratories. A total of 45 samples were assayed from five independent suppliers. All bulk samples had an expected concentration of 500,000 IU/g. No vitamin D concentrations were statistically lower than the expected range (P < .05; ANOVA).
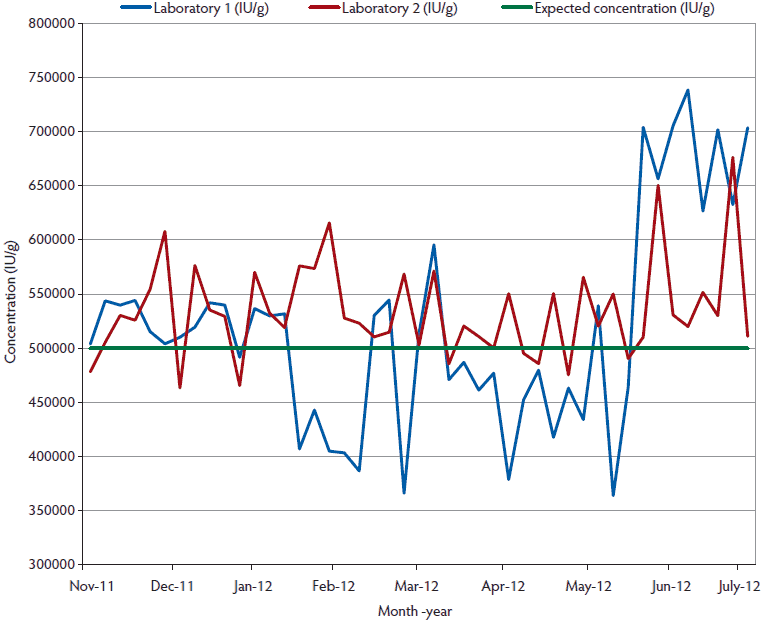
Figure 3: Month effect of vitamin D swine premix samples for dietary inclusion from five different suppliers. All premix samples had an expected concentration of 500,000 IU/g. Vitamin D concentration in samples received in February (P = .04), March (P < .01), and April 2012 (P = .03) were statistically lower than those received in June and July 2012 (least squares means).
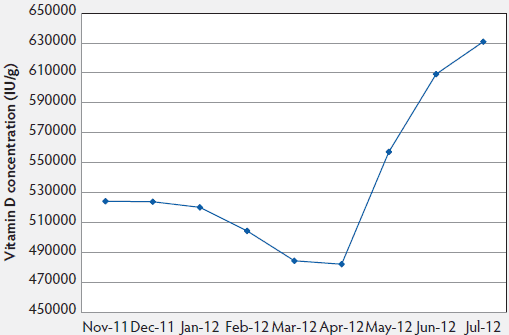
Figure 4:. The interaction of bulk vitamin D source and the laboratory that performed an assay for vitamin D content. Five bulk vitamin D sources (each sampled nine times over 9 months), each with an expected concentration of 500,000 IU/g, were divided into two equal aliquots and tested for vitamin D content at two laboratories.
Discussion
The two sources of vitamin D available to swine are dietary supplementation or synthesis in the skin from 7-dehydrocholesterol. Hypovitaminosis D can be caused by a lack of supply (sunlight or dietary) or lack of physiologic absorption or conversion. When skin is exposed to ultraviolet-B (UV-B) sunlight, 7-dehydrocholesterol is converted to vitamin D3.7 During winter months, the angle of the sun’s light prevents atmospheric penetration of nearly all UV-B irradiation north of 31° latitude. Animals not exposed to sunlight, or in northern latitudes during the winter months, require supplementary vitamin D to prevent potential disease processes such as rickets or osteomalacia. Plant-based diets have low concentrations of endogenous vitamin D. Therefore, swine diets, especially for animals housed in confinement facilities, must be supplemented with vitamin D to meet physiological needs in the absence of UV-B irradiation.
The metabolically active form of vitamin D is 1,25-dihydroxyvitamin D. The concentration of its precursor, 25(OH)D, in serum is considered the best indicator of vitamin D status of an animal. The half-life of the active hormonal form of vitamin D is only 4 to 6 hours, while the precursor’s half-life is approximately 2 to 3 weeks.8 Animals with darker skin pigmentation have higher concentrations of melanin, which is known to block a portion of the UV-B rays reaching the skin, decreasing vitamin D synthesis in the skin. It was interesting to note that serum vitamin D levels were lower in outdoor sows than in the outdoor finisher pigs sampled. The outdoor sows sampled had black skin, while the finishers were of white breeds, presumably illustrating the role melanin has in blocking the conversion of 7-dehydrocholesterol to vitamin D in the skin.
