| Original research | Peer reviewed |
Cite as: Estienne M, Clark-Deener S, Williams K. Growth performance and hematology characteristics in pigs treated with iron at birth and weaning and fed a nursery diet supplemented with a pharmacological level of zinc oxide. J Swine Health Prod. 2019;27(2):64-75.
Also available as a PDF.
SummaryObjective: To determine effects of an iron dose at weaning on growth and hematology in pigs fed zinc. Materials and methods: Weaned pigs (n = 144) were allocated to treatments in a 2 × 2 × 2 factorial arrangement (6 pens/treatment, 3 pigs/pen), with factors being pig size (large or small); number of 100 mg iron dextran doses (1 [birth] or 2 [birth and weaning]); and dietary zinc (100 or 2000 ppm). Average daily gain (ADG), feed intake (ADFI), and gain to feed ratio (G:F) were determined. Blood samples were collected at weaning and 7 and 49 days post-weaning. Results: Anemia (hemoglobin < 9.0 g/dL) at weaning tended to be greater (P = .07) for large pigs and hemoglobin (P = .02) and hematocrit (P = .05) were greater in small pigs. Hematology was largely unaffected by number of iron doses or diet. Large pigs displayed greater ADG (P < .001) but poorer G:F (P = .002). Zinc-supplemented pigs had greater (P = .002) ADG and G:F from day 0 to 21. From day 22 to 49, G:F (P = .005) was greater for controls. Overall, zinc tended to increase ADFI in large (P = .09) but not small (P = .46) pigs. Growth was largely unaffected by number of iron doses. Implications: Anemia at weaning was common, especially for larger pigs, but was not exacerbated by zinc. An iron dose at weaning had minimal effects on growth. Dietary zinc enhanced growth early post-weaning but effects waned as pigs aged. | ResumenObjetivo: Determinar los efectos de una dosis de hierro al destete en el crecimiento y hematología en cerdos alimentados con zinc. Materiales y métodos: Cerdos al destete (n = 144) fueron distribuidos en tratamientos en un arreglo factorial de 2 × 2 × 2 (6 corrales/tratamiento, 3 cerdos/corral), siendo los factores tamaño del cerdo (grande o chico); número de dosis de 100 mg de hierro dextran (1 [nacimiento] o 2 [nacimiento y destete]); y zinc dietético (100 o 2000 ppm). Se determinaron la ganancia diaria promedio (ADG por sus siglas en inglés), consumo de alimento (ADFI por sus siglas en inglés), y relación ganancia alimento (G:F por sus siglas en inglés). Se tomaron muestras de sangre al destete y a los 7 y 49 días post destete. Resultados: La anemia (hemoglobina < 9.0 g/dL) al destete tendió a ser mayor (P = .07) en los cerdos grandes y la hemoglobina (P = .02) y los hematocritos (P = .05) fueron mayores en cerdos pequeños. La hematología en su mayor parte no se vio afectada por el número de dosis de hierro o dieta. Los cerdos grandes tuvieron una mayor ADG (P < .001) pero una menor G:F (P = .002). Los cerdos suplementados con zinc tuvieron mayor (P = .002) ADG y G:F del día 0 al día 21. Del día 22 al 49, la G:F (P = .005) fue mayor en los controles. En general, el zinc tendió a incrementar el ADFI en cerdos grandes (P = .09) pero no en pequeños (P = .46). El crecimiento en general no se vio afectado por el número de dosis de hierro Implicaciones: La anemia al destete fue común, especialmente en los cerdos grandes, pero no se agravó por el zinc. Una dosis de hierro al destete tuvo efectos mínimos en el crecimiento. El zinc dietético mejoró el crecimiento al inicio del destete pero los efectos decrecieron al avanzar la edad de los cerdos. | ResuméObjectif: Déterminer les effets d’une dose de fer au sevrage sur la croissance et des données hématologiques chez des porcs nourris avec du zinc. Matériels et méthodes: Des porcs sevrés (n = 144) ont été répartis à des traitements dans un arrangement factoriel (6 enclos/traitement, 3 porcs/enclos), avec les facteurs étant la taille des porcs (gros ou petit); le nombre de doses de 100 mg de fer dextran (1 [naissance] ou 2 [naissance et sevrage]); et quantité de zinc alimentaire (100 ou 2000 ppm). Le gain moyen quotidien (ADG), la quantité de nourriture consommée (ADFI) et le ratio gain/aliment consommé (G:F) ont été déterminés. Des échantillons sanguins étaient obtenus au sevrage et 7 et 49 jours post-sevrage. Résultats: De l’anémie (hémoglobine < 9.0 g/dL) au moment du sevrage avait tendance à être plus importante (P = .07) pour les gros porcs et l’hémoglobine (P = .02) et l’hématocrite (P = .05) étaient plus élevés chez les petits porcs. Les valeurs hématologiques étaient dans l’ensemble peu affectées par le nombre de doses de fer ou la diète. Les gros porcs montraient un ADG plus important (P < .001) mais un plus faible G:F (P = .002). Les porcs supplémentés en zinc avaient un ADG et un G:F (P = .002) plus élevés du jour 0 au jour 21. Du jour 22 au jour 49, le G:F était plus élevé pour les animaux témoins (P = .005). De manière globale, le zinc avait tendance à augmenter l’ADFI chez les gros (P = .09) mais pas les petits (P = .46) porcs. La croissance était largement non-affectée par le nombre de doses de fer. Implications: L’anémie au moment du sevrage est fréquente, spécialement chez les porcs plus gros, mais n’était pas exacerbée par le zinc. Une dose de fer au moment du sevrage avait des effets minimaux sur la croissance. Le zinc alimentaire augmentait la croissance tôt après le sevrage mais les effets diminuèrent à mesure que les porcs vieillissaient. |
Keywords: swine, nursery, iron, zinc, hematology
Search the AASV web site
for pages with similar keywords.
Received: July 5, 2018
Accepted: September 26, 2018
Iron deficiency anemia can quickly develop in suckling pigs because the newborn animal has limited capacity for storing iron, sow colostrum and milk have relatively insignificant iron concentrations, and modern genotypes have the capacity for rapid growth rates. To prevent anemia, it is standard industry practice to provide supplemental iron to newborn pigs, usually by administering an intramuscular (IM) injection of 100 to 200 mg iron dextran.1 Recent research, however, has demonstrated that many pigs, particularly the largest, fastest-growing animals in a litter, are iron deficient or anemic at weaning despite having received supplemental iron during the first week of life.2,3 After weaning, anemic pigs grow slower than non-anemic counterparts during the nursery phase of production.3,4 An additional iron dose at weaning could be important, particularly for nursery pigs that consume diets supplemented with pharmacological levels of zinc to enhance growth performance.5-8 Liver iron concentrations were decreased and anemia developed in pigs consuming diets with high levels of zinc.9 On commercial farms, elevated concentrations of zinc oxide (approximately 2000 ppm) in nursery diets were associated with a greater risk of anemia in pigs.3 Thus, the objective of this experiment was to determine the effects of an additional 100 mg iron supplement at weaning on growth performance and hematology characteristics in nursery pigs fed a diet supplemented with 2000 ppm of zinc.
Materials and methods
The protocol for this experiment was reviewed and approved by the Virginia Tech Institutional Animal Care and Use Committee.
Study animals and housing
A total of 144 Duroc-sired pigs (74 male and 70 female) farrowed by 18 Yorkshire × Landrace sows were enrolled in this study. Within 24 hours after birth, pigs were ear notched for identification, weighed, and needle teeth were clipped and tails docked. All pigs received an IM injection of 100 mg iron hydrogenated dextran (Iron-100; Durvet, Inc, Blue Springs, Missouri) in the neck muscle behind the ear using a 20 gauge, 1.27 cm-long needle. At 7 days of age, boar pigs were bilaterally castrated using a sterile scalpel. Pigs were not provided creep feed during the suckling period.
Pigs were moved at weaning to an environmentally controlled nursery unit. Pen floors were galvanized steel bar slats. Nursery pens measured 0.91 × 1.22 m2 and each contained a nipple drinker and stainless steel feeder with four feeding spaces.
