| Brief communication | Peer reviewed |
Cite as: Seo HW, Duy DT, Park C, et al. Immunological, virological, and pathological evaluation of a single dose versus two doses of a one-dose porcine circovirus type 2 subunit vaccine under experimental conditions. J Swine Health Prod. 2015;23(4):214–219.
Also available as a PDF.
SummaryThis study compared use of a single dose to two doses of a one-dose porcine circovirus type 2 (PCV2) vaccine. Two doses of the PCV2 vaccine administered at 1 and 3 weeks of age (extra-label use) provides no additional protection, compared to one dose administered at 3 weeks of age. | ResumenEste estudio comparó el uso de una dosis única contra dos dosis de la vacuna de una dosis del circovirus porcino tipo 2 (PCV2 por sus siglas en inglés). Dos dosis de la vacuna PCV2 administrada en las semanas 1 y 3 de edad (uso fuera de etiqueta) no proveen protección adicional, comparado con una dosis administrada a las 3 semanas de edad. | ResumésDans cette étude nous avons comparé l’utilisation d’une dose unique à celle de deux doses d’un vaccin à dose unique contre le circovirus porcin de type 2 (CVP2). Deux doses du vaccin CPV2 administrées à 1 et 3 semaines d’âge (utilisation en dérogation) ne fournissent pas de protection additionnelle, comparativement à une administration unique donnée à 3 semaines d’âge. |
Keywords: swine, porcine circovirus type 2, vaccine, PCV2
Search the AASV web site
for pages with similar keywords.
Received: May 29, 2014
Accepted: January 15, 2015
Porcine circovirus type 2 (PCV2) has been identified in association with several conditions in pigs, including postweaning multisystemic wasting syndrome, porcine dermatitis and nephropathy syndrome, porcine reproductive disorders, and porcine respiratory disease complex.1 These syndromes and diseases are collectively referred to as porcine circovirus-associated disease (PCVAD).1 Since the first commercial PCV2 vaccine (Circoflex; Boehringer Ingelheim Vetmedica Inc, St Joseph, Missouri) was introduced in Korea in 2008,2 an additional four commercial PCV2 vaccines have also been marketed: Fostera PCV (Zoetis, Florham Park, New Jersey), Circovac (Merial, Lyon, France), and Porcilis PCV and Circumvent PCV (MSD Animal Health, Summit, New Jersey). Currently, Circumvent PCV is the only two-dose vaccine available in Korea, but it is used rarely because of adverse reactions such as lethargy and loss of appetite (personal communication with producers).
Under Korean field conditions, many swine producers believe that vaccinating twice instead of once provides better PCVAD control. Because PCVAD usually occurs in pigs 6 to 12 weeks of age,2 many swine producers vaccinate pigs twice at 1 and 3 weeks of age with the dose recommended for a one-dose PCV2 vaccine (Circoflex) (personal communication with producers). An alternative vaccination schedule is possible at 2 and 4 weeks of age; however, Korean swine producers prefer 1 and 3 weeks of age, as mandatory classical swine fever vaccination is administered at 4 weeks of age. However, to the knowledge of the authors, no studies have compared single versus dual dosing of a one-dose PCV2 vaccine in Korea. Hence, the objective of this study was to determine the immune response, viral load, and lesions in pigs vaccinated with either a single dose or two doses of a one-dose PCV2 vaccine administered at 1 and 3 weeks of age.
Materials and methods
All animal protocols were approved by the Seoul National University Institutional Animal Care and Use Committee.
Thirty colostrum-fed, crossbred, conventional piglets were purchased at 5 days of age from a commercial farm. Upon arrival at a research facility, all piglets used in this study tested negative for porcine reproductive and respiratory syndrome virus (PRRS virus) and Mycoplasma hyopneumoniae by serological testing (ELISA; PRRS X3 Ab test and M. hyo Ab test, respectively; Idexx Laboratories Inc, Westbrook, Maine). All piglets also tested negative for PCV2 viremia by real-time polymerase chain reaction (PCR) and seronegative against PCV2 by commercial ELISA for PCV2 IgG (Synbiotics, Lyon, France).
