ORIGINAL RESEARCH
Studies on the ex vivo survival of Lawsonia intracellularis
Alison Collins, BAgSc; Robert J Love, BVSc, PhD; Javier Pozo, DVM; Sionagh H Smith, BVMS, PhD; Steven McOrist, BVSc, MVSc, PhD, Dip ECVP
AC, RJL: University of Sydney, Camden, NSW, Australia; JP: University of Leon, Leon, Spain; SHS: University of Pennsylvania, Philadelphia, PA 19104; SMcO: VPS, 33 Flemington Street, Glenside, SA 5065, Australia, Email stevemcorist@vps.com.au
Collins A, Love RJ, Pozo J, et al. Studies on the ex vivo survival of Lawsonia intracellularis. Swine Health Prod. 2000;8(5):211-215. Also available in PDF format (104k).
Summary
Objective: To examine the ex vivo survival of Lawsonia intracellularis in conditions relevant to cleaning procedures on pig farms.
Methods: Two co-cultured strains of L. intracellularis were suspended in vitro for 30 minutes at 20 degrees C in various disinfectants according to label recommendations for usage, then washed in saline. Disinfectant-treated and Control bacteria were then added to fresh cells for co-culture for a standard 5-day incubation. Further cell-free suspensions were exposed to air at 5 degrees C, 20 degrees C, or 37 degrees C for up to 8 days, then added to fresh cells for co-culture for a standard 5-day incubation. Lawsonia intracellularis within co-cultures were determined to be viable by visualization after indirect immunoperoxidase staining that incorporated specific monoclonal antibody. Groups of weaned pigs were inoculated orally with L. intracellularis-infected feces, either on the day the feces were collected, or after storage for 1, 2, or 5 weeks at temperatures between 5 degrees C and 15 degrees C.
Results: Mixing of suspensions of L. intracellularis with the quaternary ammonium compound, cetrimide (3.3% wt per vol), for 30 minutes, produced no detectable Lawsonia on reculture; mixing with 1% povidone-iodine for 30 minutes produced no or few (< 1% of controls) detectable Lawsonia on re-culture. Mixing of suspensions with either 1% potassium peroxymonosulfate or a 0.33% phenolic mixture for 30 minutes were less effective at the recommended concentrations tested, as small numbers of L. intracellularis (1%-18% of controls) were detected on reculture of some strains. Detectable numbers of viable L. intracellularis were present after exposure of cell-free suspensions to air for up to 6 days at 5 degrees C. Colonization of the intestine with L. intracellularis was detected in pigs inoculated orally with feces stored for up to 2 weeks at temperatures between 5 degrees C and 15 degrees C.
Implications: Lawsonia intracellularis can probably survive in extracellular conditions for 1 to 2 weeks at 5 degrees -15 degrees C. Pure cultures of L. intracellularis were fully susceptible to a quaternary ammonium disinfectant (3% cetrimide), less so to 1% povidone-iodine, but not to 1% potassium peroxymonosulfate or a 0.33% phenolic mixture.
Keywords  : Lawsonia intracellularis,
disinfection, survival, feces
: Lawsonia intracellularis,
disinfection, survival, feces
Received: February 28, 2000
Accepted: June 28, 2000
Porcine proliferative enteropathy (PPE, ileitis) is a common enteric disease affecting pigs raised under various management systems worldwide. The causative agent, Lawsonia intracellularis, is a gram-negative, obligate intracellular bacterium in the Desulfovibrio family.1,2 Infection of pigs with this bacterium is consistently associated with proliferative mucosal lesions in the ileum and large intestine, which have clinical or subclinical effects on weight gain, feed conversion, and fecal consistency.3 Clinical observations generally include diarrhea and variation in the weights of growing pigs.4
Field and challenge-exposure studies indicate that the infection may persist in pigs for at least 10 weeks, with numerous organisms being passed in the feces.5,6 Therefore, when any pigs in a group or pen are infected, in-contact pigs are likely to become infected. Preliminary investigations of the on-farm epidemiology of PPE suggest that infection is most common among 6- to 16-week-old pigs. These weaner-grower pigs, as well as any L. intracellularis-infected breeding stock, may play an important role in transmission to younger, susceptible animals via infected feces.7-9 Questionnaires and statistical analysis of data from herds with and without pathological evidence of PPE were used to demonstrate a strong association between the occurrence of PPE and the co-mingling of pigs within the previous 30 days.10 Because there may be a relatively long-term excretion of L. intracellularis into pens, information is needed on the possible ex vivo survival time of the organism, and disinfectants that may be effective, to optimize cleaning procedures.