Study design
Pigs were weaned at approximately 21 days of age, weighed, and divided into equal groups of the largest and smallest pigs (average body weight [SE]; 7.91 [0.06] and 5.38 [0.06] kg, respectively). Pigs were housed in groups of 3 pigs/pen in six blocks of eight pens each, with litter of origin and sex balanced across pens. Pens were allocated to a 2 × 2 × 2 factorial arrangement of treatments with 6 replicate pens per treatment combination (total of 48 pens). The factors were: 1) pig size (large or small); 2) number of 100 mg iron dextran IM doses (1 dose administered within 24 hours after birth or 2 doses [one administered within 24 hours after birth and the other at weaning]); and, 3) level of dietary zinc (100 or 2000 ppm).
The amount of iron administered to pigs soon after birth varies in the swine industry with the most common doses ranging from 100 to 200 mg. For this experiment, 100 mg was used because lower doses are less likely to be toxic and to induce oxidative stress. Greater doses of parenteral iron have also been demonstrated to increase liver secretion of hepcidin which perturbs systemic iron metabolic processes.10,11 Finally, the 100 mg dose given soon after birth would likely increase the number of pigs that were anemic at weaning, allowing for evaluation of how these individuals responded to dietary zinc supplementation.
Experimental diets
Pigs were allowed ad libitum access to a 3-phase feeding regimen with all diets meeting the requirements for the various nutrients12 and zinc was adjusted as to the concentrations previously indicated. For each phase, a basal diet was first prepared containing most of the corn and all the common ingredients for each experimental diet. Zinc oxide (Maximo 720; Zinc Nacion, Monterrey, Nuevo León, Mexico) or an equal amount of ground corn was then added to the basal diet to create the zinc oxide and control diets, respectively (Table 1).
Table 1: Composition of zinc oxide and control diets fed to nursery pigs for 49 days*
| Dietary phase: | I | II | III |
|---|---|---|---|
| Days fed: | 0 - 7 | 8 – 21 | 22 - 49 |
| Feed component, % | |||
| Ground corn | 41.95 | 54.76 | 64.76 |
| Soybean oil | 3.00 | 3.00 | 3.00 |
| Dried whey | 25.00 | 10.00 | 0.00 |
| Menhaden fish meal | 4.00 | 2.00 | 0.00 |
| Soycomil† | 3.00 | 2.00 | 2.00 |
| Soybean meal | 19.85 | 24.90 | 26.65 |
| Dicalcium phosphate | 1.00 | 1.00 | 1.25 |
| Calcium carbonate | 0.70 | 1.00 | 1.00 |
| Salt | 0.20 | 0.20 | 0.20 |
| Lysine-HCL | 0.40 | 0.30 | 0.30 |
| DL-methionine | 0.12 | 0.06 | 0.06 |
| Vitamin-trace mineral‡ | 0.50 | 0.50 | 0.50 |
| Zinc Oxide or ground corn | 0.28 | 0.28 | 0.28 |
| Totals | 100.00 | 100.00 | 100.00 |
| Calculated analysis, % | |||
| Crude protein | 20.55 | 20.31 | 19.55 |
| Lysine | 1.53 | 1.37 | 1.27 |
| Methionine | 0.46 | 0.39 | 0.37 |
| Calcium | 0.88 | 0.83 | 0.74 |
| Phosphorous | 0.75 | 0.65 | 0.61 |
* Zinc oxide or control diets were prepared by mixing zinc oxide (Maximo 720; Zinc Nacion, Monterrey, Nuevo León, Mexico) or ground corn, respectively, with basal diet consisting of the major portion of the ground corn and all other common ingredients. Control diets contained 14.2 ppm of copper, 113 ppm of iron, and 113 ppm of zinc.
† Archer Daniels Midland Co (Decatur, Illinois).
‡ ANS Swine Breeder Premix manufactured for Agri-Nutrition Services, Inc (Shakopee, Minnesota). Trace minerals in sulfate forms were in a polysaccharide complex.
Data and sample collection and blood assay
Pigs were weighed at weaning and then weekly for 49 days. Average daily gain (ADG) was determined for the periods of day 0 to 7, day 8 to 21, day 22 to 49, and the entire trial. Feed additions were recorded so that average daily feed intake (ADFI) and the gain to feed ratio (G:F) could be calculated for each period and the entire trial.
Before the second injection of iron was administered to the appropriate pigs, a blood sample was collected from the barrow weighing closest to the average weight of pigs in each pen at weaning and at 7 and 49 days post-weaning. The same pig was used for each collection. On each occasion, barrows were placed supine on a v-board and approximately 7 mL of blood was collected via jugular venipuncture (20 gauge, 2.54 cm-long needle) into a Vacutainer tube (Becton, Dickinson and Company, Franklin Lakes, New Jersey) containing EDTA. Blood was used for complete blood counts conducted with a Coulter Multisizer 3 cell counter (Beckman Coulter, Inc, Brea, California) by Animal Laboratory Services of the Virginia-Maryland College of Veterinary Medicine (Blacksburg, Virginia). The following hematological determinations were made: number of red blood cells, reticulocytes, white blood cells, neutrophils, lymphocytes, monocytes, eosinophils, basophils, and platelets, percentage of reticulocytes, hemoglobin concentration, hematocrit, mean corpuscular volume, mean corpuscular hemoglobin concentration, red blood cell distribution width, and mean platelet volume.
Statistical analysis
Data were subjected to ANOVA using the PROC MIXED procedure of SAS (SAS Institute Inc, Cary, North Carolina). Body weights, ADG, ADFI, and G:F were analyzed using a model that included pig size, number of iron doses, diet, and all two- and three-way interactions as possible sources of variation. Block was included as a random variable and pen served as the experimental unit. A repeated measures model was used for analyzing hematological characteristics and included pig size, number of iron doses, diet, day, and all two-, three-, and four-way interactions as possible sources of variation. Block was included as a random variable and individual pig was the experimental unit. Individual means were compared using the LSMEANS option of PROC MIXED and were adjusted using the Tukey-Kramer procedure. Differences in means were considered statistically significant at P < .05 and tendencies declared at P < .1.
Additionally, the percentage of large and small pigs that were anemic at weaning (hemoglobin concentrations < 9.0 g/dL)2 were compared using chi-square analysis. Hematological measures in anemic and non-anemic pigs at weaning were compared using ANOVA with a model that included anemic status (yes or no) as the main source of variation. Block and weaning weight were used as covariates.
Results
Incidence of anemia in pigs at weaning
The blood sample from one small pig clotted before laboratory analyses were conducted, so data presented here represents a total of 23 small pigs and 24 large pigs. The proportion of animals that were anemic tended to be greater (P = .07) for large (10 of 24; 41.7%) compared to small (4 of 23; 17.4%) pigs. Hemoglobin concentrations (P = .004), hematocrit (P = .004), the number of red blood cells (P = .01), and mean corpuscular volume (P = .04) were less and the number of reticulocytes tended (P = .06) to be less in anemic pigs as compared to non-anemic pigs (Table 2). In contrast, red blood cell distribution width was greater (P = .003) in the anemic individuals.
