| Original research | Peer reviewed |
Cite as: Stiehler T, Heuwieser W, Pfützner A, et al. The course of rectal and vaginal temperature in early postpartum sows. J Swine Health Prod. 2015;23(2):72–83.
Also available as a PDF.
SummaryObjectives: To investigate the course of body temperature in early postpartum sows and possible factors that may influence it, and to examine the influence of a vaginal temperature logger on body temperature by including a control group of sows without loggers. Materials and methods: The study was conducted on a commercial pig farm from January to May 2013. A total of 156 sows received a vaginal temperature logger for 6 days post partum and 43 sows remained without loggers (negative control group). Vaginal temperature was measured at 10-minute intervals. Rectal temperature, feed intake, general condition, and vaginal discharge were evaluated daily. Results: The sows showed a clear circadian rhythm of vaginal temperature, with minimal mean temperature 39.0°C (standard deviation [SD] 0.5°C) from 5:00 am to 6:00 am and maximum mean temperature 39.4°C (SD 0.5°C) from 1:00 pm to 7:00 pm (P < .05). Day post partum (P < .01), time of day (P < .01), age (P < .01), general condition (P < .01), vaginal discharge (P < .01), and treatment for postpartum dysgalactia syndrome (P < .01) had effects on rectal and vaginal temperature. Implications: Measurement of body temperature should be made at the same time every day. Use of vaginal temperature loggers is a practicable method for on-farm studies to gain more information about the course of body temperature in postpartum sows. Body temperature should not be used as the single criterion for the decision to administer medical treatment. | ResumenObjetivos: Investigar el seguimiento de la temperatura corporal en hembras al inicio del post-parto y los posibles factores que puedan influenciarla, y examinar la influencia de un aparto de registro (logger) de la temperatura vaginal en la temperatura corporal al incluir un grupo control de hembras sin loggers. Materiales y métodos: El estudio se realizó en una granja comercial de cerdos de enero a mayo 2013. Un total de 156 hembras recibieron loggers vaginales durante 6 días post parto y 43 hembras permanecieron sin loggers (grupo control negativo). La temperatura vaginal se midió en intervalos de 10 minutos. Diariamente se evaluaron la temperatura rectal, consumo de alimento, condición general, y secreción vaginal. Resultados: Las hembras mostraron un ritmo circadiano claro de la temperatura vaginal, con un promedio mínimo de temperatura de 39.0°C (desviación estándar [SD] 0.5°C) de 5:00 am a 6:00 am y una temperatura máxima promedio de 39.4°C (SD 0.5°C) de 1:00 pm a 7:00 pm (P < .05). Día de postparto (P < .01), hora del día (P < .01), edad (P < .01), condición general (P < .01), secreción vaginal (P < .01), y tratamiento para el síndrome de agalactia post parto (P < .01), tuvieron efectos en la temperatura rectal y vaginal. Implicaciones: La temperatura corporal debería medirse a la misma hora cada día. La utilización de loggers de temperatura vaginal es un método viable para estudios en granja para obtener más información sobre el curso de la temperatura corporal en hembras en postparto. La temperatura corporal no debería utilizarse como el criterio único en la toma de decisión para administrar tratamiento médico. | ResuméObjectifs: Examiner l’évolution de la température corporelle chez des truies en début de période post partum et les facteurs potentiels qui pourraient l’influencer, ainsi qu’évaluer l’influence d’un capteur de température vaginale sur la température corporelle en incluant un groupe témoin de truies sans capteur. Matériels et méthodes: L’étude a été réalisée sur une ferme commerciale de janvier à mai 2013. Au total, 156 truies ont eu un capteur de température vaginale pendant 6 jours en période post-partum et 43 truies n’ont pas eu de capteur (groupe témoin négatif). La température vaginale a été mesurée à des intervalles de 10 minutes. La température rectale, la quantité d’aliment consommé, la condition générale, ainsi que l’écoulement vaginal ont été évalués quotidiennement. Résultats: Les truies ont clairement montré un rythme circadien en ce qui regarde la température vaginale, avec une température minimale moyenne de 39,0°C (écart-type [ET] 0,5°C) entre 05:00 et 06:00 H et une température maximale moyenne de 39,4°C (ET 0,5°C) entre 13:00 et 19:00 H (P < 0,05). Le jour post partum (P < 0,01), le moment de la journée (P < 0,01), l’âge (P < 0,01), la condition générale (P < 0,01), l’écoulement vaginal (P < 0,01), et le traitement pour le syndrome de dysgalactie post-partum (P < 0,01) avaient des effets sur les températures rectale et vaginale. Implications: La prise de température corporelle devrait être faite au même moment à chaque jour. L’utilisation de capteur de température vaginale est une méthode pratique pour les études à la ferme afin d’obtenir plus d’informations sur l’évolution de la température corporelle chez les truies en période post-partum. La température corporelle ne devrait pas être le seul paramètre à utiliser pour décider de l’administration d’un traitement médical.
|
Keywords: swine, postpartum dysgalactia syndrome, rectal temperature, temperature logger, vaginal temperature
Search the AASV web site
for pages with similar keywords.
