| Brief communication | Peer reviewed |
Cite as: Pepin B, Liu F, Main R, et al. Collection of oral fluid from individually housed sows. J Swine Health Prod. 2015;23(1):35–37.
Also available as a PDF.
SummaryOral-fluid sampling was attempted on 513 individually housed, mixed-parity sows. Younger sows (P < .01) and re-sampling (P < .001) were associated with successful collection. Diagnostic results on samples collected on 2 successive days were correlated. Oral-fluid sampling in breeding herds would facilitate surveillance and animal welfare. | ResumenSe intentó tomar muestras de fluidos orales en 513 hembras de paridad mixta, alojadas individualmente. Las hembras más jóvenes (P < .01) y el re-muestreo (P < .001) se asociaron con la recolección exitosa. Se correlacionaron los resultados diagnósticos de muestras recolectadas en 2 días consecutivos. El muestreo de fluido oral en hatos de cría facilitaría la vigilancia y el bienestar animal. | ResuméUn échantillonnage de fluide oral fut tenté sur 513 truies de parité mixte logées individuellement. Les truies plus jeunes (P < 0,01) et un ré-échantillonnage (P < 0,001) étaient associés à un prélèvement réussi. Les résultats diagnostiques sur des échantillons prélevés 2 jours consécutifs étaient corrélés. L’échantillonnage de fluide oral dans des troupeaux de reproducteurs faciliterait la surveillance et le bien-être des animaux. |
Keywords: swine, oral fluids, surveillance, porcine reproductive and respiratory syndrome virus, enzyme-linked immunosorbent assay
Search the AASV web site
for pages with similar keywords.
Received: March 13, 2014
Accepted: June 30, 2014
Testing oral-fluid samples by antibody-based assays or polymerase chain reaction- (PCR-) based assays is an effective and efficient method to survey for a variety of infectious agents, including porcine reproductive and respiratory syndrome virus (PRRSV),1-5 influenza A virus,6-9 porcine circovirus type 2,10 and others.11-13 Oral fluids are commonly collected from pens of animals,14 but can also be collected from individual animals. Thus, it has been reported that most boars could be trained for oral-fluid collection by providing the boars repeated exposure to the collection process.1,5
The premise of this study was that collection of oral fluid on commercial sites of individually housed sows could facilitate breeding-herd surveillance for infectious diseases and improve animal and worker welfare by reducing the need to restrain sows for sample collection. However, to the knowledge of the authors, there is no published data on the collection of oral-fluid samples from individually housed sows and, likewise, there is little data on the repeatability of test results on successive oral-fluid samples collected from the same individual in commercial settings. Therefore, the purpose of this study was not only to evaluate the concept that oral-fluid collection in breeding herds is plausible, but also to provide basic collection parameters in relation to parity, a training effect, and diagnostic repeatability.
Materials and methods
The study was conducted with the approval of the Iowa State University Institutional Animal Care and Use Committee.
The study involved 513 individually housed, mixed-parity, gestating sows on two separate commercial farms. No criteria or specifications were used to select animals for participation. The only requirement was that oral fluids had not previously been collected from any of these animals, ie, they were “untrained” for rope collection. Three parameters were of interest: the relationship between sow age (parity) and successful oral-fluid collection, the effect of re-sampling (“training”) on collection, and the repeatability of diagnostic test results on two successive oral-fluid samples collected from the same animal.
The study was carried out by attempting oral-fluid collection on 2 successive days under the same conditions, ie, ropes were placed at approximately 7:00 am, prior to feeding. Oral fluids were collected by hanging a ⅝ -inch (1.59-cm) diameter 100% cotton rope at the front of each crate for 30 to 45 minutes. To harvest the oral fluid, the rope was first gathered in a plastic bag and then grasped tightly while pulling the rope from the bag. A volume of ≥ 1.0 mL was defined as a successful collection. After sampling was completed, paired oral-fluid samples (Day 1 and Day 2) from 48 animals were randomly selected by a random number generator on the basis of sow sequence number from the order in which the ropes were placed for the successfully collected animals. The selected samples were then completely randomized using a random number generator, submitted to the Iowa State University Veterinary Diagnostic Laboratory (ISU-VDL), and tested for PRRSV by real-time reverse transcriptase PCR (RT-PCR) (TetraCore, Inc, Rockville, Maryland) and for anti-PRRSV antibodies (HerdChek X3 Oral Fluid ELISA; Idexx Laboratories, Inc, Westbrook, Maine) using procedures routinely performed in the laboratory.