Table 2: Least Squares Means (SE) of hematology characteristics in weaned pigs classified as anemic (n = 14) or not anemic (n = 33) based on circulating hemoglobin concentrations*
| Hematological parameter | Reference values† | Anemic | Not anemic | P‡ |
|---|---|---|---|---|
| Hemoglobin, g/dL | 9 - 14 | 7.90 (0.26) | 10.35 (0.18) | .004 |
| Hematocrit, % | 26 - 41 | 27.21 (0.89) | 35.02 (0.64) | .004 |
| Red blood cells, × 106 cells/µL | 5.3 - 8.0 | 5.31 (0.15) | 6.22 (0.10) | .01 |
| Mean corpuscular volume, fL | 42 - 62 | 51.55 (1.66) | 56.42 (0.39) | .04 |
| Mean corpuscular hemoglobin, g/dL | NR | 29.16 (0.25) | 29.54 (0.17) | .28 |
| Red blood cell distribution width, % | NR | 28.24 (0.74) | 20.19 (0.58) | .003 |
| Reticulocytes, % | NR | 4.22 (0.58) | 5.70 (0.41) | .11 |
| Reticulocytes, × 103 cells/µL | NR | 228.33 (35.40) | 352.89 (24.22) | .06 |
| White blood cells, × 103 cells/µL | 8.7 - 37.9 | 8.93 (0.81) | 9.02 (0.56) | .93 |
| Neutrophils, × 103 cells/µL | 2.5 - 23 segmented 0.0 - 3.1 bands | 2.79 (0.58) | 3.71 (0.40) | .26 |
| Lymphocytes, × 103 cells/µL | 2.2 - 16 | 5.06 (0.41) | 4.71 (0.36) | .39 |
| Monocytes, × 103 cells/µL | 0.001 - 5 | 0.30 (0.04) | 0.27 (0.02) | .50 |
| Eosinophils, × 103 cells/µL | 0 - 1.8 | 0.31 (0.07) | 0.25 (0.05) | .47 |
| Basophils, × 103 cells/µL | 0 - 0.5 | 0.10 (0.02) | 0.07 (0.01) | .22 |
| Platelets, × 103 cells/µL | NR | 617.47 (43.98) | 676.59 (33.15) | .30 |
| Mean platelet volume, fL | NR | 8.60 (0.64) | 8.86 (0.57) | .62 |
* Pigs with circulating hemoglobin < 9.0 g/dL were classified as anemic.2 Blood was sampled from large or small (body weight [SE], 7.91 [0.13] and 5.36 [0.19] kg, respectively) barrows at weaning and the proportion of animals that were anemic tended to be greater (P = .07) for large (10 of 24; 41.7%) compared to small (4 of 23; 17.4%) pigs.
† Reference values from Bhattarai et al.2
‡ Hematological measures were compared using ANOVA with a model that included anemic status (yes or no) as the main source of variation. Block and weaning weight were used as covariates.
NR = not reported.
Hematology characteristics
Except for the interaction of pig size and day, there were no interactions of main effects on hematology characteristics. Hemoglobin concentrations (P = .01), hematocrit (P = .05), mean corpuscular volume (P < .001), and red blood cell distribution width (P = .004) (Figure 1) as well as the number of red blood cells (P = .06) and mean corpuscular hemoglobin (P = .009) (Figure 2) were affected or tended to be affected by the interaction of pig size and day. Hemoglobin concentration (P = .02) and hematocrit (P = .05) were less (P < .05) in large versus small pigs on day 0, but not on other days; mean corpuscular volume was less and red blood cell distribution width was greater in large pigs versus small pigs on days 0 (P < .001 and P < .001, respectively) and 7 (P = .008 and P = .002, respectively), but not on day 49 (Figure 1). For large pigs, the number of red blood cells tended to increase (P = .06) from day 0 to day 7 and then remained similar (P = .78) until day 49; mean corpuscular hemoglobin concentration increased (P = .007) from day 0 to day 7 and further increased (P < .001) to day 49. For small pigs, the number of red blood cells tended to increase (P = .06) from day 0 to day 7 and from day 7 to day 49 (P = .06); mean corpuscular hemoglobin concentrations increased from day 0 to day 7 (P < .001) and tended to increase (P = .08) until day 49 (Figure 2).
Figure 1: Hematology characteristics in blood collected from large and small pigs at weaning (day 0) and at days 7 and 49 post-weaning. Data were subjected to ANOVA for repeated measures. The model included size of pig, number of iron treatments, diet, day and all two-, three- and four-way interactions as possible sources of variation. Interaction of pig size and day for A, hemoglobin concentrations (P = .01); B, hematocrit (P = .05); C, mean corpuscular volume (P < .001); and D, red blood cell distribution width (P = .004). For each characteristic, days on which values differed (P < .05) for large and small pigs are indicated by an *.
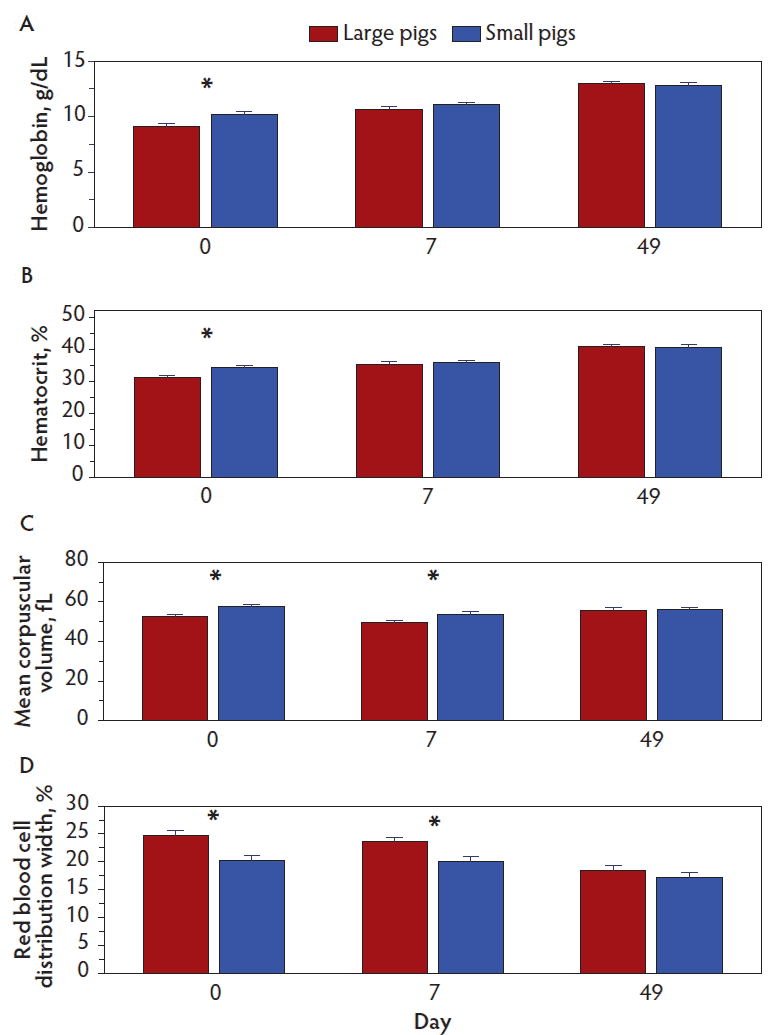
Figure 2: Red blood cell and mean corpuscular hemoglobin concentrations in large and small pigs at weaning (day 0) and days 7 and 49 post-weaning. Data were subjected to ANOVA for repeated measures. The model included size of pig, number of iron treatments, diet, day, and all two-, three-, and four-way interactions as possible sources of variation. Interaction of pig size and day for A, red blood cell concentration (P = .06) and B, mean corpuscular hemoglobin concentration (P = .009). Within pig size for each hematology characteristic, bars with different superscripts differ (Figure A: P = .06 and Figure B: P < .05).