Received: January 16, 2014
Accepted: August 7, 2014
Postpartum diseases of sows remain an important problem in the pig industry, affecting animal health and welfare of sows and piglets. Many terms are used to describe these disorders, such as mastitis, metritis, and agalactia,1 swine urogenital disease,2 periparturient hypogalactic syndrome,3 or postpartum dysgalactia syndrome (PPDS).4,5 The diverse terms reflect the variability of the etiology and clinical signs, which are mastitis, metritis, constipation, cystitis, anorexia, and pyrexia.6 Today, the term “postpartum dysgalactia syndrome” has been accepted in English-speaking areas to describe this postpartum disorder of sows.4,5,7 A common method to identify sick animals is measurement of rectal temperature in the first 3 days post partum, but temporary hyperthermia is often observed in postpartum sows.8
In recent studies, various methods of continuously measuring body temperature in pigs were evaluated. Hanneman et al9 measured body temperature by a sensor inserted at the base of the ear. In this study, a circadian temperature rhythm was demonstrated and quantified in growing-finishing pigs, but the procedure was invasive. The use of infrared thermography is fast and practical, but not suitable for observation of the health status of individual animals.10,11 Vaginal temperature loggers were recently validated for measuring vaginal temperature in cows12,13 and sows14 after parturition and in gilts after vaccination.15 Only minor differences were observed between rectal and vaginal temperature in cows.12,13 In sows, correlation between rectal and vaginal temperature was high (r = .80; P < .01), but vaginal temperature was 0.3°C higher than rectal temperature, which has to be considered when interpreting measures of vaginal temperature generated with this technology. In gilts, a linear correlation existed both in vaccinated gilts (r = .86, n = 21; P < .001) and non-vaccinated gilts (r = .65, n = 22; P < .001), and most of the differences between rectal and vaginal temperature were within two standard deviations.15
To date, there is a dearth of information about factors that might influence body temperature in early postpartum sows under field conditions. Therefore, the overall objectives of this study were to continuously measure body temperature of postpartum sows in a commercial setting, investigate plausible factors that may influence body temperature, and, in addition, examine whether body temperature in sows showing no overt clinical signs can be considered pyrexia, as has been described for healthy postpartum dairy cows.16
Materials and methods
This study was approved by the Institutional Animal Care and Use Committee of the Clinic of Animal Reproduction, Freie Universität Berlin. Sows were managed according to the guidelines set by the International Cooperation and Harmonisation of Technical Requirements for Registration of Veterinary Medical Products.17
Herd and facilities
The study was carried out on a commercial pig farm with 1370 sows in Brandenburg, Germany. The herd was positive for Actinobacillus pleuropneumoniae, Haemophilus parasuis, Mycoplasma hyopneumoniae, porcine circovirus type 2, and porcine reproductive and respiratory syndrome virus (PRRSV). The sows were regularly vaccinated against H parasuis, Clostridium perfringens Type A, Escherichia coli, and PRRSV. The sows were moved to farrowing crates approximately 7 days prior to expected farrowing, with five farrowing rooms in three barns. The front third of the farrowing crates had solid concrete floors, with a covered and heated region for the piglets and a fully slatted floor in the back region. When moved into the farrowing room, sows were fed a lactation ration (energy, 13.0 MJ per kg; crude protein, 17.5%; crude fiber, 6%; crude ash, 6%; crude oil and fats, 5%) at 6:00 am and at 1:00 pm, with continuous access to water from a nipple drinker. The amount fed was increased after farrowing. Routine management of piglets included ear notching for identification, iron injection (1 mL Belfer iron (III)-hydroxid-dextran-complex, 100 mg per mL; bela-pharm GmbH & Co KG, Vechta, Germany), and castration of the male piglets during the first 6 days. Sows and piglets remained in the crates until the piglets were weaned at 28 days of age.
Study design
Every Thursday, 12 ± 2 sows, including 2 ± 1 gilts (mean ± standard deviation) were enrolled in the study for the duration of 16 weeks. On this farm, most sows farrowed Wednesday and Thursday, with farrowing not complete in many sows until Thursday afternoon. Only sows that had finished farrowing and completely expelled the placenta were included in the study. Included sows farrowed either on Wednesday or Thursday morning. On the first day after farrowing, in 10 ± 2 of these sows, a microprocessor-controlled temperature logger was inserted into the vagina, as recently validated for sows.14 A group of sows enrolled as negative controls (2 ± 1 sows) did not receive a logger. The temperature logger (Minilog 8; Vemco, Ltd, Halifax, Nova Scotia, Canada; size = 92 × 20 mm, weight = 40.5 grams) was attached to a modified vaginal controlled internal drug release device (CIDR-blank; InterAg, Hamilton, New Zealand). A part of the plastic frame of the CIDR was removed and the flexible part was pulled over the logger. A thin cord was attached to the plastic frame of the CIDR so that it could easily be pulled out after use. Before use, the combination of CIDR and logger was submerged in a povidone iodine solution for a minimum of 5 minutes (Braunol; B. Braun, Melsungen AG, Melsungen, Germany) and immediately on removal, without rinsing, inserted into the vagina of the standing or lying sow with the help of a tubular speculum (tubular speculum for pigs (length 40 cm, inner diameter 2.5 cm; WDT, Garbsen, Germany). The logger was pushed through the speculum with a CIDR applicator and positioned caudal to the cervix. This procedure required no restraint or sedation of the study animals. The logger remained in the vaginal cavity for a total of 6 days, measuring vaginal temperature at 10-minute intervals. The number of piglets born alive, stillborn piglets, and the birth weight of the litters were noted at enrolment, with each litter weighed as a group. Piglet mortality, medical treatment of sows, number of piglets weaned, and litter weight at weaning were documented.
Daily observation of the animals, measurement of rectal temperatures, and medical treatment were conducted by the herd manager (investigator), who had completed 3 years of education comparable to a veterinary technician course. Before starting the study, the investigator underwent a 3-day training period with the first author, including 20 sows each day, assessing general health status, feed intake, and vaginal discharge, measuring rectal temperature, and implementing medical treatment. In the first 2 weeks of the study, the first author and investigator examined all study animals together to ensure that the measurements and examinations were performed consistently. In addition, every Thursday, when new study animals were enrolled, observations were performed simultaneously by the first author and the investigator. A data capture form was attached above the farrowing crate of each sow. From day 1 to day 6 of the study, the animals were clinically examined by the investigator. First, feed intake was evaluated by visual examination on a three-point scale (complete intake of feed, partial intake, or no feed intake). Second, the investigator scored general condition of the sow on a three-point scale: healthy, sow was attentive, standing up for feeding, nursing the piglets; slightly reduced, sow seemed apathetic, did not nurse the piglets, ie, remained in ventral recumbency, not allowing the piglets to nurse, but stood up for feeding; severely reduced, sow was somnolent, remained recumbent, did not nurse the piglets, and did not stand up for feeding. Rectal temperature was measured twice daily (morning and afternoon) and vaginal discharge was evaluated by the investigator. Vaginal discharge was characterised as purulent or mucopurulent to simplify scoring. On day 7 of the study, the loggers were removed, which required no restraint or sedation. Litters were reweighed at weaning (day 28).
For analysis, a sow was categorized as ill when three clinical criteria were abnormal: reduced general condition, reduced feed intake, and vaginal discharge with a rectal temperature > 40.0°C.18 Not all sows categorized as ill were given medical treatment, as illness was classified retrospectively after the study ended. The herd manager provided all medical treatment of ill animals to ensure that treatment decisions were comparable. The standard operating procedure for treatment of PPDS included an intramuscular (IM) injection of 5 mg per kg body weight (BW) enrofloxacin (Floxibac, 100 mg per mL; Chanelle Pharmaceuticals Manufacturing Ltd, Loughrea, Co Galway, Ireland) once daily for 3 consecutive days; an IM injection of 50 mg per kg BW metamizol (Metapyrin, 500 mg per mL; Serumwerk Bernburg AG, Bernburg, Germany) once daily for 3 consecutive days; and a single IM injection of 10 mg dinoprost (Dinolytic, 5 mg per mL; Zoetis Deutschland GmbH, Berlin, Germany) on the first day of treatment. Sows with purulent vaginal discharge for more than 1 day, but without other overt clinical signs, received only a single injection of 10 mg dinoprost. For further analysis, the single injection of dinoprost was classified as medical treatment.