The effect of sow age (parity) and re-sampling (training) on successful oral-fluid collection was analyzed using a logistic regression model, logit(p) = α + β1×1 + β2×2 + β3×1×2, where P = probability of successful oral-fluid collection; α = intercept; β1 = regression coefficient for day; β2 = regression coefficient for parity; and β3 = regression coefficient for interaction of parity and day (SAS version 9.2; SAS Institute Inc, Cary, North Carolina). In this model, day, parity, and the interaction of parity and day are fixed effects and sow ID is a random effect. This logistic regression model was also used to predict oral-fluid collection success from the collected data. Logistic regression was used in the analysis because the logit link provided the means to evaluate the probability of successful oral-fluid collection (yes or no) in the context of the covariates that could affect this probability. This approach factored in the influence of day, sow parity, the interaction of day and parity, and the random effects of individual animals while accounting for the uneven distribution of sows in each parity level, providing a better prediction of success rates by parity than the raw field data alone. To analyze the diagnostic repeatability of diagnostic test results, a Pearson’s correlation coefficient was used. A value of P < .01 was considered statistically significant.
Results
Oral fluids were collected on Day 1 from 119 of 513 individually housed sows (23.2%). On Day 2, samples were collected from 245 of the same 513 animals (47.8%). Only four animals that provided a successful collection on Day 1 did not provide a sample on Day 2. Parity was associated with oral-fluid collection (P < .01; logistic regression), with lower collection success observed at higher parities (Table 1). The total number of animals from which an oral-fluid sample was collected was significantly higher on Day 2 than on Day 1 (P < .001; logistic regression). This increase in response was observed at all parity levels.
Table 1: Percent success of oral-fluid collection from individual sows in individual housing by parity and by Day 1 and Day 2 of collection*
| % successful oral-fluid collection | |||||
|---|---|---|---|---|---|
| Actual collection | Predicted collection† | ||||
| Parity‡ | No. of sows | Day 1 | Day 2§ | Day 1 | Day 2 |
| 0 | 41 | 14.6 | 36.6 | 29.5 | 61.8 |
| 1 | 89 | 34.8 | 67.4 | 25.1 | 57.2 |
| 2 | 94 | 25.5 | 50.0 | 21.3 | 52.4 |
| 3 | 71 | 33.8 | 56.3 | 17.8 | 47.5 |
| 4 | 72 | 16.7 | 47.2 | 14.9 | 42.8 |
| ≥ 5 | 146 | 15.1 | 33.6 | 12.3 | 38.1 |
* Sows were individually housed in conventional gestational confinement, and oral-fluid samples were collected on an individual-animal basis on 2 successive days. A cotton rope was hung directly in front of each sow. Each sow in the study was positioned next to another study animal. Each individual had its own feeder and watering system. A successful collection was defined as collecting an oral-fluid volume ≥ 1.0 mL.
† Predicted oral-fluid collection success was based on analysis of the field collection data using a logistic regression model, (logit(p) = α + β1×1 + β2×2 + β3×1×2), where P = probability of successful oral-fluid collection; α = intercept; β1 = regression coefficient for day; β2 = regression coefficient for parity, and β3 = regression coefficient for interaction of parity and day.
‡ Parity was significantly associated with sampling success (P < .01; logistic regression).
§ Collection rate significantly higher on Day 2 (P < .001; logistic regression).
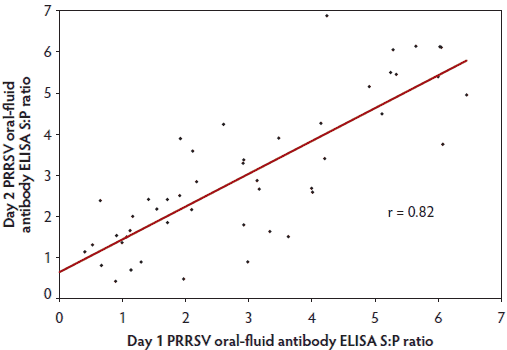
Testing showed that all oral-fluid samples (n = 96 from 48 animals) were negative for PRRSV by RT-PCR, but positive for PRRSV antibody by oral-fluid ELISA. Therefore, the analysis of diagnostic repeatability on paired samples (Day 1 versus Day 2) was based only on the sample-to-positive (S:P) ratios of the PRRS ELISA. The analysis of the ELISA S:P ratios (Figure 1) revealed a strong correlation between Day 1 and Day 2 results (Pearson’s correlation coefficient = 0.82) and no significant difference between days (P > .05; paired t test).