Preliminary pen exposure studies have evaluated infection in sentinel pigs in contact with challenged pigs.11,12 Transmission has occurred to pigs housed in pens in the same room, but not to pigs housed in separate rooms. In one study, transmission occurred despite the use of routine cleaning procedures between pens (washing/disinfection of pens, use of footbaths and dedicated boots/clothing).12 Oral inocula for these and other challenge studies have consisted of cecal contents, or co-cultured pure L. intracellularis where available. In the present study, a variety of in vitro and in vivo methods were used to establish the ability of L. intracellularis to survive in feces in environmental conditions likely to occur on pig farms.
Materials and methods
Standard co-cultures and monitoring of infection
Three strains of Lawsonia intracellularis were used: NCTC 12657 and LRI89/5/83 had been isolated from acute PPE lesions in different 5-month-old British pigs, and the third strain, 51/89, had been isolated from chronic PPE lesions in a 10-week-old pig. The strains were isolated and maintained in co-culture in the rat intestinal epithelial cell line IEC-18 (ATCC CRL 1589) using methods described previously.13 Intestinal material was first inoculated onto cultured cells, and after their initial growth, the bacteria were released from lysed cells, then passaged onto fresh cells. Each isolate used in this study had been passaged between seven and 16 times after initial isolation. During this isolation process, each strain was tested for Chlamydia and Mycoplasma species and other bacterial contaminants, as described previously.13 Quantitative assessment of the numbers of L. intracellularis within the cells on each coverslip co-culture was made by counting the number of infected cells after an indirect immunoperoxidase stain that incorporated a monoclonal antibody (IG4) specific to L. intracellularis.14 Challenge-exposure studies have indicated that intracellular bacteria visualized by this staining method are viable and infective for pigs.1
Evaluation of disinfectant efficacy
Disinfectants selected for testing were:
- potassium peroxymonosulfate (oxidizing agent)
- sodium hypochlorite (chlorine-releasing agent)
- phenolic mixture: orthophenyl phenol and orthobenzyl chlorophenol
- povidone-iodine
- hydrogen peroxide and peracetic acid mixture (acidic oxidizing agent)
- cetrimide: a mixture of akyl trimethyl ammonium bromides (quaternary ammonium agent).
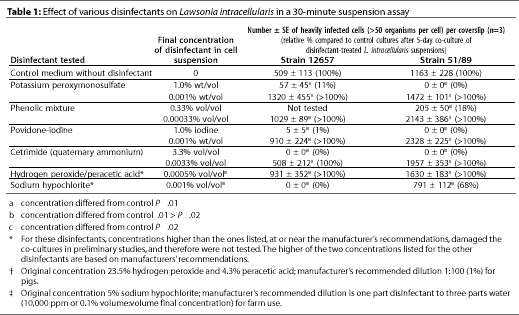
Disinfectant concentrations are shown in Table 1.
Two strains of L. intracellularis (NCTC 12657 and 51/89) were freshly harvested from 5-day-old co-cultures and adjusted in cell culture medium to an approximate concentration of 104 organisms per mL. Separate 1-mL suspensions were added to 1-mL volumes of each diluted disinfectant in small plastic tubes, with no organic load present. In preliminary experiments, the recommended concentrations of hydrogen peroxide/peracetic acid and sodium hypochlorite damaged the co-cultures, and a single, lower concentration of each of these disinfectants was tested (Table 1). A solution of 50:50 vol per vol bacteriological medium in water instead of disinfectant was added to control suspensions. Each of the four remaining disinfectants was diluted in sterile, distilled water to the concentration recommended by the manufacturer, and to 0.001 of that concentration (Table 1).Each tube was incubated at 20 degrees C for 30 minutes without agitation, then centrifuged at 12,000 g for 5 minutes, and the pellet was washed twice in sterile phosphate-buffered saline, pH 7.6. After being resuspended in medium with 7% bovine serum, suspensions were inoculated onto triplicate monolayers which were incubated for the standard 5-day co-culture, after which the coverslips were harvested and the infected cell count established as described above.