After the piglets were weaned, the study sows were routinely moved to the breeding centre for insemination in the following estrus. Pregnancy diagnosis was performed via ultrasound by the herd veterinarian 4 weeks after insemination. The results of this examination were used to analyse the pregnancy rate of animals with and without loggers for a preliminary indication of whether or not use of the vaginal loggers might have a detrimental effect on subsequent reproductive performance of the sows.
Statistical analysis
Data were recorded in Excel (Office 2010; Microsoft Deutschland GmbH, Munich, Germany) and analysed using SPSS for Windows (Version 20.0; SPSS Inc, Munich, Germany). Vaginal temperatures below 38.0°C were considered artefacts and excluded from analysis.
To study plausible factors that might be associated with body temperature (rectal and vaginal temperatures), their effects were tested in a univariate analysis of variance: day postpartum, hour of the day (actual time, vaginal temperature only) or time of day (morning or afternoon, rectal temperature only), age, logger (sows with or without loggers), piglets born alive, litter weight at day 1, litter weight at day 28, mean weight gain of the litter, vaginal discharge, feed intake, general condition, illness, medical treatment, PPDS treatment, and week of study. Fixed effects tested included the day postpartum (1 to 6), hour of the day (1 to 24), or time of day (1 = morning, 2 = afternoon), age (1 = gilt; 2 = parity 2 to 4; 3 = parity 5 or greater), logger (0 = no vaginal logger; 1 = vaginal logger), piglets born alive (eight to 19), vaginal discharge (0 = no vaginal discharge; 1 = vaginal discharge), illness (0 = fewer than three clinical signs, 1 = three clinical signs), medical treatment (0 = no; 1 = yes), PPDS treatment (0 = no; 1 = yes), and week of study (1 to 16). The three-point scales of feed intake and general condition were converted into a two-point scale to minimize the problem of subjectivity. Therefore feed intake (1 = complete intake; 2 = partly or no intake) and general condition (1 = healthy; 2 = slightly or severely reduced) were tested as fixed effects. Litter weight at day 1 and at day 28 and mean litter weight gain were tested as covariates. All variables with a univariable P < .20 were included in a repeated measures linear mixed model with repeating day post partum (one for rectal temperature and one for vaginal temperature, respectively). Models were constructed in a backward stepwise manner using the scale-identity structure. Pearson’s and Spearman’s correlation (ρ) between the parameters were determined. If two variables were highly correlated (r > .5), only the variable with the smaller P value was included in the final model.
Results
A total of 199 sows were included in the study. In 156 sows, a vaginal temperature logger was inserted after parturition. The remaining 43 sows (negative controls) did not receive a temperature logger. A total of 15 sows were excluded from vaginal temperature analysis because of logger losses (n = 10, 6.4%) or technical problems with the loggers (n = 5, 3.2%). These 15 sows, however, were included in the rectal temperature analysis. A total of 34 gilts and 165 pluriparous sows were used in the study, with 100 sows in parities 2 to 4 and 65 sows in parities 5 to 10 (mean parity 3.8, SD 2.3, including gilts).
Temperature loggers recorded 118,127 ten-minute readings of vaginal temperature from 141 sows. Of these readings, 468 (0.4%) were below 38.0°C and excluded from analysis. For further statistical analyses of vaginal temperature, only 18,509 readings from day 1 to 6 were used because rectal temperature was measured and examinations performed during this period, whereas vaginal temperature alone was measured until the morning of day 7.
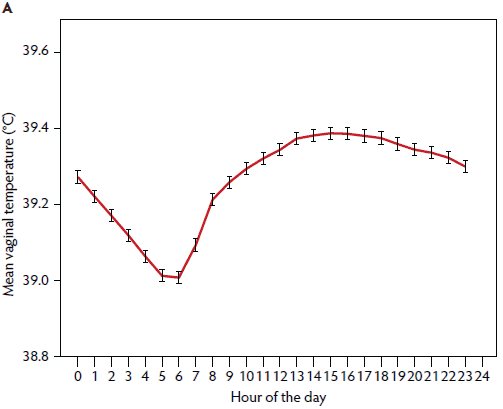
Vaginal temperature showed a clear circadian rhythm, with the lowest temperature in the morning from 5:00 am to 6:00 am and the highest temperature in the afternoon from 1:00 pm to 7:00 pm (Figure 1). Vaginal temperature began to increase parallel with activities in the farrowing pen (eg, feeding at 6:00 am, cleaning of farrowing pens, and medical treatments in the morning). Temperature rose until 3:00 pm and stayed high until 7:00 pm (Figure 1).
Figure 1: In a study conducted on a commercial pig farm, January to May 2013, a total of 156 sows received a vaginal temperature logger for 6 days post partum and 43 sows did not receive a logger (negative control group). Vaginal temperature was measured at 10-minute intervals. Fifteen sows were excluded from vaginal temperature analysis because of logger losses or technical problems with the loggers. Mean vaginal temperature was measured for all sows for all study days (A, n = 141) and for days 1 to 6 post partum (B, n = 141).
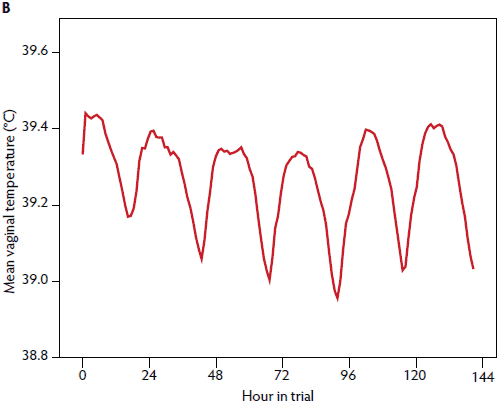
A total of 18 sows (9.0%) were treated within the first 6 days postpartum with dinoprost (n = 10, 5.0%) because of vaginal discharge or with antibiotic and anti-inflammatory drugs for 3 consecutive days as described (PPDS treatment; n = 8, 4.0%).