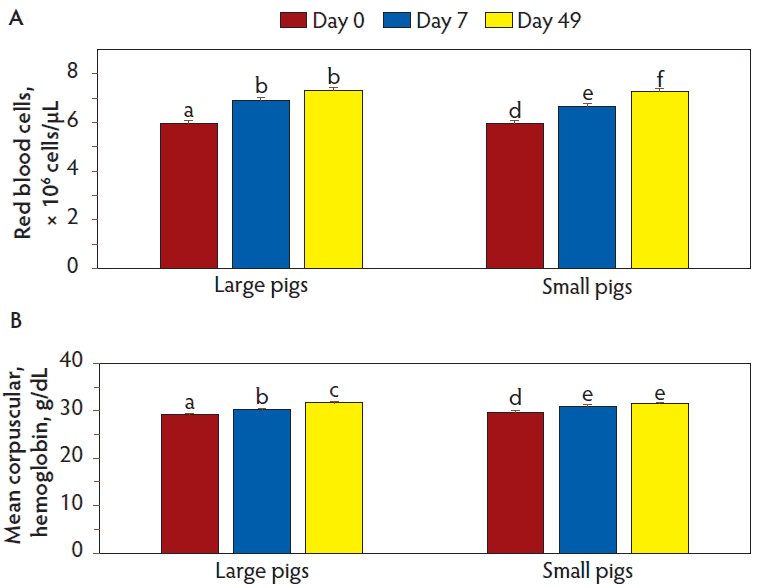
Table 3 contains hematology characteristics in nursery pigs as affected by the main effects of pig size, number of iron doses, diet, and day (day 0, 7, or 49 post-weaning). Mean corpuscular volume was less (P < .001) and hemoglobin (P = .07) and mean corpuscular hemoglobin (P = .09) tended to be less in large compared to small pigs. In contrast, red blood cell distribution width was greater (P < .001) in the large versus small pigs. Size of pig did not affect other hematology characteristics. Except for a tendency (P = .09) for the number of white blood cells to be greater in pigs receiving a dose of iron only at birth compared to at birth and at weaning, hematology characteristics were not affected by the number of iron doses. Mean corpuscular hemoglobin tended to be greater (P = .06) in zinc-supplemented pigs compared to controls, but diet did not affect other hematology characteristics.
Table 3: Hematology characteristics of large and small nursery pigs treated intramuscularly with 100 mg iron dextran at birth or at birth and at weaning and fed control or zinc supplemented (2000 ppm) diets for 49 days
| Size of pig | Iron treatments | Diet | Day† | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hematological parameter | Large (n = 24) | Small (n = 23) | SE | P‡ | Birth (n = 24) | Birth + weaning (n = 23) | SE | P‡ | Control (n = 24) | Zinc (n = 23) | SE | P‡ | 0 (n = 47) | 7 (n = 47) | 49 (n = 47) | SE | P‡ |
| Red blood cells, × 106 cells/µL* | 6.80 | 6.63 | 0.14 | .23 | 6.72 | 6.71 | 0.14 | .93 | 6.72 | 6.71 | 0.14 | .98 | 5.95a | 6.90b | 7.30c | 0.10 | < .001 |
| Hemoglobin, g/dL* | 10.92 | 11.35 | 0.24 | .07 | 11.22 | 11.05 | 0.24 | .46 | 11.03 | 11.24 | 0.24 | .36 | 9.66a | 10.88b | 12.87c | 0.20 | < .001 |
| Hematocrit, %* | 35.85 | 36.90 | 0.70 | .14 | 36.64 | 36.11 | 0.70 | .45 | 36.27 | 36.48 | 0.70 | .77 | 32.74a | 35.60b | 40.78c | 0.70 | < .001 |
| Mean corpuscular volume, fL* | 52.72 | 55.89 | 0.86 | < .001 | 54.70 | 53.90 | 0.86 | .35 | 54.18 | 54.43 | 0.86 | .77 | 55.12a | 51.85b | 55.94a | 0.54 | < .001 |
| Mean corpuscular hemoglobin, g/dL* | 30.33 | 30.72 | 0.23 | .09 | 30.54 | 30.50 | 0.23 | .86 | 30.30 | 30.74 | 0.23 | .06 | 29.44a | 30.58b | 31.55c | 0.16 | < .001 |
| Red blood cell distribution width, %* | 22.29 | 19.21 | 0.84 | < .001 | 20.89 | 20.61 | 0.84 | .74 | 21.27 | 20.23 | 0.84 | .22 | 22.55a | 21.85a | 17.85b | 0.48 | < .001 |
| Reticulocytes, % | 3.75 | 3.93 | 0.30 | .56 | 3.97 | 3.71 | 0.30 | .40 | 4.01 | 3.67 | 0.30 | .27 | 5.36a | 2.74b | 3.43a | 0.33 | < .001 |
| Reticulocytes, × 103 cells/µL | 249.9 | 255.9 | 19.8 | .76 | 261.9 | 243.9 | 19.8 | .36 | 261.9 | 243.9 | 19.8 | .37 | 318.4a | 190.3b | 249.9c | 21.6 | < .001 |
| White blood cells, × 103 cells/µL | 13.27 | 12.61 | 0.59 | .26 | 13.45 | 12.43 | 0.59 | .09 | 13.14 | 12.73 | 0.59 | .49 | 8.97a | 14.35b | 15.50b | 0.64 | < .001 |
| Neutrophils, × 103 cells/µL | 4.73 | 4.78 | 0.37 | .89 | 4.96 | 4.55 | 0.37 | .27 | 4.91 | 4.60 | 0.37 | .40 | 3.46a | 5.86b | 4.94b | 0.51 | < .001 |
| Lymphocytes, × 103 cells/µL | 8.36 | 6.69 | 1.02 | .10 | 8.27 | 6.79 | 1.02 | .15 | 7.04 | 8.01 | 0.92 | .34 | 4.82a | 8.59b | 9.18b | 1.28 | .001 |
| Monocytes, × 103 cells/µL | 0.60 | 0.66 | 0.06 | .24 | 0.65 | 0.61 | 0.06 | .52 | 0.65 | 0.61 | 0.06 | .54 | 0.27a | 0.73b | 0.89c | 0.05 | < .001 |
| Eosinophils, × 103 cells/µL | 0.28 | 0.27 | 0.04 | .70 | 0.29 | 0.26 | 0.04 | .40 | 0.29 | 0.26 | 0.04 | .40 | 0.26 | 0.31 | 0.24 | 0.05 | .31 |
| Basophils, × 103 cells/µL | 0.11 | 0.13 | 0.02 | .29 | 0.13 | 0.11 | 0.02 | .17 | 0.12 | 0.12 | 0.02 | .92 | 0.08a | 0.12b | 0.15b | 0.01 | < .001 |
| Platelets, × 103 cells/µL | 501.6 | 509.4 | 33.9 | .82 | 513.0 | 498.0 | 33.9 | .66 | 506.3 | 504.7 | 33.9 | .96 | 664.6a | 515.3b | 336.6c | 30.1 | < .001 |
| Mean platelet volume, fL | 8.59 | 8.71 | 0.27 | .65 | 8.66 | 8.64 | 0.27 | .93 | 8.73 | 8.58 | 0.27 | .59 | 8.75 | 8.45 | 8.76 | 0.28 | .45 |
* Affected (P < .05) or tended to be affected (P = .06) by interaction of pig size and day.
† For the main effect of day, values with different superscripts (a,b,c) differ (P < .05).
‡ Data were subjected to ANOVA for repeated measures. The model included size of pig, number of iron treatments, diet, day, and all two-, three-, and four-way interactions as possible sources of variation.
Other than the number of eosinophils (P = .31) and mean platelet volume (P = .45), all hematology measures were affected by day. Red blood cell number, hemoglobin concentration, hematocrit, mean corpuscular hemoglobin concentration, and the number of monocytes increased (P < .001) from day 0 to day 7 and further increased (P < .001) from day 7 to day 49. The number of white blood cells (P < .001), neutrophils (P < .001), lymphocytes (P = .009), and basophils (P = .01) increased from day 0 to day 7, and then remained similar until day 49. The number of platelets decreased (P < .001) from day 0 to day 7 and further decreased (P < .001) from day 7 to day 49. Red blood cell distribution width was similar (P = .28) on days 0 and 7 and then decreased (P < .001) from day 7 to day 49. Reticulocyte numbers decreased (P < .001) from day 0 to day 7 and then tended to increase (P = .09) from day 7 to 49, however, numbers on day 49 were less (P < .001) than numbers on day 0. Mean corpuscular volume decreased (P < .001) from day 0 to day 7 but increased (P < .001) to levels on day 49 that were not different (P = .29) from those on day 0. Reticulocyte percentage decreased (P < .001) from day 0 to 7 and tended to increase (P = .09) to day 49, however levels at day 49 were less (P < .001) than levels at day 0.