A total of 30 pigs were randomly divided into six groups (five pigs per group) using the random number generation function in Excel (Microsoft Corporation, Redmond, Washington) (Table 1). Sample size was calculated assuming a 90% power (1 - β = .90) of detecting a difference at the 5% level of significance (α = .05), which was based on expected results of ELISA antibody titers, virus load determined by PCR, and lymphoid lesions represented by scores.3 The treatment timeline is shown in Table 1. Pigs in Group 1 and Group 2 were administered one 1.0-mL dose of Circoflex intramuscularly in the right side of the neck at 3 weeks of age. Pigs in Group 3 and Group 4 were administered two 1.0 mL doses of Circoflex intramuscularly in the same anatomic site, at 1 and 3 weeks of age. At 49 days of age (day 0; day of challenge), each pig in groups 1, 3, and 5 was inoculated intranasally with 2 mL of PCV2b (strain SNUVR000463; 5th passage; 1.0 × 105 median tissue culture infective doses per mL). Group 5 pigs served as the positive control group (challenged but not vaccinated). Group 6 pigs were unchallenged and unvaccinated (no vaccine administered) and served as the negative control group. Groups were housed in separate rooms within the same facility. Blood samples were collected at study days -42, -28, 0, 7, 14, 21, and 42.
Table 1: Means (standard deviation) of lymphoid lesion scores and numbers of lymphoid porcine circovirus type 2 (PCV2) antigen-positive cells in pigs vaccinated with either a single dose or two doses of a one-dose PCV2 vaccine and challenged with PCV2*
| Group | Age at vaccination | Challenge | Lymphoid lesion score† | No. of positive lymphoid cells‡ | |
|---|---|---|---|---|---|
| 1 week | 3 weeks | 7 weeks | |||
| 1 | None | 1 mL | Yes | 0.6 (0.55)a | 6.0 (4.42)a |
| 2 | None | 1 mL | None | 0b | 0b |
| 3 | 1 mL | 1 mL | Yes | 0.4 (0.55)a,b | 4.6 (3.57)a,b |
| 4 | 1 mL | 1 mL | None | 0b | 0b |
| 5 | None | None | Yes | 1.4 (0.54)c | 20.6 (7.27)c |
| 6 | None | None | None | 0b | 0b |
* Group 1 and 2 pigs were vaccinated with a one-dose PCV2 vaccine (Circoflex; Boehringer Ingelheim Vetmedica Inc, St Joseph, Missouri) at 3 weeks of age. Group 3 and Group 4 pigs were vaccinated with two doses of the same one-dose PCV2 vaccine at 1 and 3 weeks of age. Group 1 and Group 3 pigs were inoculated intranasally with a PCV2b strain at 7 weeks of age. Blood samples were collected from pigs with anticoagulant for PCV2-specific interferon-γ-secreting cells and without anticoagulant for serological testing at study days -42, -28, 0 (day of challenge), 7, 14, 21, and 42.
† Pigs in all groups were euthanized at 13 weeks of age. Superficial inguinal lymph node was collected for histopathology and immunohistochemistry. Lymphoid lesion scores: 0 = no lymphoid depletion or granulomatous replacement; 1 = mild lymphoid depletion; 2 = moderate lymphoid depletion; and 3 = severe lymphoid depletion and histiocytic replacement. Scores were compared among groups using Fisher’s exact test.
‡ Numbers of lymphoid cells positive for PCV2 antigen per unit area (0.25 mm2) were counted using an NIH ImageJ 1.45s program (http://imagej.nih.gov/ij/download.html). Numbers of positive cells were compared among groups using Tukey’s test.
abc Within a column, values with different superscript letters are significantly different (P < .05).
The QIAamp DNA Mini Kit (Qiagen Inc, Valencia, California) was used to extract DNA from serum samples. The DNA extracts were used to quantify numbers of PCV2 genomic DNA copies by real-time PCR as previously described.4 The number of copies of PCV2 genomic DNA per mL of serum was converted to log10 for analysis.
All pigs were euthanized for necropsy at day 42. Superficial inguinal lymph nodes were collected for histopathology and immunohistochemistry.