A total of 191 of the 199 study animals were inseminated after weaning. Seven of the remaining eight sows (4.0%) were slaughtered after weaning due to age (n = 3), lameness (n = 1), small litter size (n = 1), cachexia (n = 1), or infertility (n = 1). One sow died in the farrowing pen in the third week of lactation. Among 151 sows with vaginal temperature loggers, 146 sows became pregnant after the first insemination, and five sows after a second insemination. Among 40 sows without loggers, 39 sows became pregnant after the first insemination, and one sow after a second insemination. The logger had no effect on fertility after weaning (P = 1.0).
Factors associated with rectal temperature
In the univariate analysis, the influence of all plausible factors on rectal temperature was tested (Table 1). Medical treatment and PPDS treatment were correlated (P = .65) and therefore only PPDS treatment was included in the mixed model. Mean litter weight gain was excluded (P = .35). In the repeated measures linear mixed model (Table 1), the day post partum (1 to 6), time of day (morning or afternoon), age (gilt, parity 2 to 4, parity ≥ 5), vaginal temperature logger (yes or no), general condition (healthy or slightly to severely reduced), vaginal discharge (yes or no), PPDS treatment (yes or no), and week of the study (1 to 16) remained significant (Figures 2, 3, 4, 5, and 6). The highest rectal temperature recorded from day 1 to 6 post partum was in gilts, whereas the lowest rectal temperature recorded throughout the trial was in sows of parity ≥ 5 (Table 2; Figure 3).
Table 1: Factors influencing rectal temperature in postpartum sows (n = 199)*
| Factor | P | |
|---|---|---|
| Univariable analysis | ANOVA | |
| Day post partum | < .01 | < .01 |
| Time of day | < .01 | < .01 |
| Age (parity) | < .01 | < .01 |
| Vaginal temperature logger | < .01 | .02 |
| Liveborn piglets | .02 | NS |
| Weight of piglets day 1 | .16 | NS |
| Weight of piglets day 28 | .13 | NS |
| Mean weight gain of piglets | .35 | ND |
| Vaginal discharge | < .01 | < .01 |
| Feed intake | < .01 | NS |
| General condition | < .01 | < .01 |
| Illness† | < .01 | ND |
| PPDS treatment‡ | < .01 | < .01 |
| Week of study | < .01 | .03 |
* Study described in Figure 1. Vaginal temperature was measured at 10-minute intervals. Rectal temperature, feed intake, general condition, and vaginal discharge were evaluated daily. Effects of variables were tested using univariable analysis and repeated measures ANOVA in a multivariate linear mixed model. Piglets were weighed as a litter at 1 and 28 days of age. Litter weight was tested as a covariate.
† Assessment of illness was based on the combination of general behavior, feed intake, and vaginal discharge on at least 1 day of the 6-day trial.
‡ Enrofloxacin (Floxibac, 100 mg/mL; Chanelle Pharmaceuticals Manufacturing Ltd, Loughrea, Co Galway, Ireland), 50 mg/kg, IM, 3 consecutive days; metamizol (Metapyrin, 500 mg/mL; Serumwerk Bernburg AG, Bernburg, Germany), 50 mg/kg BW, IM, 3 consecutive days); and dinoprost (Dinolytic, 5 mg/mL; Zoetis Deutschland GmbH, Berlin, Germany), 10 mg IM, single injection first day of treatment.
ANOVA = analysis of variance; ND = not done; NS = not significant (P > .05); PPDS = postpartum dysgalactia syndrome; BW = body weight; IM = intramuscular.
Table 2: Rectal and vaginal temperatures in °C (mean ± standard deviation) in postpartum sows by parity (n = 199)*
| Parity | Temperature measurement | Days postpartum | |||||
|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | ||
| All n = 199 | RTam (°C) | 39.0 ± 0.5a | 38.9 ± 0.5b | 38.6 ± 0.5c | 38.6 ± 0.4cd | 38.5 ± 0.4d | 38.6 ± 0.4cd |
| RTpm (°C) | 39.1 ± 0.4a | 38.8 ± 0.4b | 38.7 ± 0.4cd | 38.7 ± 0.4cd | 38.7 ± 0.5cde | 38.8 ± 0.5be | |
| VT (°C) | 39.4 ± 0.5a | 39.3 ± 0.5b | 39.3 ± 0.4c | 39.2 ± 0.5d | 39.2 ± 0.5d | 39.3 ± 0.5e | |
| 1 n = 34 | RTam (°C) | 39.2 ± 0.3a | 39.1 ± 0.4a | 38.9 ± 0.3b | 38.9 ± 0.3bd | 38.9 ± 0.3cd | 38.8 ± 0.4cd |
| RTpm (°C) | 39.2 ± 0.2a | 39.1 ± 0.3b | 39.0 ± 0.3cd | 38.9 ± 0.4c | 38.9 ± 0.4c | 39.1 ± 0.4bd | |
| VT (°C) | 39.4 ± 0.3a | 39.5 ± 0.5bc | 39.5 ± 0.4c | 39.5 ± 0.3d | 39.5 ± 0.4d | 39.5 ± 0.5b | |
| 2-4 n = 100 | RTam (°C) | 39.0 ± 0.4a | 38.8 ± 0.5b | 38.7 ± 0.4c | 38.6 ± 0.4d | 38.5 ± 0.4d | 38.6 ± 0.4d |
| RTpm (°C) | 39.1 ± 0.4a | 38.8 ± 0.4b | 38.7 ± 0.4cd | 38.7 ± 0.4c | 38.7 ± 0.4de | 38.8 ± 0.5e | |
| VT (°C) | 39.4 ± 0.5a | 39.4 ± 0.5b | 39.3 ± 0.4c | 39.3 ± 0.5c | 39.2 ± 0.5de | 39.3 ± 0.5ce | |
| > 4 n = 65 | RTam (°C) | 39.0 ± 0.5a | 38.7 ± 0.4b | 38.4 ± 0.4c | 38.4 ± 0.4c | 38.4 ± 0.5c | 38.4 ± 0.4c |
| RTpm (°C) | 39.1 ± 0.6a | 38.7 ± 0.3b | 38.5 ± 0.3c | 38.6 ± 0.5bc | 38.6 ± 0.5b | 38.6 ± 0.5bc | |
| VT (°C) | 39.4 ± 0.5a | 39.1 ± 0.4bc | 39.1 ± 0.4bc | 39.0 ± 0.4d | 39.0 ± 0.4bcd | 39.1 ± 0.5e | |
* Study described in Figure 1 and Table 1. Vaginal temperatures were recorded continuously using a vaginal temperature logger.