Figure 1: The random selection of 48 sows from the study participants that provided consecutive oral-fluid samples for the 2 days of the study (described in Table 1) showed a strong correlation (Pearson correlation, r = 0.82) between sample-to-positive ratio (S:P) values in the antibody enzyme-linked immunosorbent assay (ELISA) for porcine reproductive and respiratory virus syndrome (PRRSV) with repeat testing on the same individual animals. Each data point represents the S:P ratio values for one animal on Day 1 and Day 2 of the study.
Discussion
Routine collection of oral-fluid samples from individually housed boars has been documented in both experimental and field studies.1,5 In these studies, individual boars were trained for oral-fluid collection by hanging the rope at the front of the pen for 20 minutes daily for 2 or 3 days. Thereafter, most boars were compliant with oral-fluid collection. Although assurance of PRRSV-free semen requires testing by RT-PCR serum samples or blood swabs from boars at the time of semen collection, oral-fluid sampling from non-donor boars provides a mechanism for disease monitoring while avoiding the necessity of collecting blood.1,5 This decreases the frequency of restraining animals for sample collection and increases worker safety.1,15
Although this is a “proof of concept” study, the findings suggested that the behavior seen in boars also applies to individually housed sows in commercial herds. In particular, repeated exposure of sows to the rope produced a measurable training effect regardless of animal age. It was also observed that younger females were more likely to interact with the rope, which is supported by both the observed and the statistically predicted oral-fluid successful collection rates. This suggests the possibility of training animals prior to entry into the breeding herd during isolation or quarantine. Of course, the advantages of oral-fluid collection in boars also apply to sow herds for more consistent and safer disease monitoring.
Accurate surveillance depends on the repeatability and reproducibility of the diagnostic assays used. In this study, quantitative analysis of testing results showed a strong correlation (Pearson’s correlation coefficient of 0.82) between samples collected from the same individuals on 2 consecutive days. This further increases confidence in the process of surveillance in sows using oral-fluid samples.
These baseline results suggest that oral-fluid samples can be collected from individually housed sows, but that further studies on the optimization of oral-fluid collection in the sow unit (gestation and farrowing) would be of value. Potential future studies include further evaluation of training methods and an assessment of the duration of the training effect. Regardless of the approach, more extensive surveillance of the sow herd will be necessary if we are to achieve control of agents such as PRRSV and porcine epidemic diarrhea virus.
Implications
• Oral-fluid collection is most likely to be successful in younger sows.
• Regardless of age, improved collection success on re-sampling suggests that sows could be trained for oral-fluid collection, eg, during quarantine.
• The strong correlation (r = 0.82) observed between PRRS oral-fluid antibody test results on different samples from the same animal strengthens the validity of oral-fluid testing.
• The use of oral fluids for monitoring PRRSV in breeding herds is plausible and could improve the current level of surveillance in most breeding herds by facilitating sample collection from animals and reducing the need to collect blood samples.
Acknowledgements
The authors would like to thank the veterinarians who provided their assistance in finding field sites for participation in this study.
Conflict of interest
None reported.
Disclaimer
Scientific manuscripts published in the Journal of Swine Health and Production are peer reviewed. However, information on medications, feed, and management techniques may be specific to the research or commercial situation presented in the manuscript. It is the responsibility of the reader to use information responsibly and in accordance with the rules and regulations governing research or the practice of veterinary medicine in their country or region.
References
1. Kittawornrat A, Prickett J, Chittick W, Wang C, Engle M, Johnson J, Patnayak D, Schwartz T, Whitney D, Olsen C, Schwartz K, Zimmerman J. Porcine reproductive and respiratory syndrome virus (PRRSV) in serum and oral fluid samples from individual boars: Will oral fluid replace serum for PRRSV surveillance? Virus Res. 2010;154:170–176.