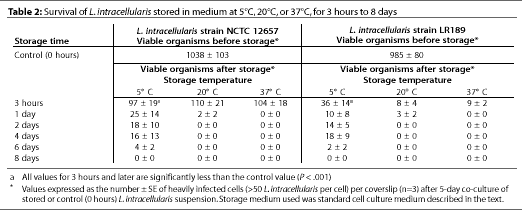
Bacterial survival in bacteriological medium in air
Two strains of L. intracellularis (NCTC 12657 and LR189) were harvested fresh from 5-day co-cultures, freed from cell material by differential centrifugation, and adjusted in medium to suspensions of approximately 104 organisms per mL. The suspensions were stored separately in plastic vessels at 5 degrees C, 20 degrees C, or 37 degrees C. After 3 hours, 1, 2, 4, 6, or 8 days, 1-mL samples of each suspension were added to fresh, 1-day-old IEC-18 cell monolayers and incubated microaerobically at 37 degrees C in triplicate. After the standard 5-day co-culture, the coverslips were harvested and infected cell counts were determined as described above.
Bacterial survival in feces in air
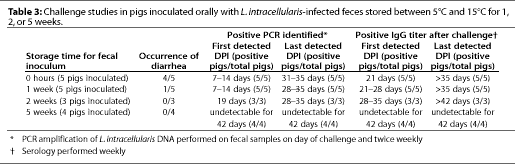
Feces were collected (Day 0) from an adult pig naturally infected with an Australian strain of L. intracellularis. The diagnosis of L. intracellularis infection in the source pig was confirmed by routine pathologic techniques, including histology and indirect immunoperoxidase staining of ileum tissues. Indirect immunoassay of the feces for L. intracellularis content, performed as described previously,6 indicated between 1 x 106 and 107 organisms per g of feces on Day 0. The feces were stored in an open plastic container in a room with a diurnal temperature range of 5 degrees -15 degrees C. Drying of the feces during storage did not allow representative staining and comparisons of fecal smears on Days 7, 14, or 35. Oral inocula were prepared from 10 g of feces resuspended 1:2 vol per vol in sterile phosphate-buffered saline, pH 7.6. Four groups of 3- to 4-week-old pigs were weaned into separate pens in one isolation building, and fed a commercial, wheat-based, pelleted diet, without antibiotics, throughout the trial. There was no direct contact between pigs in different pens, and pigs could not contact the feces of pigs in other pens.
Each of the four groups of pigs was inoculated orally with freshly prepared fecal suspensions so that
- one group (n = 5) received a fecal suspension of fresh feces;
- one group (n = 5) received a fecal suspension of 1-week-old feces;
- one group (n = 3) received a fecal suspension of 2-week-old feces;
- one group (n = 4) received a fecal suspension of 5-week-old feces; and
- a fifth group of unexposed pigs (n = 10) housed in the same isolation building served as controls.
All pigs were monitored for possible signs of clinical infection, including reduced weight gain and diarrhea.
Individual fecal samples were collected from exposed pigs twice weekly and from unexposed controls weekly for PCR amplification of L. intracellularis DNA. Total DNA was extracted from 0.2 g of each fecal sample by use of commercial silica-based columns, according to the manufacturer's instructions ("Wizard," Promega; St. Paul, Minnesota). Five µL of each extract was incorporated as DNA template into a PCR reaction designed to detect L. intracellularis. We used the PCR reaction mixture, buffers, cycles, and PCR product detection methods of Jones, et al., in a total volume of 50 µL as described elsewhere,15 except that primers designated LINTf2c and LINT821r were used. These primers were designed to be complementary to hypervariable regions of the 16S rDNA sequence of L. intracellularis (Genbank accession no. L15739). The sequence of primer LINTf2c was 5'-GGGGGAAGAAATGGATGGGC-3' and primer LINT821r was 5'-TTAACTCCCCAGCACCTAGCACC-3'. The accuracy of these primers was verified by confirmatory DNA sequencing of the PCR product from L. intracellularis and by their inability to amplify from, or hybridize to, DNA extracted from laboratory isolates of Campylobacter mucosalis, C. hyointestinalis, C. coli, C. jejuni, Escherichia coli and Desulfovibrio desulfuricans, incorporated into our standard reactions. Positive control DNA from cultured L. intracellularis (NCTC 12657) was used in each batch of reactions, with a positive PCR product of 402 bp. Blood was collected from all pigs in the study at weekly intervals, and a specific indirect immunofluorescence assay for anti-Lawsonia intracellularis IgG was performed on the serum, as described elsewhere.15
Statistical analysis
Counts for each disinfectant concentration were compared to those of controls by the paired, two-tailed Student T test. Counts for each time point in the air survival study were compared to those of the starting points by the paired, two-tailed Student T test.