Figure 2: Mean rectal temperature (± standard error of the mean) measured morning and afternoon in postpartum sows (n = 199). Study described in Figure 1 and Table 1.
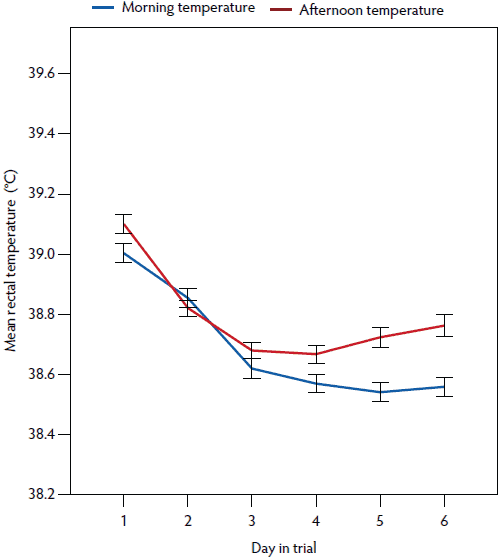
Figure 3: Mean rectal temperature (± standard error of the mean) measured in the morning (A) and mean vaginal temperature measured continuously (B) in postpartum sows. Parity 1, rectal temperature, n = 34, vaginal temperature, n = 28; Parity 2-4, rectal temperature, n = 100, vaginal temperature, n = 71; Parity > 4, rectal temperature, n = 65, vaginal temperature, n = 42. Study described in Figure 1 and Table 1.
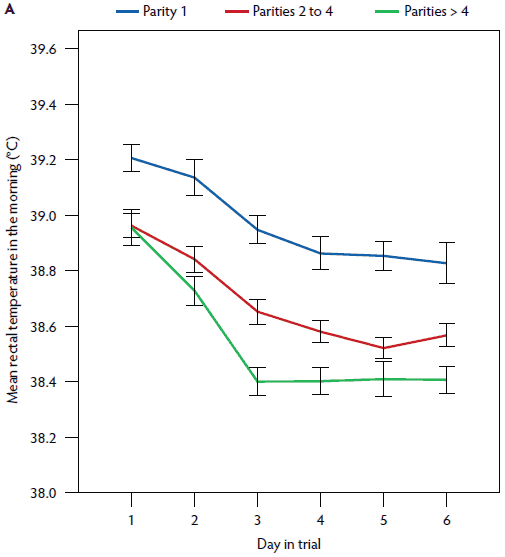
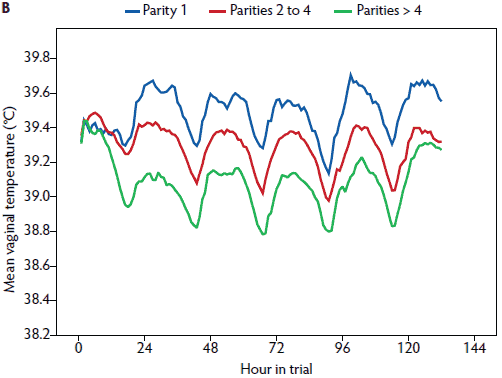
Figure 4: Mean rectal temperature (± standard error of the mean) measured in the morning (A) and in the afternoon (B) in postpartum sows with (n = 141) and without (n = 43) vaginal temperature loggers. Study described in Figure 1 and Table 1.
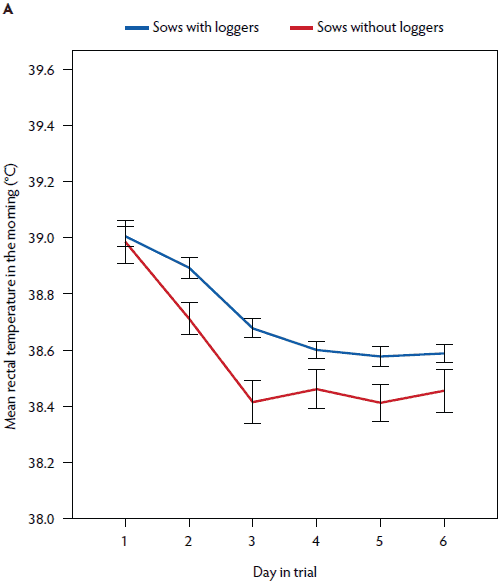
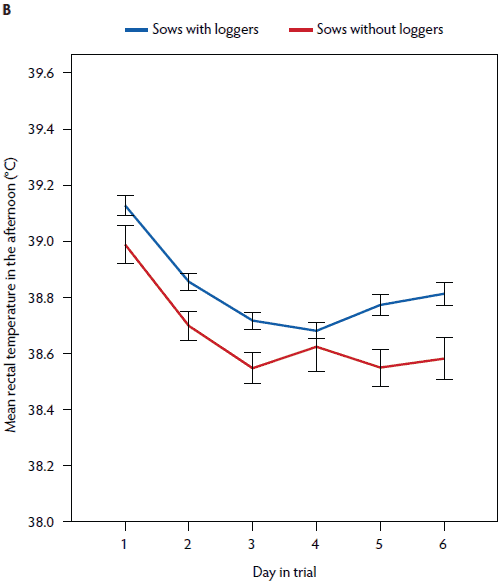
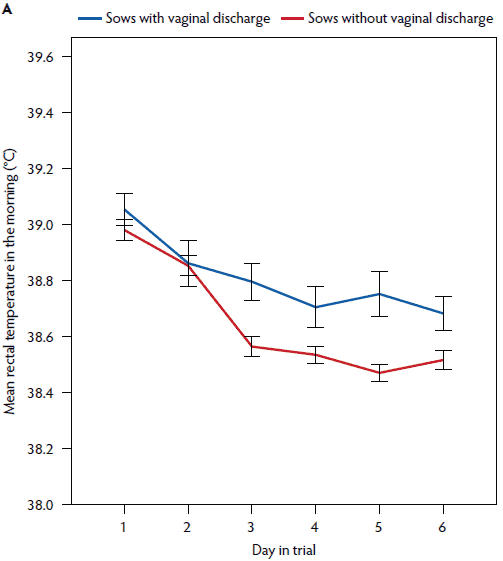
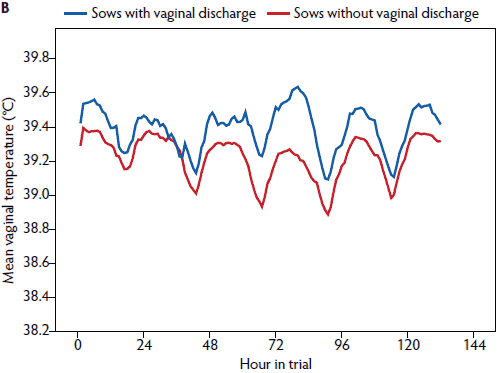
Figure 5: Mean rectal temperature (± standard error of the mean) measured in the morning (A) and mean vaginal temperature measured continuously (B) in postpartum sows with vaginal discharge (rectal temperature, n = 58; vaginal temperature, n = 33) and without vaginal discharge (rectal temperature, n = 141; vaginal temperature, n = 108).
abcde Means within a row with different superscripts differ (P < .05; repeated measures analysis of variance).