Serum samples were tested using a commercial PCV2 ELISA IgG (Synbiotics) and serum virus neutralization.5 Serum samples were considered positive for PCV2 IgG antibody if the reciprocal ELISA titer was greater than 350, according to the manufacturer’s instructions. Neutralizing antibody (NA) data were converted to log2 for analysis. The numbers of PCV2-specific interferon-γ-secreting cells (IFN-γ-SCs) were determined in peripheral blood mononuclear cells (PBMCs) by the enzyme-linked immunospot (ELISPOT) method as previously described.6 Whole PCV2b (the strain used for challenge) at a multiplicity of infection of 0.01 was used as a stimulant of PBMCs. Phytohemagglutinin (10 µg per mL; Roche Diagnostics GmbH, Mannheim, Germany) and phosphate buffered saline were used as positive and negative controls, respectively.
For morphometric analysis of histopathological lesion scores and numbers of PCV2-positive cells in lymph nodes, the superficial inguinal lymph node was collected from each pig and three sections of that lymph node were examined blindly as previously described.7,8 Lymphoid lesions were scored on a scale from 0 to 3: 0, no lymphoid depletion or granulomatous replacement; 1, mild lymphoid depletion; 2, moderate lymphoid depletion; and 3, severe lymphoid depletion and histiocytic replacement.7 The number of lymphoid cells positive for PCV2 antigen per unit area (0.25 mm2) of lymph node was counted using an NIH ImageJ 1.45s program (http://imagej.nih.gov/ij/download.html).8
Continuous data (PCV2 DNA [log10 PCV2 genomic copies per mL] determined by real-time PCR; PCV2 ELISA titer; number of IFN-γ-SCs per 106 PBMCs determined by ELISPOT assay, and numbers of lymphoid cells positive for PCV2 antigen per unit area [0.25 mm2] determined by immunohistochemistry) were analyzed using a one-way analysis of variance (ANOVA). If the ANOVA showed a significant effect, Tukey’s test for multiple comparisons was performed at each time point. Fisher’s exact test was used for discrete data (lymphoid lesion score). A value of P < .05 was considered to be significant.
Results
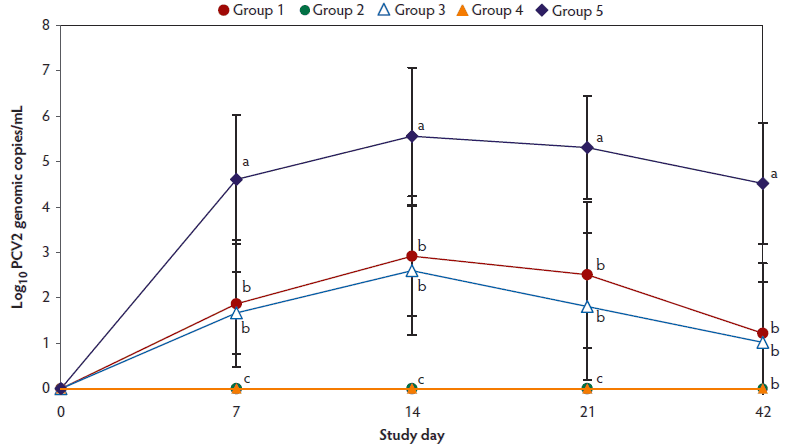
No PCV2 DNA was detected in the serum samples of pigs tested at days -42, -28, and 0. On days 7 to 42, the numbers of genomic copies of PCV2 in serum were significantly lower (P < .05) in Group 1 and Group 3 (vaccinated, challenged pigs) than in Group 5 (unvaccinated, challenged pigs) (Figure 1). However, numbers of genomic copies of PCV2 in serum did not differ between one-dose (Group 1) and two-dose (Group 3) vaccinated, challenged pigs. No PCV2 DNA was detected in serum of pigs in groups 2, 4, and 6 throughout the experiment.
Figure 1: Means (with standard deviation) of the log10 transformed number of genomic copies of PCV2 DNA in serum of pigs in the study described in Table 1. Different letters indicate significant differences among groups (P < .05; one-way ANOVA).