Results
Evaluation of disinfectant efficacy
The efficacy of various disinfectants against L. intracellularis, as indicated by subsequent co-culture of the organism, is summarized in Table 1. Mixing of cetrimide at the final concentration of 3.3% (wt per vol) in suspensions of the L. intracellularis strains resulted in no detectable organisms remaining. Mixing of other disinfectants with the suspensions, at the concentrations tested, resulted in small numbers of at least one strain of L. intracellularis being detected in subsequent re-culture, indicating that the disinfectant did not kill all the bacteria in the test suspensions. Mixing of 1% povidone-iodine with L. intracellularis did result in complete or marked reduction of detectable bacteria (< 1% of controls) on subsequent re-culture. The acidic oxidizing agent (hydrogen peroxide/peracetic acid) and the chlorine-releasing agent (sodium hypochlorite) considerably damaged the cell monolayers, despite the washing procedure. We therefore tested reduced concentrations of these agents, which still markedly reduced antibacterial activity: after exposure of strain 12657 to the reduced dose of sodium hypochlorite, no bacteria were detectable on re-culture.
Bacterial survival of bacteriological medium in air
Viable L. intracellularis were detectable for up to 6 days in cell-free suspensions kept in culture medium in plastic vessels in air at 5 degrees C, but for only 1 day when kept at higher temperatures (Table 2).
Bacterial survival in feces in air
At the time of oral challenge, no pigs had detectable L. intracellularis DNA in their feces, or serum IgG antibodies to L. intracellularis (Table 3). Four of five pigs inoculated with fresh feces on Day 0 developed moderate to severe diarrhea beginning 14 days after exposure and persisting for 1 to 4 weeks. One of five pigs challenged with 1-week-old feces developed diarrhea 28 days after exposure, which persisted for 3 weeks. Diarrhea did not occur in other pigs.
PCR products consistent with Lawsonia intracellularis DNA were amplified from control DNA material and from the feces of pigs exposed to infected feces after 0, 1, or 2 weeks' storage, but not from pigs exposed to infected feces stored for 5 weeks (Table 3). Similarly, serum IgG antibodies to L. intracellularis were detected beginning 21 days after exposure in pigs exposed to infected feces after 0, 1, or 2 weeks' storage, but not in control pigs nor in pigs exposed to infected feces stored for 5 weeks (Table 3). L. intracellularis shedding in feces was detected between 5 and 12 days later, and serum IgG antibody development was detected 7 days later, in pigs inoculated with feces stored for 2 weeks compared with pigs given fresh or 1-week-old feces.
Discussion
As L. intracellularis cannot be cultured directly from feces, indirect detection methods were employed. The absence of detectable L. intracellularis DNA in feces or serum antibodies against L. intracellularis at the time of inoculation demonstrated that none of the inoculated pigs had been previously infected with L. intracellularis. The similar temporal pattern of shedding of L. intracellularis in feces and the development of serum IgG antibodies to L. intracellularis indicate that no uncontrolled transmission of infection occurred between our trial pigs.
Our observations of in-vitro data and experimental infections of pigs with stored feces suggest that L. intracellularis may be viable outside the host for up to 2 weeks under conditions similar to those found on pig farms. The absence of clinical signs and delay of seroconversion and fecal shedding in pigs inoculated with feces stored for 2 weeks suggest that the infectivity of the inoculum might have been reduced. However, previous challenge studies have indicated that only a moderate challenge inoculum (106-108 organisms) is required to initiate pathogenic intestinal infection, and persistent fecal shedding can still occur.1,6,16 Our results demonstrating survival and infectivity of L. intracellularis in stored feces suggest that fecal contamination of pens, troughs, and equipment could cause a continuing cycle of infection among new pigs, with subsequent fecal shedding of organisms. This emphasizes the need for thorough cleaning and disinfection of pens and equipment before the introduction of a new batch of pigs. These PCR and serologic studies further corroborate the persistent nature of L. intracellularis infections in pigs exposed to infected feces or other inocula.5,6 In the absence of adequate biosecurity, spread of infection among groups of pigs in a single building or farm may be rapid and far-reaching.