RTam = rectal temperature measured in the morning; RTpm = rectal temperature measured in the afternoon; VT = vaginal temperature measured continuously using temperature logger (24-hour average).
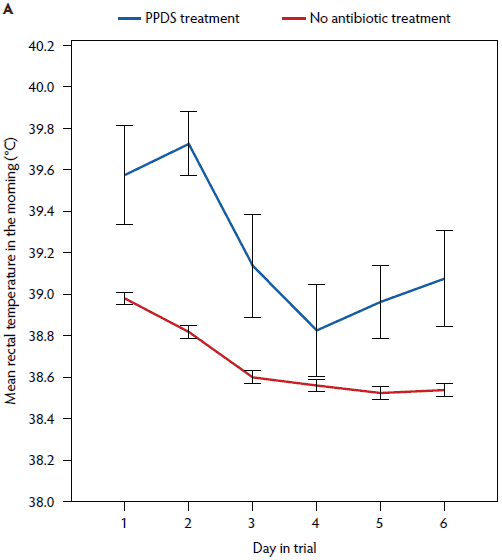
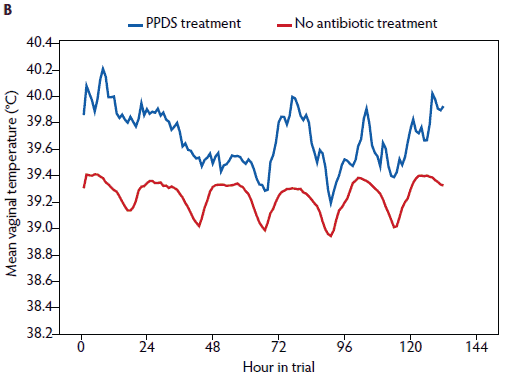
Figure 6: Mean rectal temperature (± standard error of the mean) measured in the morning (A) and vaginal temperature measured continuously (B) in sows treated for postpartum dysgalactia (PPDS) (rectal temperature, n = 8; vaginal temperature, n = 7) and sows not treated for PPDS (rectal temperature, n = 191; vaginal temperature, n = 134). PPDS treatment described in Table 1.
Factors associated with vaginal temperature
In a univariate analysis, all variables tested had an effect on vaginal temperature (Table 3). Litter weight on day 28 and the mean litter weight gain were correlated (r = .88), and therefore mean litter weight gain was excluded from further analysis. In the repeated measures linear mixed model (Table 3), day post partum (1 to 6), hour of day (1 to 24), age (gilt, parity 2 to 4, parity ≥ 5), feed intake (complete or partly to no intake), general condition (healthy or slightly to severely reduced), vaginal discharge (yes or no) and PPDS-treatment (yes or no) remained significant (Figures 1, 3, 5, and 6). The factor “age” was associated with vaginal temperature. Thus, the highest vaginal temperature recorded from day 1 to 6 post partum was in gilts, and the lowest vaginal temperature throughout the trial was in sows at parity ≥ 5 (Table 2, Figure 3).
Table 3: Factors influencing vaginal temperature in postpartum sows (n = 141)*
| Factor | P | |
| Univariable analysis | Repeated measures ANOVA | |
| Day post partum | < .01 | < .01 |
| Time of day | < .01 | < .01 |
| Age (parity) | < .01 | < .01 |
| Liveborn piglets | .02 | NS |
| Weight of piglets at day 1 | < .01 | NS |
| Weight of piglets at day 28 | < .01 | NS |
| Mean weight gain of piglets | < .01 | ND |
| Vaginal discharge | < .01 | < .01 |
| Feed intake | < .01 | < .01 |
| General condition | < .01 | < .01 |
| Illness† | < .01 | NS |
| PPDS treatment | < .01 | < .01 |
| Week of study | < .01 | NS |
* Study, statistical analysis, and PDDS treatment described in Figure 1 and Table 1.
† Illness based on the combination of general behavior, feed intake, or vaginal discharge on at least 1 day of the 6-day trial.
PPDS = postpartum dysgalactia syndrome; NS = not significant (P > .05); ND = not done.
Fever in postpartum sows
In summary, rectal temperature in the first 6 days post partum was > 39.5°C at least once in 59 of 199 sows (29.6%). Among these, 17 sows (8.5%) had a rectal temperature > 40.0°C. In 127 of 141 sows (90.1%), at least one vaginal temperature measurement was > 39.5°C, the threshold frequently used in previous studies.1,6 Among these 127 sows, vaginal temperature was between 39.8°C and 40.0°C (68.8%), between 40.1°C and 40.3°C (61.7%), and > 40.3°C (40.4%) at least once in 97, 87, and 57 sows, respectively.
Discussion
A sufficient number of sows with a vaginal temperature logger (n = 156) were used in this study to examine the course of continuously measured vaginal temperature, because information on rectal temperature based on repeated measures at certain time points is already available.19 In former studies, temperature sensors for measuring body temperature were often inserted under general anesthesia, eg, near the carotid artery20 or the femoral or pulmonary artery, in the urinary bladder, or by the tympanic method,21 and animals were housed under laboratory conditions.9,15 In this study, temperature loggers inserted into the vagina allowed continuous recording of vaginal temperature under field conditions, as recently validated for cows and sows.13,14 Previous trials used only three,22 nine,9 27,23 or 43 animals.15 Furthermore, a larger number of sows (n = 141), including gilts (n = 28) were used in this study to investigate the effect of plausible factors on vaginal temperature. The loggers were attached to a modified vaginal CIDR device as described for cows, dogs, and sows.12,14,24 This minimized expulsion of loggers from the vagina during the study period (10 loggers; 6.4%), while in another study, loggers were not fixed in the vagina and slid out easily.15 In the current study, insertion of the loggers was minimally invasive and did not require anaesthesia. To the knowledge of the authors, this is the first study using this methodology in sows for 6 days post partum.