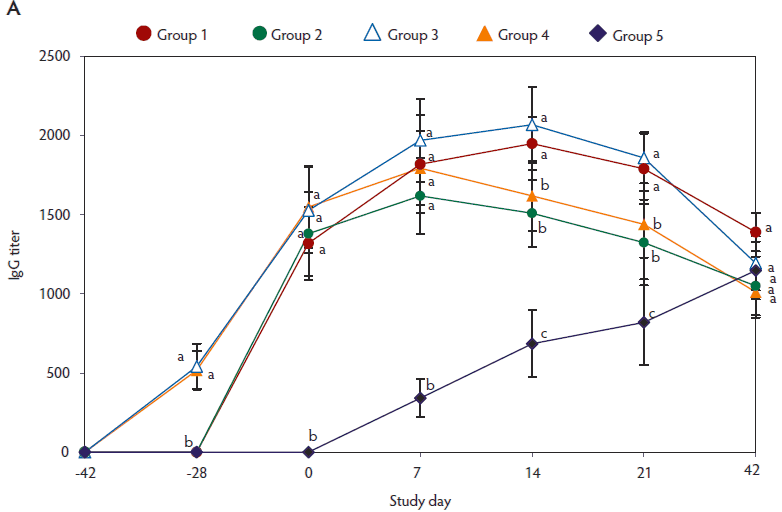
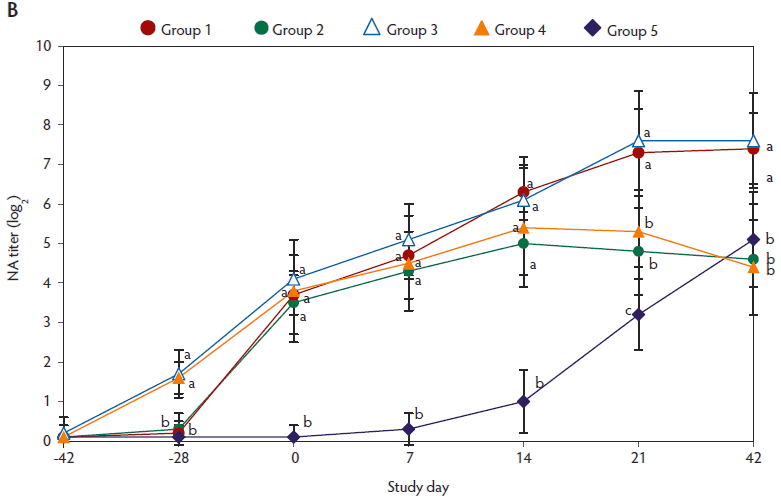
On day -28, anti-PCV2 IgG antibody titers (Figure 2A) and genomic mean NA titers (Figure 2B) were significantly higher (P < .05) in pigs vaccinated with two doses of the vaccine (Group 3 and Group 4), than in pigs vaccinated with a single dose of PCV2 vaccine (Group 1 and Group 2). From day 0 to 21, anti-PCV2 IgG antibody titers (Figure 2A) and genomic mean NA titers (Figure 2B) were significantly higher (P < .05) in vaccinated pigs (groups 1, 2, 3, and 4), than in unvaccinated challenged pigs (Group 5). On days 0 and 7, numbers of PCV2-specific IFN-γ-SCs were significantly higher (P < .05) in vaccinated pigs (groups 1, 2, 3, and 4) than in unvaccinated challenged pigs (Group 5) (Figure 2C).
Figure 2: Means (with standard deviation) for anti-PCV2-IgG antibody titers (panel A); log2 transformed group means (with standard deviation) for neutralizing antibody (NA) titers (panel B); and mean (with standard deviation) of PCV2-specific interferon-γ-secreting cells (IFN-γ-SCs) in peripheral blood mononuclear cells (PBMCs) (panel C) in the study described in Table 1. Different superscript letters indicate significant differences among groups (P < .05; one-way ANOVA).