The ability of some disinfectants to reduce the numbers of Lawsonia was partially tested, but further work is needed. Formalin- or glutaraldehyde-based disinfectant compounds were not tested because of their toxicity to humans. Testing methods vary widely, from suspension assays similar to that used in this study, to carrier tests on bacteria placed on a selection of different surfaces. The latter method is not compatible with cell-dependent organisms such as L. intracellularis. This preliminary study indicated that quaternary ammonium compounds and iodine may be effective at the recommended concentrations, but phenol disinfectants may be of limited benefit. Gram-negative organisms have selective membrane porins which are necessary for enteric bacteria to resist toxic effects of fatty acids and bile salts,17 and which may be responsible for bacterial resistance to certain disinfectants, including phenols. The efficacy of quaternary ammonium compounds may be diminished in the presence of organic material; this factor could not be tested fully in our cell culture-dependent system. The possible causes for variations in infected cell counts at low concentrations of some disinfectants (Table 2) could not be determined. Further work is needed on these aspects of disinfectant activity against Lawsonia.
Although effective antibiotics are now known, and on-farm medication programs are available,18 eradication of L. intracellularis infection by medication and livestock control procedures has not been described. Detailed cleaning regimens, including the removal of pigs and feces from pens and the use of disinfectants, should aid disease control in "all-in-all-out" and continuous flow systems. Contact between the disinfectant and the organism is an important aim of disinfection in a pig farm. Full biosecurity may be achieved only where pens are cleaned and left empty for 2 weeks, and incoming pigs are known to be free of infection. On the basis of our results, we recommend washing pens with hot water to remove all organic material, applying quaternary ammonium- or iodine-based disinfectant for at least 30 minutes before re-washing, then leaving the pens empty for at least 2 weeks. It has been suggested that pens with slatted floors may carry a higher risk of transmission of Lawsonia.19 This may be partly due to relaxed cleaning procedures on these floor systems, but some cleaning procedures, such as high-pressure spray washing, may spread pig feces from slatted floors to adjacent pens during washing. We therefore emphasize that our recommended procedure be applied in a thorough and complete manner.
Our results have shown that L. intracellularis has a surprisingly strong capacity to survive outside the host. This may partly explain its common occurrence and apparent ability to persist on pig farms under various management systems. The ex vivo survival of L. intracellularis in air and feces at 5 degrees -15 degrees C is relevant, since these temperatures regularly occur on farms throughout winter periods. Other studies have shown broadly similar survival patterns (10-20 days) in the microaerobic Campylobacter fetus and the anaerobic Brachyspira hyodysenteriae, in cultures exposed to air in the laboratory, and in feces. The survival time of these pathogens in infected feces was also enhanced at cool temperatures (5 degrees -10 degrees C) and by dilution in water.20,21 The microaerobic L. intracellularis retained some viability when stored at 5 degrees -15 degrees C for up to 2 weeks, despite the possibly toxic effects of atmospheric oxygen and feces. Microaerobic or anaerobic bacteria may be protected within larger portions of feces. In studies with dysenteric feces, B. hyodysenteriae could not be isolated from smaller portions of feces air dried at 5 degrees C for 12 hours.20 Eradication programs developed for swine dysentery may be adaptable for proliferative enteropathy.
Acknowledgements
We thank Jens Kjaer and Mike Muirhead for their helpful advice, Matt van Dijk for his technical assistance, and Rebecca Mackie for her helpful scientific assistance.
References--refereed
1. McOrist S, Jasni S, Mackie RA, Neef N, MacIntyre N, Lawson GHK. Reproduction of porcine proliferative enteropathy with pure cultures of ileal symbiont intracellularis. Infect Immun. 1993;61:4284-4292.