Growth performance
There were a few two-way interactions of main effects on various growth measures, however, there were no three-way interactions among pig size, number of iron doses, and diet. Between day 22 and 49 post-weaning, ADFI (P = .06), but not ADG (P = .25) or G:F (P = .61), tended to be affected by an interaction between pig size and diet (Figure 3). In large (P = .06) but not small (P = .91) pigs, ADFI was greater when the diet was supplemented with 2000 ppm of zinc. For the period between day 22 and 49, G:F (P = .05), but not ADG (P = .29) or ADFI (P = .80), was affected by an interaction of the number of iron doses and diet (Figure 4). Dietary supplementation with 2000 ppm of zinc decreased (P = .05) G:F only in pigs receiving the extra dose of iron at weaning. For the entire experiment (day 0 to 49), the interaction between pig size and diet tended to affect (P = .09) ADFI but not ADG (P = .30) or G:F (P = .71) (Figure 5). The zinc diet tended to increase ADFI in large (P = .09) but not small (P = .46) pigs.
Figure 3: Growth performance between 22 and 49 days post-weaning in pigs classified as large or small at weaning (21 days of age) and that were fed control diets or diets supplemented with 2000 ppm of zinc. Data were subjected to ANOVA using a model that included pig size, number of iron doses, diet, and all two- and three-way interactions as possible sources of variation. Interaction between pig size at weaning and diet for A, ADG (P = .25), B, ADFI (P = .06), and C, G:F (P = 0.61). An * indicates differences between diets within size of pig (P = .06). ADG = Average daily gain; ADFI = average daily feed intake; G:F = gain to feed ratio.
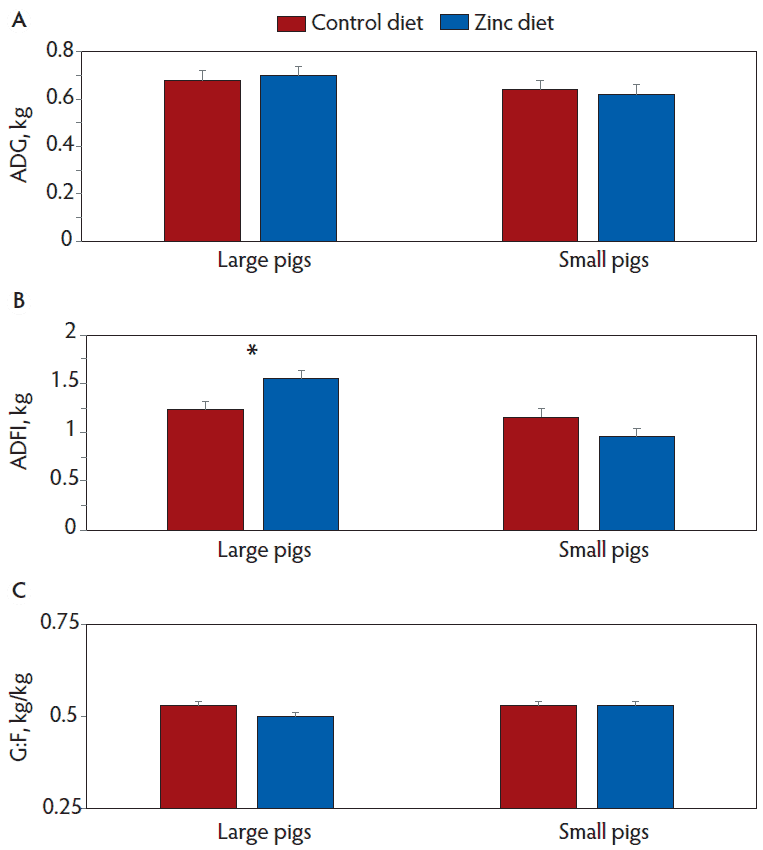
Figure 4: Growth performance between days 22 and 49 post-weaning in pigs that received 100 mg iron dextran at birth or 100 mg iron at birth and at weaning and were fed control diets or diets supplemented with 2000 ppm of zinc. Data were subjected to ANOVA using a model that included pig size, number of iron doses, diet, and all two- and three-way interactions as possible sources of variation. Interaction between the number of iron treatments and diet for A, ADG (P = .29), B, ADFI (P = .80), and C, G:F (P = .05). An * indicates differences between diets within number of iron doses (P = .05). ADG = Average daily gain; ADFI = average daily feed intake; G:F = gain to feed ratio.
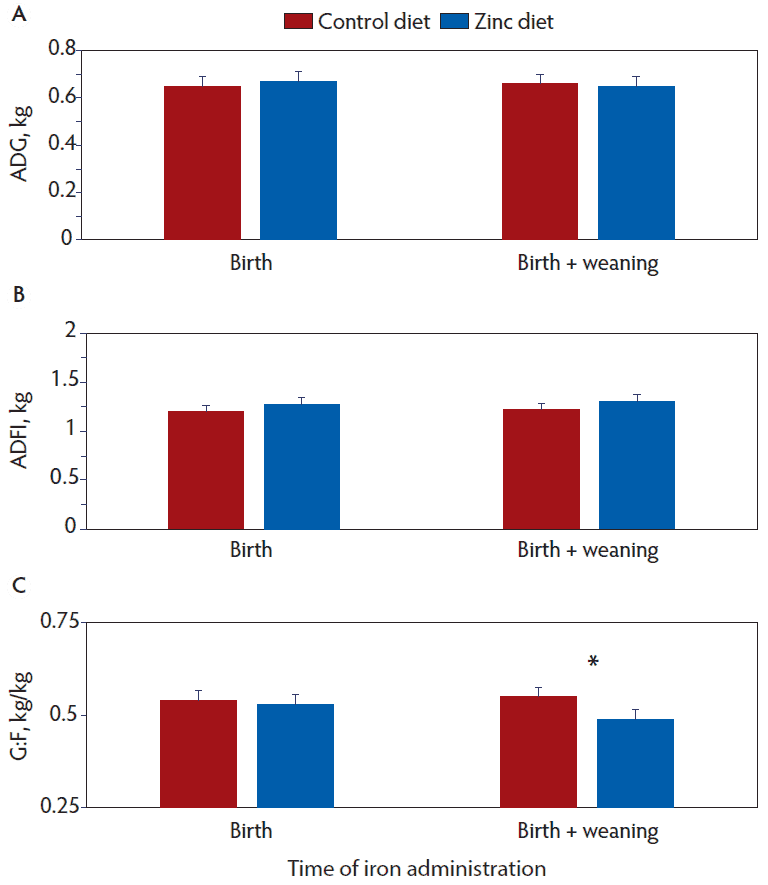
Figure 5: Growth performance between days 0 and 49 post-weaning in pigs classified as large or small at weaning (21 days of age) and that were fed control diets or diets supplemented with 2000 ppm of zinc. Data were subjected to ANOVA using a model that included pig size, number of iron doses, diet, and all two- and three-way interactions as possible sources of variation. Interaction between size of pig and diet for A, ADG (P = .30), B, ADFI (P = .09), and C, G:F (P = .72). An * indicates differences between diets within size of pig (P = .09). ADG = Average daily gain; ADFI = average daily feed intake; G:F = gain to feed ratio.