Results of this serum survey provide evidence that 25(OH)D concentrations may be highly variable across all ages of confined swine. The combined mean calculated from the January and June samples by age revealed lower or near low serum 25(OH)D values compared to previously published reference intervals for swine in all groups except finishing pigs. In the nursery category, the combined overall mean for June and January results was 11.4 ng per mL, whereas previous reports recommend 18 to 30 ng per mL 25(OH)D in a pig 2 to 4 weeks of age. For the grower category, the combined overall mean was 19.5 ng per mL, with a reference value of 18 to 30 ng per mL. The serum data also highlight that individual swine within a population may be functioning on suboptimal serum vitamin D concentrations, yet not showing clinical signs of deficiency. Hypovitaminosis D can cause clinical metabolic bone disease if serum 25(OH)D concentrations are low for extended periods. Clinical signs of hypovitaminosis D in swine are related to low blood calcium and phosphorous levels and include tremors, weakness, seizures, and sudden death. Gross lesions may include flexible, rubbery bones, broken bones, and expansion of the costochondral junctions, commonly called the “rachitic rosary.”
The subset of outdoor pigs tested had significantly greater 25(OH)D levels (P < .01) than their counterparts raised in confinement. The complementary outdoor pig samples raise the question as to whether current diet formulations are providing adequate dietary vitamin D for physiological needs of animals raised in confinement. Implications of subclinical hypovitaminosis D in swine are currently unknown. However, studies in human medicine indicate the importance of vitamin D in anti-cancer regimens and a beneficial function in the immune system.1,4,9 Vitamin D insufficiency could diminish immunological response to naturally occurring disease insults or to vaccination. Researchers have recently been successful in treating dairy cows with intramammary 25(OH)D doses for mastitis;10 but the efficacy of vitamin D treatment administered to enhance immunity is unknown at this time.
The two forms of vitamin D available to swine are either sunlight and conversion within the skin or dietary supplements. Because the majority of US swine production is indoors, the focus needs to be on vitamin D supplementation. Several types of supplements are available. These include powdered supplements that can be mixed into total feed rations (as evaluated in this project), oral drenches for piglets at processing or weaning, and liquid products that can be supplied through drinking water.
The timing of supplementation and quality of supplements are important considerations. Some sources of vitamin D3 added to feed may contain large quantities of inactive metabolites, such that the quantity of pure vitamin D has been overestimated.11 Laboratory tests to measure vitamin D concentrations in feed supplements have been challenging. Results of various analytical methods are variable.12 High-performance liquid chromatography with UV detection is considered the gold standard for quantifying vitamin D3, yet various extraction techniques are available and may result in different quantities detected by similar analytic methods. Therefore, accurate analytical test protocols are essential to determine the active amount of vitamin D present. In this study, we found that two specific laboratories provided different results for the same sample. An interaction between company laboratory and dietary premix source was detected. However, our results also showed that no single premix or supplier (US or foreign) was associated with a significantly lower than expected concentration of vitamin D3 in assays from either laboratory.
Vitamin D3 is susceptible to degradation by heat and moisture, especially if direct contact occurs with minerals such as ferrous sulfate and manganese oxide.13 Several reports have indicated that feedstuffs contaminated with mold or mycotoxins may be associated with rickets. In chickens, it is thought that these factors interfere with absorption of vitamin D from the intestinal tract, or possibly interfere with metabolism of vitamin D.14 Therefore, managing vitamin D premixes prior to and after inclusion into vitamin-trace mineral premixes or complete swine diets is an important quality-control procedure to prevent hypovitaminosis D. Anecdotally, this quality-control concern was identified as a contributor in two metabolic bone disease cases in swine in Iowa.15
Not only is the quality of vitamin D supplementation important, but the timing of supplementation is crucial. Chronic hypovitaminosis D prevents dietary calcium and phosphorus from being absorbed efficiently, resulting in hyperparathyroidism, which causes calcium stores in the bone to become depleted to help restore blood calcium concentrations. Once clinical signs and physiologic changes are observed, feed supplementation will not quickly reverse the effects of hypovitaminosis D.
Dietary vitamin D recommendations for swine from the National Research Council (NRC) range from 150 to 220 IU (3.75 to 5.50 μg) per kg of diet (depending on stage of production).16 The 2012 NRC requirements were increased to 800 IU (20 μg) per kg of diet for sows, but were not adjusted for growing pigs. One also may have to consider whether all studies determining the vitamin D requirement of pigs were performed in the absence of UV-B irradiation. The swine industry typically feeds three to five times the NRC-recommended level of vitamin D in the diet, and still the 25(OH)D serum concentration in confined herds is well below that of pigs raised outdoors. The importance of this observation to health and productivity of the pigs remains to be determined. In a study with broiler chicks, it was noted that the NRC recommendation was three to five times less than the amount of vitamin D needed to support the rapid growth of these birds in a low-stress environment.17
Data from the serum survey confirm lower values of serum 25(OH)D than historical reference ranges, raising the possibility that current feeding or production practices provide inadequate vitamin D to swine or that there is a need to re-evaluate the requirements or the reference ranges. It seems especially critical to evaluate the requirement, since no individual supplement or supplier (US or foreign) was associated with a concentration of vitamin D statistically lower than the expected 500,000 IU per g. However, the interaction of laboratory and sample assay illustrated the importance of the laboratory testing method used to obtain accurate results for the same sample.