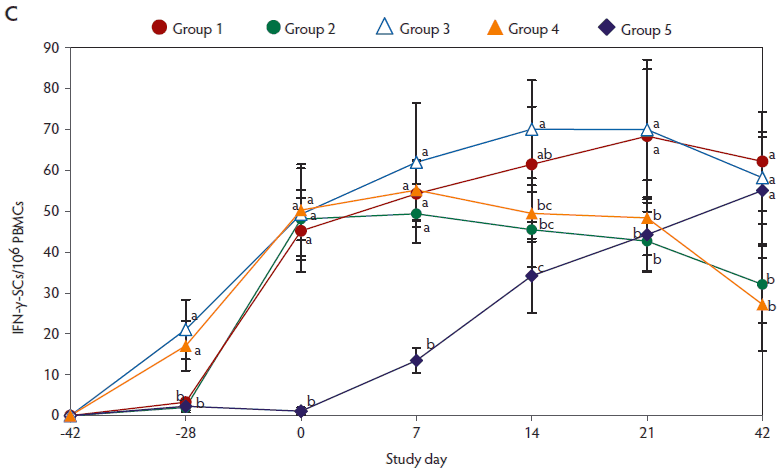
No anti-PCV2 IgG antibodies or PCV2-specific NA or IFN-γ-SCs were detected in Group 6 (negative control).
The number of lymphoid cells positive for PCV2 antigen was significantly lower (P < .05) in the vaccinated groups (Group 1 and Group 3) than in the positive control group (Group 5) (Table 1). However, lymphoid lesion scores and the number of lymphoid cells positive for PCV2 did not differ between challenged pigs administered one dose (Group 1) or two doses (Group 3) of PCV2 vaccine.
Discussion
Commercial PCV2 vaccines approved for single-dose administration have become more popular because a one-dose PCV2 vaccine requires less labor and reduces stress to animals. A major disadvantage of using a single dose is that it does not generate an immunological booster response.9 Therefore, in Korea, some swine producers prefer to vaccinate pigs twice, at 1 and 3 weeks of age, with the one-dose vaccine, because PCVAD usually occurs in pigs between 6 and 12 weeks of age under Korean field conditions.2 In this case, there are two concerns with administering an additional dose of a one-dose vaccine to very young pigs (1 week of age): the potential for interference with maternally derived antibodies and the immature immune system. Experimental evidence indicates that efficacy of PCV2 vaccines is not affected by maternally derived antibodies.10,11 Nonetheless, interference with the efficacy of the PCV2 vaccine depends on the concentration of maternally derived antibodies at the time of vaccination. High immunoperoxidase monolayer assay titers (> 10 log2) interfere with development of the humoral immune response after vaccination.12 Also, as part of the attributes of the development process of the immune system, 1-week-old pigs fail to mount a strong primary immune response when the pig is boostered at 3 weeks of age.13 This suggests that it is more effective to vaccinate pigs older than 1 week of age.
Although differences in immunological parameters at challenge were apparent between pigs vaccinated with either one or two doses of the one-dose vaccine in this study, there were no significant differences in PCV2 viremia or PCV2-associated lesions after challenge. Two additional measures, viral load and viral lesions, are critical parameters to evaluate the efficacy of PCV2 vaccines.13 High levels of PCV2 viremia are associated with development of PCV2-associated lesions.14,15 These observations demonstrate that an additional vaccination in week 1 of life yields no additional protection over a single dose vaccination in week 3. Therefore, two doses of a one-dose PCV2 vaccine administered at 1 and 3 weeks of age, which constitutes extra-label use of the vaccine, provides no additional protection compared to the labelled dose, a single vaccination administered at 3 weeks of age.
Implication
Extra-label use of Circoflex by administering two doses at 1 and 3 weeks of age instead of a single dose at 3 weeks of age is not necessary for control of PCVAD.
Acknowledgements
The author’s research was supported by the Bio-industry Technology Development Program of the Ministry of Agriculture, Food and Rural Affairs, Republic of Korea. This research was also supported by contract research funds of the Research Institute for Veterinary Science (RIVS) from the College of Veterinary Medicine and by the BK 21 PLUS Program for Creative Veterinary Science Research in the Republic of Korea. The first two authors contributed equally to this work.
Disclaimer
Scientific manuscripts published in the Journal of Swine Health and Production are peer reviewed. However, information on medications, feed, and management techniques may be specific to the research or commercial situation presented in the manuscript. It is the responsibility of the reader to use information responsibly and in accordance with the rules and regulations governing research or the practice of veterinary medicine in their country or region.