2. Kittawornrat A, Prickett J, Wang C, Panyasing Y, Ballagi A, Rice A, Main R, Johnson J, Rademacher C, Hoogland M, Rowland R, Zimmerman J. Detection of porcine reproductive and respiratory syndrome virus (PRRSV) antibodies in oral fluid specimens using a commercial PRRSV serum antibody ELISA. J Vet Diagn Invest. 2012;24:262–269.
3. Kittawornrat A, Engle M, Panyasing Y, Olsen C, Schwartz K, Ballagi A, Rice A, Lizano S, Wang C, Zimmerman J. Kinetics of the porcine reproductive and respiratory syndrome virus (PRRSV) humoral immune response in swine serum and oral fluids collected from individual boars. BMS Vet Res. 2013;9:61. doi:10.1186/1746-6148-9–61.
4. Kittawornrat A, Panyasing Y, Goodell C, Wang C, Gauger P, Harmon K, Rauh R, Desfresne L, Levis I, Zimmerman J. Porcine reproductive and respiratory syndrome virus (PRRSV) surveillance using pre-weaning oral fluid samples detects circulation of wild-type PRRSV. Vet Microbiol. 2014;168:331–339.
5. Pepin BJ, Kittawornrat A, Liu F, Gauger PC, Harmon K, Abate, S, Main R, Garton C, Hargrove J, Rademacher C, Ramirez A, Zimmerman J. Comparison of specimens for detection of porcine reproductive and respiratory syndrome virus infection in boar studs. Transbound Emerg Dis. 2013. doi:10.1111/tbed.12135.
6. Detmer SE, Patnayak DP, Jiang Y, Gramer MR, Goyal SM. Detection of influenza A virus in porcine oral fluid samples. J Vet Diagn Invest. 2011;23:241–247.
7. Goodell CK, Prickett J, Kittawornrat A, Zhou F, Rauh R, Nelson W, O’Connell C, Burrell A, Wang C, Yoon K-J, Zimmerman JJ. Probability of detecting influenza A virus subtypes H1N1 and H3N2 in individual pig nasal swabs and pen-based oral fluid specimens over time. Vet Microbiol. 2013;166:450–460.
8. Panyasing Y, Goodell CK, Giménez-Lirola L, Kittawornrat A, Wang C, Schwartz KJ, Zimmerman JJ. Kinetics of influenza A virus nucleoprotein antibody (IgM, IgA, IgG) in serum and oral fluid specimens from pigs infected under experimental conditions. Vaccine. 2013;31:6210–6215.
9. Romagosa A, Gramer M, Joo HS, Torremorell M. Sensitivity of oral fluids for detecting influenza A virus in populations of vaccinated and non-vaccinated pigs. Influenza Other Respir Viruses. 2011;6:110–118.
10. Prickett JR, Johnson J, Murtaugh MP, Puvanendiran S, Wang C, Zimmerman JJ, Opriessnig T. Prolonged detection of PCV2 and anti-PCV2 antibody in oral fluids following experimental inoculation. Transbound Emerg Dis. 2011;58:121–127.
11. Giménez-Lirola LG, Xiao CT, Zabala M, Halbur PG, Opriessnig T. Improving ante mortem diagnosis of Erysipelothrix rhusiopathiae infection by use of oral fluids for bacterial, nucleic acid, and antibody detection. J Microbiol Methods. 2013;92:113–121.
12. Mur L, Gallardo C, Soler A, Zimmerman J, Pelayo V, Nieto R, Sánchez-Vizcaíno JM, Aria M. Potential use of oral fluid samples for serological diagnosis of African swine fever. Vet Microbiol. 2013;165:135–139.
13. Ramirez A, Wang C, Prickett JR, Pogranichniy R, Yoon KJ, Main R, Johnson JK, Rademacher C, Hoogland M, Hoffmann P, Kurtz A, Kurtz E, Zimmerman J. Efficient surveillance of pig populations using oral fluids. Prev Vet Med. 2012;104:292–300.
14. White D, Rotolo M, Olsen C, Wang C, Prickett J, Kittawornrat A, Panyasing Y, Main R, Rademacher C, Hoogland M, Zimmerman J. Recommendations for pen-based oral fluid collection in growing pigs. J Swine Health Prod. 2014;22:138–141.
15. Dee S, Deen J. Establishment of a PRRS virus ELISA-negative boar population using previously exposed boars. Vet Rec. 2001;149:678–680.