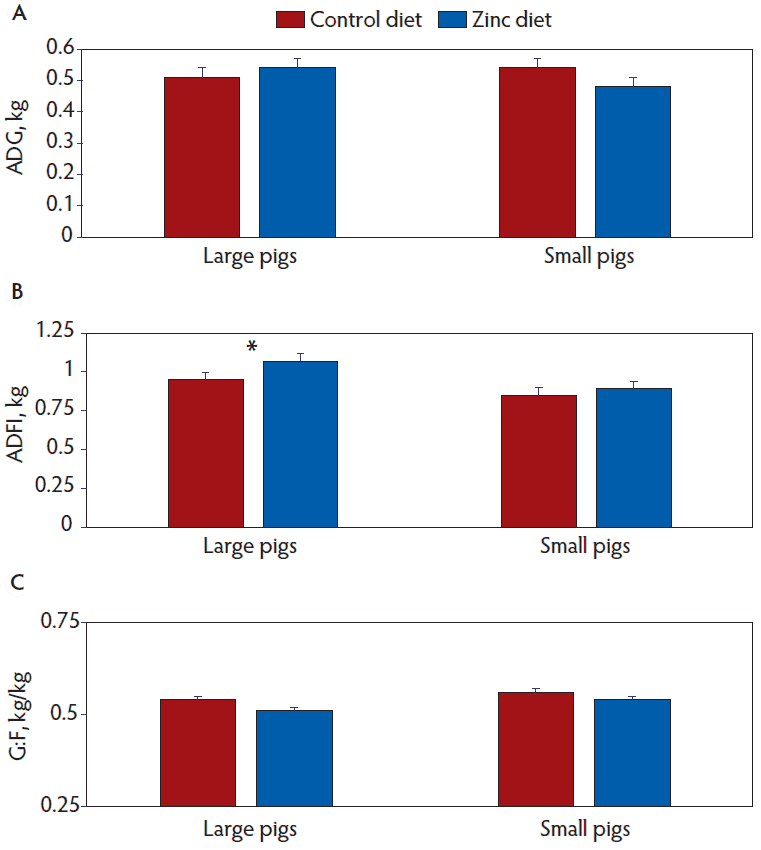
Table 4 summarizes weekly body weights and growth performance in nursery pigs as affected by the main effects of pig size, number of iron doses, and diet. Large pigs weighed more than small pigs on each week of the experiment. From day 0 to 7, pig size did not affect ADG (P = .15) or ADFI (P = .12), but G:F was greater (P = .01) in the small individuals. For days 8 to 21 and 22 to 49, ADG and ADFI were greater (P < .001) in the large versus small pigs. For days 8 to 21, G:F was similar (P = .35) between pig size groups, but from day 22 to 49, G:F was greater (P = .006) in the small individuals. For the overall trial (day 0 to 49 post-weaning), ADG and ADFI were greater (P < .001) in large pigs, and G:F was greater (P = .002) in small animals.
Table 4: Body weight and growth performance of large and small nursery pigs injected once or twice with iron dextran (100 mg) and fed control or zinc supplemented (2000 ppm) diets for 49 days
| Size of pig | Number of iron injections | Diet | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Large | Small | SE | P* | One | Two | SE | P* | Control | Zinc | SE | P* | |
| Number of pens | 24 | 24 | 24 | 24 | 24 | 24 | ||||||
| Body weights, kg | ||||||||||||
| Weaning, Day 0 | 7.91 | 5.38 | 0.06 | < .001 | 6.63 | 6.65 | 0.06 | .73 | 6.64 | 6.64 | 0.06 | .99 |
| Day 7 | 8.54 | 6.14 | 0.10 | < .001 | 7.31 | 7.37 | 0.10 | .57 | 7.19 | 7.49 | 0.10 | .004 |
| Day 14 | 10.67 | 8.03 | 0.14 | < .001 | 9.28 | 9.41 | 0.14 | .37 | 9.08 | 9.62 | 0.14 | < .001 |
| Day 21 | 14.31 | 11.10 | 0.24 | < .001 | 12.55 | 12.85 | 0.24 | .23 | 12.34 | 13.07 | 0.24 | .005 |
| Day 28 | 18.89 | 14.80 | 0.31 | < .001 | 16.67 | 17.01 | 0.31 | .28 | 16.45 | 17.23 | 0.38 | .02 |
| Day 35 | 24.07 | 19.52 | 0.43 | < .001 | 21.70 | 21.89 | 0.43 | .68 | 21.41 | 22.18 | 0.43 | .08 |
| Day 42 | 29.75 | 24.50 | 0.92 | < .001 | 27.06 | 27.19 | 0.92 | .80 | 26.72 | 27.53 | 0.92 | .12 |
| Day 49 | 33.90 | 28.66 | 0.54 | < .001 | 31.36 | 31.20 | 0.54 | .76 | 30.97 | 31.59 | 0.54 | .26 |
| Day 0 to 7 | ||||||||||||
| ADG, kg/day | 0.09 | 0.11 | 0.01 | .15 | 0.10 | 0.10 | 0.01 | .74 | 0.08 | 0.12 | 0.01 | .002 |
| ADFI, kg/day | 0.28 | 0.25 | 0.02 | .12 | 0.27 | 0.26 | 0.02 | .66 | 0.25 | 0.28 | 0.02 | .14 |
| G:F, kg/kg | 0.32 | 0.44 | 0.04 | .01 | 0.36 | 0.40 | 0.04 | .37 | 0.31 | 0.45 | 0.04 | .002 |
| Day 8 to 21 | ||||||||||||
| ADG, kg/day | 0.41 | 0.35 | 0.02 | < .001 | 0.37 | 0.39 | 0.02 | .15 | 0.37 | 0.40 | 0.02 | .02 |
| ADFI, kg/day | 0.70 | 0.59 | 0.03 | < .001 | 0.64 | 0.66 | 0.03 | .29 | 0.60 | 0.70 | 0.03 | < .001 |
| G:F, kg/kg | 0.59 | 0.61 | 0.01 | .35 | 0.59 | 0.60 | 0.01 | .64 | 0.61 | 0.58 | 0.01 | .09 |
| Day 22 to 49 | ||||||||||||
| ADG, kg/day | 0.69 | 0.63 | 0.04 | < .001 | 0.66 | 0.65 | 0.04 | .56 | 0.65 | 0.66 | 0.04 | .86 |
| ADFI, kg/day† | 1.34 | 1.15 | 0.07 | < .001 | 1.24 | 1.26 | 0.07 | .36 | 1.21 | 1.29 | 0.07 | .006 |
| G:F, kg/kg‡ | 0.51 | 0.55 | 0.01 | .006 | 0.54 | 0.52 | 0.01 | .09 | 0.54 | 0.51 | 0.01 | .005 |
| Overall, Day 0 to 49 | ||||||||||||
| ADG, kg/day§ | 0.52 | 0.47 | 0.02 | < .001 | 0.50 | 0.50 | 0.02 | .96 | 0.49 | 0.51 | 0.02 | .18 |
| ADFI, kg/day¶ | 1.01 | 0.86 | 0.04 | < .001 | 0.93 | 0.95 | 0.04 | .31 | 0.90 | 0.97 | 0.04 | < .001 |
| G:F, kg/kg | 0.52 | 0.55 | 0.01 | .002 | 0.54 | 0.53 | 0.01 | .13 | 0.55 | 0.52 | 0.01 | .01 |
* Data were subjected to ANOVA using a model that included pig size, number of iron doses, diet, and all two- and three-way interactions as possible sources of variation.
† Tendency for effect (P = .06) of interaction between size of pig and diet.
‡ Affected (P = .05) by interaction of number of iron treatments and diet.
§ Tendency for effect (P = .10) of interaction between size of pig and number of iron treatments.
¶ Tendency for effect (P = .09) of interaction of size of pig and diet.
ADG = average daily gain; ADFI = average daily feed intake; G:F = gain to feed ratio.
With the exception of a tendency for greater G:F from day 22 to 49 (P = .09) in animals receiving only one dose of iron, there were no effects (P > .13) of number of iron doses on body weights or growth performance measures.
Compared to controls, zinc-supplemented pigs had greater body weights on days 7 (P = .004), 14 (P < .001), 21 (P = .05), and 28 (P = .02), and a tendency (P = .08) for greater body weights on day 35. Body weights at days 42 (P = .12) and 49 (P = .26) were not affected by experimental diet. From day 0 to 7, zinc-fed pigs had greater (P = .002) ADG and G:F, but similar (P = .14) ADFI compared to controls. For the period between days 8 and 21, ADG (P = .02) and ADFI (P < .001) were greater in pigs fed the zinc diet, but G:F tended to be greater (P = .09) for pigs fed control diets. The ADG was similar (P = .86), ADFI was greater (P = .007), and G:F was less (P = .005) for zinc-supplemented pigs compared with controls for day 22 to 49. Over the course of the trial, pigs fed diets supplemented with zinc had greater ADFI (P < .001) but similar ADG (P = .18) and a lower G:F (P = .009) compared with controls.