References
1. Chae C. A review of porcine circovirus 2-associated syndromes and diseases. Vet J. 2005;169:326–336.
2. Chae C. Porcine circovirus type 2 and its associated diseases in Korea. Virus Res. 2012;164:107–113.
3. Chow SC, Wang H, Shao J. Comparing means. In: Chow SC, Wang H, Shao J, eds. Sample size calculations in clinical research. 2nd ed. New York: Chapman & Hall/CRC; 2008:60–92.
4. Gagnon CA, del Castillo JR, Music N, Fontaine G, Harel J, Tremblay D. Development and use of a multiplex real-time quantitative polymerase chain reaction assay for detection and differentiation of Porcine circovirus-2 genotypes 2a and 2b in an epidemiological survey. J Vet Diagn Invest. 2008;20:545–558.
5. Pogranichniy RM, Yoon KJ, Harms PA, Swenson SL, Zimmerman JJ, Sorden SD. Characterization of immune response of young pigs to porcine circovirus type 2 infection. Viral Immunol. 2000;13:143–153.
6. Seo HW, Han K, Oh Y, Park C, Chae C. Efficacy of a reformulated inactivated chimeric PCV1-2 vaccine based on clinical, virological, pathological and immunological examination under field conditions. Vaccine. 2012;30:6671–6677.
7. Fenaux M, Halbur PG, Haqshenas G, Royer P, Thomas P, Nawagitgul P, Gill M, Toth TE, Meng XJ. Cloned genomic DNA of type 2 porcine circovirus is infectious when injected directly into the liver and lymph nodes of pigs: Characterization of clinical disease, virus distribution, and pathologic lesions. J Virol. 2002;76:541–551.
8. Kim D, Ha Y, Lee YH, Chae S, Lee K, Han K, Kim J, Lee JH, Kim SH, Hwang KK, Chae C. Comparative study of in situ hybridization and immunohistochemistry for the detection of porcine circovirus 2 in formalin-fixed, paraffin-embedded tissues. J Vet Med Sci. 2009;71:1001–1004.
9. Opriessnig T, Patterson AR, Madson DM, Pal N, Halbur PG. Comparison of efficacy of commercial one dose and two dose PCV2 vaccines using a mixed PRRSV-PCV2-SIV clinical infection model 2–3-months post vaccination. Vaccine. 2009;27:1002–1007.
10. Opriessnig T, Patterson AR, Elsener J, Meng XJ, Halbur PG. Influence of maternal antibodies on efficacy of porcine circovirus type 2 (PCV2) vaccination to protect pigs from experimental infection with PCV2. Clin Vaccine Immunol. 2008;15:397–401.
11. Opriessnig T, Patterson AR, Madson DM, Pal N, Ramamoorthy S, Meng XJ, Halbur PG. Comparison of the effectiveness of passive (dam) versus active (piglet) immunization against porcine circovirus type 2 (PCV2) and impact of passively derived PCV2 vaccine-induced immunity on vaccination. Vet Microbiol. 2010;142:177–183.
12. Fort M, Sibila M, Perez-Martin E, Nofrarias M, Mateu E, Segalés J. One dose of a porcine circovirus 2 (PCV2) sub-unit vaccine administered to 3-week-old conventional piglets elicits cell-mediated immunity and significantly reduces PCV2 viremia in an experimental model. Vaccine. 2009;27:4031–4037.
13. Chae C. Commercial porcine circovirus type 2 vaccines: Efficacy and clinical application. Vet J. 2012;194:151–157.
14. Meerts P, van Gucht S, Cox E, Vandebosch A, Nauwynck HJ. Correlation between type of adaptive immune response against porcine circovirus type 2 and level of virus replication. Viral Immunol. 2005;18:333–341.
15. Meerts P, Misinzo G, Lefebvre D, Nielsen J, BØtner A, Kristensen CS, Nauwynck HJ. Correlation between the presence of neutralizing antibodies against porcine circovirus 2 (PCV2) and protection against replication of the virus and development of PCV2-associated disease. BMC Vet Res. 2006;2:6–16.