Vaginal temperatures showed a clear circadian rhythm. It can be speculated that the minimum temperature in the morning was interrupted by starting of activities in the farrowing room, eg, feeding. In the afternoon, body temperature decreased more slowly because of thermal storage and physiological activity of the sow.
A similar circadian temperature rhythm has been described previously.9 In a small study, body temperature in eight of nine growing-finishing swine exhibited a circadian rhythm with an amplitude (range between the lowest and highest values of mean temperature) of 0.18°C ± 0.02°C, mean body temperature of 38.7°C ± 0.24°C, and a maximum at 7:44 pm.9 However, on the basis of frequent body temperature measurements by a temperature transmitter at 30-minute intervals in 10- to 14-week-old pigs for 2 to 3 consecutive days, it was concluded that in pigs there is a very little innate circadian rhythm and that the observed temperature variations are mainly related to feeding and activity.23 In the present study, a circadian rhythm in sows was observed during the first 6 days post partum with an amplitude of 0.4°C. It was not possible, however, to determine whether this variation was based on an innate circadian rhythm or related to feeding or activity or both.
Infections, trauma, and injury result in an adaptive response that includes loss of appetite, apathy, and fever.25 Fever is triggered by the release of endogenous pyrogens from different regions of macrophage-like cells. These pyrogens include cytokines IL-1 and IL-6, which act at the level of the anterior hypothalamus to raise the thermoregulatory set point. In a previous study,1 it was suggested that it is possible to predict the occurrence of PPDS from elevation of rectal temperature, which occurs earlier than other clinical signs (eg, vaginal discharge, mastitis, or emaciated piglets as a consequence of insufficient milk production). It was postulated that rectal temperature > 39.4°C was an appropriate threshold to administer preventive medical treatment such as antibiotic and anti-inflammatory drugs.1 A previous publication demonstrated a physiological hyperthermia after farrowing.8 In this study, 59 (29.6%) and 17 (8.5%) of 199 sows had rectal temperatures > 39.5°C and > 40.0°C, respectively, and 57 (40.4%) sows had vaginal temperatures > 40.3°C. Only eight of these sows received antibiotic and anti-inflammatory treatment. Most of the sows with such high temperatures showed no signs of illness, eg, apathy, no feed intake, or vaginal discharge, and were therefore not treated. As evidenced by the subsequent reproductive performance of the sows, these high body temperatures might not have negative consequences. However, this fact needs to be studied on a large scale with more sows and more farms. In the study of Elmore et al,26 mean pre-farrowing body temperature of 12 mixed-breed sows was 38.83°C ± 0.3°C. Mean postfarrowing temperature was 0.6°C to 1.2°C higher and temperature returned to pre-farrowing values after piglets were weaned. These investigators measured body temperature with surgically implanted temperature transmitters in the left paralumbar fossa. King et al19 also demonstrated that sows have a significantly higher rectal temperature after farrowing (39.3°C ± 0.7°C) than before farrowing (38.3°C ± 0.6°C). Their study was conducted on 217 sows in which rectal temperature was measured three times a day. The authors speculated that the higher temperatures were caused by the physiological inflammatory reaction accompanying involution of the uterus and increasing mammary gland metabolism. Interestingly, they also postulated that healthy sows may have a body temperature > 39.7°C and that high rectal temperature without other clinical signs should not be considered evidence of disease.19 Our results are in agreement, ie, in a high number of apparently healthy sows, body temperature exceeded 39.5°C, the common threshold for pyrexia. In the study of King et al,19 a glass mercury thermometer was used, whereas we used a digital thermometer. The comparison must be interpreted with care, as it is possible that use of different methods of measuring rectal temperature biased the results. To date, it is an accepted fact that lactation hyperthermia occurs in sows after farrowing,8 hence the difficulty in defining body temperature limits for medical treatment of sows. The results of this study provide further evidence that diagnosis of PPDS in sows should include a combination of clinical signs, eg, lethargy, diminished milk production, reduced appetite, and vaginal discharge, as previously described.27 It is emphasized, however, that evaluation of clinical signs might be more subjective than measurement of rectal temperature, which is a repeatable measure.14 This has already been demonstrated for vaginal discharge in cows.28 General health condition, feed intake, and vaginal discharge are subjective parameters, but more objective parameters to validate health or illness of early postpartum sows are limited. Clinical examination of the study animals was conducted only by the investigator to reduce observer bias. In addition, during the first 2 weeks of the study and every Thursday, clinical examinations were performed simultaneously by the herd manager and the first author to provide a consistent evaluation of these parameters. Furthermore, assessments of feed intake, vaginal discharge, and general demeanour are similar to those used in a previous PPDS study as clinical parameters in examination of sows.6
In this study, general behavior, feed intake, and vaginal discharge had a significant influence on rectal and vaginal temperature. Gilts had higher rectal and vaginal temperatures than older sows from day 1 to 6. This result agrees with that of a previous study in cows.29 To the knowledge of the authors, this has not yet been described for sows. The number of piglets born alive, mean litter weight on day 1, mean weight gain, and mean litter weight on day 28 did not influence vaginal and rectal temperatures. These findings agree with those of another study.30 Insertion of a vaginal temperature logger affected rectal temperature. This could be explained as a physiologic reaction to a foreign body.
It remains unclear if the factors used as covariates occurred as a consequence of, or simultaneously with, higher body temperature (eg, illness, medical treatment). However, in these models, this is of minor consequence because both models seek large-scale associations.
Measuring rectal temperature is an objective and repeatable diagnostic method.14 The measuring process, however, should be standardized to achieve comparable values. Temperature should be measured at the same time of day. The results of this study clearly illustrated that the measures should be interpreted with caution, because rectal and vaginal temperature are associated with several factors. Body temperature is only one sign of the PPDS complex and should not be used as the single criterion for the decision to administer medical treatment in early postpartum sows. General health status, feed intake, and vaginal discharge are more subjective, and their usefulness should be evaluated in combination with body temperature in further studies. It must be emphasized that the definition of PPDS needs refinement.
Implications
• Because of the circadian rhythm of body temperature in sows, measurements should be made at the same time every day.
• Use of temperature loggers inserted into the vagina of sows is very practicable for on-farm studies to gain more information about postpartum body temperature.
• Clinical signs such as general behaviour, feed intake, and vaginal discharge are associated with increased body temperature in sows.
• Body temperature should not be used as the single criterion for the decision to administer medical treatment in early postpartum sows.
Conflict of interest
None reported.