Discussion
Incidence of anemia in pigs at weaning
Iron is a requisite component of hemoglobin, a protein molecule in red blood cells that carries oxygen from the lungs to bodily tissues and returns carbon dioxide from tissues back to the lungs. If iron levels in the body are inadequate to maintain a normal concentration of hemoglobin in the blood, iron deficiency anemia occurs. In the neonatal pig, iron deficiency anemia can be prevented by parenteral administration of iron,13 and on modern swine farms, an IM injection of 100 to 200 mg iron dextran given within a few days after birth is common.1 The timing, dosage, and number of injections of iron dextran, however, varies widely among commercial operations.1 Moreover, it is evident that despite treatment with iron early in life, a significant number of pigs, particularly the fastest growing animals within a litter, are iron deficient or anemic at weaning.2,3
For the current investigation, pigs with hemoglobin concentrations < 9.0 g/dL were classified as anemic.2 Based on that criterion, a greater proportion of large pigs (41.7%) was classified as anemic at weaning compared with small pigs (17.4%). These results are consistent with previous studies2,3 that also demonstrated an increased risk of anemia at weaning in larger, faster-growing pigs. The overall percentage of pigs in the current study that were classified anemic (29.8%) was greater than the proportion so classified (6%) in a previous study,3 although the criterion used to identify the anemic condition was similar. Perhaps this difference reflects the greater dose of iron dextran (200 mg) administered at birth on the commercial farms in the previous study3 compared to the dose (100 mg) used in the current experiment. However, the proportion of pigs classified as anemic in the current study (29.8%) was similar to the proportion of pigs (35%) categorized as either anemic (hemoglobin concentrations ≤ 9.0 g/dL) or iron deficient (hemoglobin concentrations > 9.0 and ≤ 11.0 g/dL) in a previous report.3
Red blood cell numbers, hematocrit, mean corpuscular volume, and the number of reticulocytes were less for anemic pigs compared with non-anemic counterparts, although mean values in both groups of animals were within reference ranges and considered normal.14 Red blood cell distribution width, a measure of variability in the size of cells, was greater in anemic individuals, which is consistent with iron deficiency anemia.2
Hematology characteristics
Hemoglobin concentrations and hematocrit were less in large pigs than in small pigs at weaning. In addition, mean corpuscular volume was less and red blood cell distribution width was greater at both weaning and 7 days post-weaning in large versus small pigs. These results support previous reports2,3 in which larger and faster-growing pigs are at a greater risk of developing anemia compared with small pigs. Additionally, number of red blood cells increased in large pigs from weaning to day 7 post-weaning and then remained constant through the end of the current trial. In contrast, mean corpuscular hemoglobin concentrations in large pigs increased throughout. The situation was different in small pigs in that number of red blood cells increased from weaning to day 49 post-weaning, but mean corpuscular hemoglobin concentrations increased from day 0 to day 7 and then remained constant. In general, values for other hematological characteristics reported here were affected by day post-weaning with absolute values and trends over time consistent with previous reports.15-17 With the exception of a tendency for mean corpuscular hemoglobin concentrations to be greater in zinc-fed pigs than controls, diet did not affect hematology characteristics. That hemoglobin concentrations and hematocrit were similar between groups suggests that the zinc-supplemented diets did not have an overt effect on iron absorption and utilization. Our results are in general agreement with a previous study18 in which, except for an increased percentage of lymphocytes, hematology profiles of weaned pigs fed pharmacological levels of dietary zinc were similar to control-fed animals.
In the current experiment, pigs treated at both birth and at weaning with 100 mg iron dextran tended to have fewer white blood cells than pigs that received a dose of iron at birth only. The biological significance of this finding is not readily apparent because excess iron in the blood stream of neonatal pigs can cause polyarthritis, septicemia, and colibacillosis, which one would expect to increase the number of white blood cells.19 Also, leukopenia has actually been associated with low hemoglobin concentrations in humans suffering from severe iron deficiency anemia.20
Hemoglobin concentrations were not affected by the additional iron injection at weaning in the experiment reported here. A second injection of 200 mg iron dextran administered 1 or 7 days before weaning at 21 or 28 days of age also failed to consistently increase hemoglobin concentrations in a case study conducted on 5 commercial pig farms.1 In pigs that received 200 mg of iron dextran within 24 hours after birth, hemoglobin concentrations and hematocrit increased only slightly after an additional treatment with iron at weaning (17 days of age), and by 21 days post-weaning, values were actually less compared with pigs that received iron only at birth.21 A second injection of 200 mg iron dextran at 21 days of age increased hemoglobin concentration but not hematocrit in pigs at weaning (28 days of age), but at 21 days post-weaning, neither hematology characteristics were affected by the second treatment.22 For pigs weaned and blood sampled at 34 days of age, a second injection of 200 mg iron dextran administered at 20 days of age also increased hemoglobin concentrations.23 Thus it appears that efficacy of a second iron dose at weaning to increase hemoglobin is impacted by the timing of administration relative to weaning and is probably more effective in pigs weaned at older ages. Moreover, differences in iron statuses between iron-injected and control animals could be obscured by dietary consumption of iron.
Growth performance
In the current study, pigs classified as large at weaning (approximately 7.9 kg) weighed approximately 2.5 kg more than pigs classified as small (approximately 5.4 kg). That large pigs consumed more feed and grew faster during the nursery phase of production is consistent with previous reports.16,24-27 Feed conversion efficiency is a function of body weight, and as a pig grows, it becomes less efficient at converting feed into body weight gain.28 Accordingly, in the current study, small pigs at weaning displayed greater G:F during the nursery phase of production compared to large pigs at weaning.
In general, a second injection of iron dextran did not affect the various growth performance measures. However, between days 22 and 49 post-weaning, a second treatment with iron at weaning tended to decrease G:F in zinc-fed, but not control pigs. A biological explanation for this finding is not readily apparent, but plasma zinc concentrations were increased by increasing dietary concentrations of iron.29 Perhaps by the end of the current 49-day study, blood zinc concentrations in zinc-fed pigs that also received a dose of iron at weaning were reaching a level that was detrimental to feed conversion efficiency.
Studies designed to determine the effects of additional iron administered by either increasing the dosage given at birth or by an additional treatment during the suckling period or at weaning on post-weaning growth have yielded equivocal results. Increased post-weaning ADG in pigs receiving injections of 200 mg iron at birth and 200 mg at 7 to 14 days prior to weaning compared to pigs receiving 200 mg iron only at birth were reported.22,23 In contrast, nursery growth performance was not influenced or was only slightly modified by increasing the dosage of iron given at birth from 200 to 300 mg19,29 or by injecting 200 mg at birth and 100 to 200 mg at day 17 of age or at weaning.21,29 Finally, in the current study, ADFI was increased by dietary zinc and this increase tended to be most pronounced in large size pigs. Increased ADFI in zinc-fed pigs was first detected between day 8 and 21 of the experiment and was subsequently demonstrated for the period from day 22 to 49 and the overall trial. The stimulatory effect of pharmacological concentrations of dietary zinc on feed consumption has been previously reported.7
Increased growth responses in nursery pigs provided pharmacological concentrations of dietary zinc oxide have been well-documented, where experimental diets in most studies had been fed for 28 days.5-8 For example, in a previous study,7 weaned pigs were fed diets containing zinc oxide at levels of 0, 500, 1000, 2000, or 3000 ppm for 28 days. As dietary zinc oxide increased, both early weaned pigs (< 15 days of age) and pigs weaned after 20 days of age had greater ADG and ADFI. Nonetheless, early weaned pigs also displayed greater G:F, which was not observed in the pigs weaned at 20 days of age. Responses for both pig groups plateaued at 2000 ppm.7 For pigs weaned at 21 days of age in the current experiment, enhanced ADG and G:F in response to 2000 ppm zinc was limited to the first 21 days of the experiment. Moreover, feed conversion efficiency was poorer in zinc-fed pigs compared to controls for the period from day 22 to 49. It has been suggested that if pharmacological doses of zinc are fed for too long (> 5 weeks) symptoms of toxicity and deficiencies of other trace minerals can result.12 A perturbed trace mineral balance could explain our finding that supplemental zinc decreased feed conversion efficiency from day 22 to 49 of feeding in pigs receiving a second 100 mg dose of iron at weaning.