2. McOrist S, Gebhart CJ, Boid R, Barns SM. Characterization of Lawsonia intracellularis gen. nov., sp. nov., the obligate intracellular bacterium of porcine proliferative enteropathy. Int J System Bacteriol. 1995;45:820-825.
3. Rowland AC, Lawson GHK. Intestinal adenomatosis in the pig: immunofluorescent and electron microscopic studies. Res Vet Sci. 1974;17:331-323.
4. Winkelman NL. lleitis: An update. Comp Contin Educ. 1996;18:SI9-S27.
5. McOrist S, Lawson GHK. Proliferative enteropathies: Campylobacter species in the faeces of normal and in-contact pigs. Vet Rec. 1989;124:41.
6. Smith SH, McOrist S. Development of persistent intestinal infection and excretion of Lawsonia intracellularis by piglets. Res Vet Sci. 1997;62: 6-10.
7. Rowland AC, Rowntree PGM. A haemorrhagic bowel syndrome associated with intestinal adenomatosis in the pig. Vet Rec. 1972; 91:235-241.
8. Love RJ, Love DN, Edwards MJ. Proliferative haemorrhagic enteropathy in pigs. Vet Rec. 1977;100:65-68.
9. Moller K, Jensen TK, Jorsal SE, Leser TD, Carstensen B. Detection of Lawsonia intracellularis, Serpulina hyodysenteriae, weakly beta-haemolytic intestinal spirochaetes, Salmonella enterica, and haemolytic Escherichia coli from swine herds with and without diarrhoea among growing pigs. Vet Micro. 1998;62:59-72.
13. Lawson GHK, McOrist S, Jasni S, Mackie RA. Intracellular bacteria of porcine proliferative enteropathy: Cultivation and maintenance in vitro. J Clin Microbiol. 1993;31:1136-1142.
14. McOrist S, Boid R, Lawson GHK, McConnell I. Monoclonal antibodies to intracellular campylobacter-like organisms of the porcine proliferative enteropathies. Vet Rec. 1987;121:421-422.
15. Knittel JP, Jordan DM, Schwartz KJ, Janke BH, Roof MB, McOrist S, Harris DL. Evaluation of antemortem polymerase chain reaction and serologic methods for detection of Lawsonia intracellularis-exposed pigs. Am J Vet Res. 1998;59:722-726.
17. Russell AD, Hugo WB. Chemical disinfectants. In: Linton AH, Hugo WB, Russell AD, eds. Disinfection in Veterinary and Farm Animal Practice. Oxford: Blackwell Scientific Publications; 1987:12-42.
18. McOrist S, Smith SH, Klein T. Monitored control programme for proliferative enteropathy on British pig farms. Vet Rec. 1999;144:202-204.
19. Smith SH, McOrist S, Green LE. Questionnaire survey of proliferative enteropathy on British pig farms. Vet Rec. 1998;142:690-693.
20. Chia SP, Taylor DJ. Factors affecting the survival of Treponema hyodysenteriae in dysenteric pig feces. Vet Rec. 1978;103:68-70.
21. Lindenstruth RW, Ward BQ. Viability of Vibrio fetus in hay, soil, and manure. JAVMA. 1948;113:163.
References--nonrefereed
10. Bane D, Gebhart C, Gardner I. Epidemiology of porcine proliferative enteropathy: a case study. Proc AASP Ann Meet, Quebec, Canada. 1997;429-431.
11. Jordan DM, Knittel JP, Schmoll EM, Schwartz KJ, Roof MB, Larson DJ, Hoffman LJ. A Lawsonia intracellularis transmission study using a pure culture inoculated seeder-pig sentinel model. Proc AASP Ann Meet, Quebec, Canada. 1997;243-248.
12. Winkelman NL, Pauling GE, Bagg RN, Dick CP, Paradis MA, Brennan J, Wilson J. Use of a challenge model to measure the impact of subclinical porcine proliferative enteritis on growth performance in pigs. Proc AASP Ann Meet, Des Moines, IA. 1998;209-211.
16. Knittel JP, Schwartz KJ, McOrist S, Roof MB, Jordan DM, Harris DL. Colonization of Lawsonia intracellularis in swine is dose dependent. Proc AASP Ann Meet, Nashville, TN. 1996;75-78.