Disclaimer
Scientific manuscripts published in the Journal of Swine Health and Production are peer reviewed. However, information on medications, feed, and management techniques may be specific to the research or commercial situation presented in the manuscript. It is the responsibility of the reader to use information responsibly and in accordance with the rules and regulations governing research or the practice of veterinary medicine in their country or region.
References
1. Furniss SJ. Measurement of rectal temperature to predict mastitis, metritis and agalactia (MMA) in sows after farrowing. Prev Vet Med. 1987;5:133–139.
2. Glock XTP, Bilkei G. The effect of postparturient urogenital diseases on the lifetime reproductive performance of sows. Can Vet J. 2005;46:1103–1107.
3. Hoy S. Investigations on influence of different housing factors on frequency of puerperal diseases in sows. Praktische Tierarzt. 2002;83:990–996.
4. Papadopoulos GA, Vanderhaeghe C, Janssens GPJ, Dewulf J, Maes DGD. Risk factors associated with postpartum dysgalactia syndrome in sows. Vet J. 2010;184:167–171.
5. Preissler R, Looft H, Kemper N. The postpartum dysgalactia syndrome: An overview of the genetic predisposition. Tierärztliche Umschau. 2012;67:520–527.
6. Hirsch AC, Philipp H, Kleemann R. Investigation on the efficacy of meloxicam in sows with mastitis-metritis-agalactia syndrome. J Vet Pharmacol Ther. 2003;26:355–360.
7. Kemper N, Gerjets I. Bacteria in milk from anterior and posterior mammary glands in sows affected and unaffected by postpartum dysgalactia syndrome (PPDS). Acta Vet Scand. 2009;51:26–32.
8. Martineau GP, Farmer C, Peltoniemi O. Mammary System. In: Zimmerman JJ, Karriker LA, Ramirez A, Schwartz KJ, Stevenson GW, eds. Diseases of Swine. 10th ed. Ames Iowa: Wiley-Blackwell; 2012:270–293.
9. Hanneman SK, McKay K, Costas G, Rosenstrauch D. Circadian temperature rhythm of laboratory swine. Comp Med. 2005;55:249–255.
10. Traulsen I, Naunin K, Müller K, Krieter J. Application of infrared thermography to measure body temperature of sows. Züchtungskunde. 2010;82:437–446.
11. Dewulf J, Koenen F, Laevens H, de Kruif A. Infrared thermometry is not suitable for the detection of fever in pigs. Vlaams Diergeneeskundig Tijdschrift. 2003;72:373–379.
12. Vickers LA, Burfeind O, von Keyserlingk MAG, Veira DM, Weary DM, Heuwieser W. Technical note: Comparison of rectal and vaginal temperatures in lactating dairy cows. J Dairy Sci. 2010;93:5246–5251.
13. Suthar V, Burfeind O, Maeder B, Heuwieser W. Agreement between rectal and vaginal temperature measured with temperature loggers in dairy cows. J Dairy Res. 2013;80:240–245.
14. Stiehler T, Heuwieser W, Pfützner A, Voigtsberger R, Burfeind O. Repeatability of measurements of the rectal temperature and comparison of vaginal and rectal temperature in puerperal sows [in German]. Tierärztliche Praxis Großtiere. 2013;41:217–224.
15. Schmidt M, Ammon C, Schon P, Manteuffel C, Hoffmann G. Assessment of continuous vaginal logger-based temperature monitoring in fever-induced gilts. Am J Anim Vet Sci. 2013;8:55–65.
16. Burfeind O, Suthar VS, Heuwieser W. Effect of heat stress on body temperature in healthy early postpartum dairy cows. Theriogenology. 2012;78:2031–2038.
17. Hellmann K, Radeloff I. International Cooperation on Harmonisation of Technical Requirements for Registration of Veterinary Medicinal Products (VICH). 2000; http://www.fda.gov/downloads/AnimalVeterinary/GuidanceCompliance Enforcement/GuidanceforIndustry/UCM052417.pdf. Accessed 16 December 2014.
18. Kiss D, Bilkei G. A new periparturient disease in Eastern Europe, Clostridium difficile causes postparturient sow losses. Theriogenology. 2005;63:17–23.
19. King GJ, Willoughby RA, Hacker RR. Fluctuations in rectal temperature of swine at parturition. Can Vet J. 1972;13:72–74.
20. Ingram DL, Mount LE. Effects of food intake and fasting on 24-hourly variations in body temperature in young pig. Pflügers Archiv: European Journal of Physiology. 1973;339:299–304.
21. Hanneman SK, Jesurum-Urbaitis JT, Bickel DR. Comparison of methods of temperature measurement in swine. Lab Anim. 2004;38:297–306.
22. Jessen C, Kuhnen G. Seasonal variations of body temperature in goats living in an outdoor environment. J Therm Biol. 1996;21:197–204.
23. Ingram DL, Legge KF. Variations in deep body temperature in the young unrestrained pig over the 24 hour period. J Physiol-London. 1970;210:989–998.
24. Maeder B, Arlt S, Burfeind O, Heuwieser W. Continuous vaginal temperature measurement in bitches before parturition. Reprod Biol. 2013;13(Suppl2):31–32.
25. Kluger MJ, Kozak W, Conn CA, Leon LR, Soszynski D. Role of fever in disease. In: Kluger MJ, Bartfai T, Dinarello CA, eds. Molecular Mechanisms of Fever. Annals of the New York Academy of Sciences. 1998;856:224–233. doi:10.1111/j.1749-6632.1998.tb08329.x.
26. Elmore RG, Martin CE, Riley JL, Littledike T. Body temperatures of farrowing swine. JAVMA. 1979;174:620–622.
*27. Larsen I, Thorup F. The diagnosis of MMA. Proc IPVS Cong. Copenhagen, Denmark. 2006;256.
28. Sannmann I, Burfeind O, Suthar V, Bos A, Bruins M, Heuwieser W. Technical note: Evaluation of odor from vaginal discharge of cows in the first 10 days after calving by olfactory cognition and an electronic device. J Dairy Sci. 2013;96:5773–5779.
29. Suthar V, Burfeind O, Bonk S, Voigtsberger R, Keane C, Heuwieser W. Factors associated with body temperature of healthy Holstein dairy cows during the first 10 days in milk. J Dairy Res. 2012;79:135–142.
30. van Gelder KN, Bilkei G. The course of acute phase proteins and cortisol in mastitis metritis agalactia (MMA) of the sow and sow performance. Tijdschrift Voor Diergeneeskunde (Amsterdam). 2005;130:38–41.
* Non-referred reference.