Implications
- The incidence of anemia at weaning was significant, especially for the largest individuals, but was not exacerbated by dietary zinc supplementation.
- A second dose of iron dextran administered at weaning had minimal effects on nursery growth performance.
- Pharmacological levels of dietary zinc enhanced nursery growth performance during the early post-weaning period but waned as the pigs aged.
- Supplemental zinc decreased G:F from day 22 to 49 in pigs that received a second dose of iron at weaning, perhaps due to trace mineral imbalances occasioned by the prolonged feeding of zinc.
- Zinc supplementation resulted in increased feed consumption by larger, faster-growing pigs.
Acknowledgement
Funding for this work was provided by the Virginia Agricultural Experiment Station and the Hatch Program of the US Department of Agriculture’s National Institute of Food and Agriculture and the National Pork Board (Project NPB #17-052).
Conflict of interest
None reported.
Disclaimer
Scientific manuscripts published in the Journal of Swine Health and Production are peer reviewed. However, information on medications, feed, and management techniques may be specific to the research or commercial situation presented in the manuscript. It is the responsibility of the reader to use information responsibly and in accordance with the rules and regulations governing research or the practice of veterinary medicine in their country or region.
References
1. Almond G, Byers E, Seate J, Boyer P. Supplemental iron dextran injections: Influence on hemoglobin concentrations and piglet growth. J Swine Health Prod. 2017;25:308-312.
2. Bhattarai S, Nielson JP. Early indicators of iron deficiency in large piglets at weaning. J Swine Health Prod. 2015;23:10-17.
3. Perri AM, Friendship RM, Harding JCS, O’Sullivan TL. An investigation of iron deficiency and anemia in piglets and the effect of iron status at weaning on post-weaning performance. J Swine Health Prod. 2015;24:10-20.
4. Bhattarai S, Nielsen JP. Association between hematological status at weaning and weight gain post-weaning in piglets. Livest Sci. 2015;182:64-68.
5. Smith JW II, Tokach MD, Goodband RD, Nelssen JL, Richert BT. Effects of the interrelationship between zinc oxide and copper sulfate on growth performance of early-weaned pigs. J Anim Sci. 1997;75:1861-1866.
6. Hill GM, Cromwell GL, Crenshaw TD, Dove CR, Ewan RC, Knabe DA, Lewis AJ, Libal GW, Mahan DC, Shurson GC, Southern LL, Veum TL. Growth promotion effects and plasma changes from feeding high dietary concentrations of zinc and copper to weanling pigs (regional study). J Anim Sci. 2000;78:1010-1016.
7. Hill GM, Mahan DC, Carter SD, Cromwell GL, Ewan RC, Harrold RL, Lewis AJ, Miller PS, Shurson GC, Veum TL. Effect of pharmacological concentrations of zinc oxide with or without the inclusion of an antibacterial agent on nursery pig performance. J Anim Sci. 2001;79:934-941.
8. Case CL, Carlson MS. Effect of feeding organic and inorganic sources of additional zinc on growth performance and zinc balance in nursery pigs. J Anim Sci. 2002;80:1917-1924.
9. Cox DH, Hale OM. Liver iron depletion without copper loss in swine fed excess zinc. J Nutr. 1962;77:225-228.
10. Lipinski P, Starzynski RR, Canonne-Hergaux F, Tudek B, Olinski R, Kowalczyk P, Dziaman T, Thibaudeau O, Gralak MA, Smuda E, Wolinski J, Usinska A, Zabielski R. Benefits and risks of iron supplementation in anemic neonatal pigs. Am J Path. 2010;177:1233-1243.
11. Starzynski RR, Laarakkers CMM, Tjalsma H, Swinkels DW, Pieszka M, Stys A, Mickiewicz M, Lipinski P. Iron supplementation in suckling piglets: How to correct iron deficiency anemia without affecting plasma hepcidin levels. PloS One 8:e64022. doi:10.1371/journal.pone.0064022
12. National Research Council. Nutrient Requirements of Swine. 11th ed. Washington, DC: National Academy Press; 2012.
13. Ullrey DE, Miller ER, West DR, Schmidt DA, Seerley RW, Hoefer JA, Luecke RW. Oral and parenteral administration of iron in the prevention and treatment of baby pig anemia. J Anim Sci. 1959;18:256-263.
14. Friendship RM, Lumsden JH, McMillan I, Wilson MR. Hematology and biochemistry reference values for Ontario swine. Can J Comp Med. 1984;48:390-393.
15. Petrovic V, Novotny J, Hisira V, Link R, Leng L, Kovac G. The impact of suckling and post-weaning period on blood chemistry of piglets. Acta Vet Brno. 2009;78:365-371.
16. Callahan SR, Cross AJ, DeDecker AE, Lindemann MD, Estienne MJ. Effects of group-size-floor space allowance during the nursery phase of production on growth, physiology, and hematology in replacement gilts. J Anim Sci. 2017;95:201-211.
17. Ventrella D, Dondi F, Barone F, Serafini F, Elmi A, Giunti M, Romagnoli N, Forni M, Bacci M. The biomedical piglet: Establishing reference intervals for haematology and clinical chemistry parameters of two age groups with and without iron supplementation. BMC Vet Res. 2017; 13:23 doi:10.1186/s12917-017-0946-2
18. Abonyi FO, Ogoenyi EE, Eze JI, Machebe NS. Growth performance, haematology and insulin profile of weanling pigs fed graded levels zinc oxide supplemented diet. Indian J Anim Res. 2015;49:638-644.
19. Murphy KA, Friendship RM, Dewey CE. Effects of weaning age and dosage of supplemented iron on the hemoglobin concentrations and growth rate of piglets. J Swine Health Prod. 1997;5:135-138.
20. Tichelli A, Gratwohl A, Speck B. Iron-deficiency anemia: diagnosis and therapy. Schweiz Med Wochenschr. 1992;122:461-465.
21. Peters JC, Mahan DC. Effects of neonatal iron status, iron injections at birth, and weaning in young pigs from sows fed either organic or inorganic trace minerals. J Anim Sci. 2008;86:2261-2269.
*22. Kamphues J, Manner K, Netzer C. Effects of a 2nd iron injection in suckling piglets on iron retention and performance before and after weaning. Proc 12th IPVC. The Hague, The Netherlands. 1992; 601.
23. Haugegaard J, Wachmann H, Kristensen PJ. Effect of supplementing fast-growing, late-weaned piglets twice with 200 mg iron dextran intramuscularly. Pig J. 2008;61:69-73.
24. McConnell JC, Eargle JC, Waldorf RC. Effects of weaning weight, co-mingling, group size and room temperature on pig performance. J Anim Sci. 1987;65:1201-1206.
25. Mahan DC. Effect of weight, split-weaning, and nursery feeding programs on performance responses of pigs to 105 kilograms body weight and subsequent effects on sow rebreeding interval. J Anim Sci. 1993;71:1991-1995.
26. Wolter BF, Ellis M. The effects of weaning weight and rate of growth immediately after weaning on subsequent pig growth performance and carcass characteristics. Can J Anim Sci. 2001;81:363-369.
27. de Grau A, Dewey C, Friendship R, de Lange K. Observational study of factors associated with nursery pig performance. Can J Vet Res. 2005;69:241-245.
28. Patience JF, Rossoni-Serao MC, Gutierrez NA. A review of feed efficiency in swine: biology and application. J Anim Sci Biotechnol. 2015;6:33. doi:10.1186/s40104-015-0031-2
29. Jolliff JS, Mahan DC. Effect of injected and dietary iron in young pigs on blood hematology and postnatal pig growth performance. J Anim Sci. 2011;89:4068-4080.
* Non-refereed reference.