Implications
• Subclinical hypovitaminosis D is more common than previously thought.
• Under the conditions of this study, vitamin D premixes supplied to swine nutrition are not significantly lower in vitamin D than the expected range.
Acknowledgements
The authors would like to thank the Iowa Pork Producers Association for funding this project and the following nutritional companies for providing samples: Archer Daniels Midland, Devenish Nutrition, DSM Nutrition, Ralco Nutrition, and Trouw Nutrition. We would also like to thank Drs Steve Henry and Jeff Blythe for their knowledge and assistance on this project and also DSM for bulk-source vitamin D analysis testing.
Conflict of interest
Authors Goff and Horst are co-owners of GlycoMyr, Inc, a company that manufactures a vitamin D supplement for neonatal and weanling pigs.
Disclaimer
Scientific manuscripts published in the Journal of Swine Health and Production are peer reviewed. However, information on medications, feed, and management techniques may be specific to the research or commercial situation presented in the manuscript. It is the responsibility of the reader to use information responsibly and in accordance with the rules and regulations governing research or the practice of veterinary medicine in their country or region.
References
1. van Etten E, Stoffels K, Gysemans C, Mathieu C, Overbergh L. Regulation of vitamin D homeostasis: implications for the immune system. Nutr Rev. 2008;66:S125–S134.
2. Greco DS, Stabenfeldt GH. Endocrine glands and their function. In: Cunningham JG, Klein BG, eds. Textbook of Veterinary Physiology. 4th ed. St Louis, Missouri: Saunders Publishing; 2007:428–464.
3. Thompson K. Bones and joints. Metabolic bone disease. In: Maxie MG, eds. Pathology of Domestic Animals. 5th ed. Philadelphia, Pennsylvania: Elsevier; 2007:67–88.
4. Cannell JJ, Hollis BW. Use of vitamin D in clinical practice. Altern Med Rev. 2008;13:6–20.
5. Madson DM, Ensley SM, Gauger PC, Schwartz KJ, Stevenson GW, Cooper VL, Janke BH, Burrough ER, Goff JP, Horst RL. Rickets: case series and diagnostic review of hypovitaminosis D in swine. J Vet Diagn Invest. 2012;24:1137–1144.
6. Hollis BW, Kamerud JQ, Selvaag SR, Lorenz JD, Napoli JL. Determination of vitamin D status by radioimmunoassay with an 125I-labeled tracer. Clin Chem. 1993;39:529–533.
7. Crenshaw TD. Calcium, phosphorus, vitamin D and vitamin K. In: Lewis AJ, Southern LL, eds. Swine Nutrition. 2nd ed. Boca Raton, Florida: CRC Press; 2001:187–212.
8. Holick MF. Vitamin D status: measurement, interpretation, and clinical application. Ann Epidemiol. 2009;19:73–78.
9. Van Belle TL, Gysemans C, Mathieu C. Vitamin D in autoimmune, infectious and allergic diseases: a vital player? Best Pract Res Clin Endocrinol Metab. 2011;25:617–632.
10. Lippolis JD, Reinhardt TA, Sacco RA, Nonnecke BJ, Nelson CD. Treatment of an intramammary bacterial infection with 25-hydroxyvitamin D3. PLoS One. 6(10): e25479. doi:10.1371/journal.pone.0025479.
*11. Baker DH. Nutrient bioavailability in feedstuffs: Methodology for determining amino acid and B-vitamin availability in cereal grains and soybean meal. Proc Georgia Nutrition Conference. 1978:1–12.
12. Hollis BW. Measuring 25-hydroxyvitamin D in a clinical environment: challenges and needs. Am J Clin Nutr. 2008;88:507S–510S.
*13. Hirsch A. Vitamin D - History, manufacture, analysis and metabolism: An overview. In: Vitamins – The Life Essentials. National Feed Ingredients Association (NIFA), Des Moines, Iowa. 1982.
14. Ledoux DR, Brown TP, Weibking TS, Rottinghaus GE. Fumonisin toxicity in broiler chicks. J Vet Diagn Invest. 1992;4:330–333.
*15. Safley W, Armbrecht P. A case of rickets in pigs. Proc Winter IVMA. Altoona, Iowa. 2013;179–182.
16. National Research Council. Nutrient Requirements of Swine.10th rev ed. Washington, DC: National Academic Press; 1998:115.
*17. Rowland R. Vitamin D Function. In: Vitamins – The Life Essentials. National Feed Ingredients Association (NIFA), Des Moines, Iowa. 1982.
* Non-refereed references.
