CASE STUDY
Dermal necrosis and mortality from alkali exposure in a group of finisher pigs
Ross P. Cowart, DVM, MS; Stan W. Casteel, DVM, PhD; Lanny W. Pace, DVM, PhD
RCP: Department of Veterinary Medicine and Surgery, A308 Clydesdale Hall, College of Veterinary Medicine, University of Missouri-Columbia, Columbia, Missouri 65211; Email CowartR@missouri.edu; SWC,LWP: Veterinary Medical Diagnostic Laboratory, Columbia, MO.
Cowart RP, Casteel SW, Pace, LW. Dermal necrosis and mortality from alkali exposure in a group of finisher pigs. Swine Health Prod. 2000;8(5):225-227. Also available in PDF format (92k).
Summary
A swine producer reported severe chemical skin burns and high death losses when a group of finisher pigs was exposed to a combination of hydrated lime and sodium bicarbonate. Hydrated lime, an alkalinizing agent commonly used in industry and agriculture, is derived from limestone. We report clinical and experimental evidence that sodium bicarbonate can intensify the caustic effects of this form of lime.
Keywords  : swine, hydrated
lime, alkali burns
: swine, hydrated
lime, alkali burns
Received: April 3, 2000
Accepted: July 5, 2000
Lime is a generic term referring to several compounds derived from limestone, which is a sedimentary rock or fossil consisting primarily of calcium carbonate or a combination of calcium carbonate and magnesium carbonate. Heat treatment of limestone releases carbon dioxide leaving calcium oxide, also known as quicklime. When mixed with water, quicklime forms calcium hydroxide, commonly known as hydrated or slaked lime.
Limestone, quicklime, and hydrated lime are widely used in industry and agriculture as alkalinizing agents.1 The beneficial effects of lime are well recognized. It reduces moisture and odors in animal facilities,2 and when applied to poultry litter2 or sawdust bedding for dairy cows,3 hydrated lime raises the pH, discouraging bacterial proliferation.
The caustic nature of lime and lime products is also well recognized. The pH of a saturated solution of limestone and water ranges between 8 and 9, depending on the solubility of the particular preparation. Quicklime rapidly hydrates in water, forming a solution of calcium hydroxide which may vary in pH from 11 to 13, depending on concentration and temperature.1
Alkaline compounds cause liquefaction necrosis of tissues, producing extensive, penetrating damage in a rapidly progressive manner. Saponification of fats and solubilization of proteins allow deep penetration into tissues. The extent of injury depends on the specific tissue exposed, pH, and contact time. Although esophageal injury may occur at pH 11.8,4 a higher pH is probably required to injure intact skin. Hydration of quicklime (calcium oxide) is an exothermic reaction,5 giving this compound the potential for causing both thermal and caustic burns.
Alkali burns have been reported in a human exposed to calcium hydroxide in wet cement,6 and another who fell into a caustic lime pit.7 The risk of corneal or conjunctival damage makes ocular exposure to alkali of special concern in human medicine.8 Reports of alkali burns in animals are rare, although a case involving swine was reported from the Netherlands.9
Case description
A farrow-to-finish swine producer reported that 140 of 220 pigs housed in a finishing barn had died during a 2-day period. The producer had moved 220 mixed-breed barrows and gilts, ranging in weight from 70-90 kg (154-198 lb), onto empty confinement floors of an open-front, naturally ventilated building with a solid concrete floor. Six pens, each measuring 4.3 m x 21.3 m (14' x 70') had been scraped clean of manure. On the advice of a feed salesman, 22.7 kg (50 lb) of hydrated lime (Ca(OH)2 ) was spread in each pen immediately before the pigs were allowed access. Within 5 hours, the pigs seemed uncomfortable and five had died. As the pigs had rolled in the hydrated lime and had it on their skin, the producer washed them with water and a commercial laundry detergent. He contacted the feed salesman, who suggested that the wrong type of lime had been used, and advised the producer to neutralize the lime by applying sodium bicarbonate (NaHCO3 ) to the skin of the pigs. By the following morning, 140 of the 220 pigs had died.
The next day, two mixed-breed barrows weighing approximately 90 kg (198 lb) were presented to the University of Missouri Veterinary Teaching Hospital, as representative of the herd problem. Both pigs had painful, necrotic skin lesions on the ventral aspect of the abdomen and thorax, the caudal and plantar aspects of the rear legs, and the face and the ears (Figures 1 and 2). One pig was euthanized and necropsied immediately. A complete blood count and a serum chemistry profile on the remaining pig revealed no abnormalities.
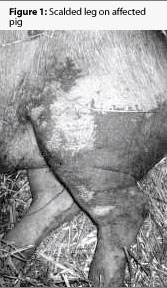
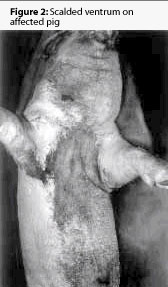
Gross necropsy on the first pig revealed extensive epidermal necrosis, and mild to moderate subcutaneous edema associated with some skin lesions. Mild nasal turbinate atrophy was observed, and there were slightly depressed and reddened lobules in the cranial lung lobes. The stomach mucosa was slightly hyperemic. No other gross lesions were apparent.
Histopathologic findings included full-thickness epidermal necrosis and superficial dermal necrosis. Underlying the necrotic dermis was a mixture of polymorphonuclear and mononuclear inflammatory cells, including multifocal aggregates of eosinophils. Scattered hemorrhages were observed in the deeper dermis. Mild, multifocal peribronchiolar lymphoid hyperplasia was observed in the lungs.
Skin lesions of the hospitalized pig were cleansed with dilute povidone iodine solution and treated with silver sulfadiazine ointment. The pig had a stiff gait and was reluctant to move, but attitude and appetite remained normal. The gait and skin lesions gradually improved, and the pig was discharged from the hospital on the ninth day after admission. Unfortunately, the 78 other surviving pigs were lost to follow-up.
Because deaths and abnormal behavior had been observed after the pigs had contacted the hydrated lime, but before sodium bicarbonate was applied, we questioned which compound caused the dermal necrosis. An experiment was designed to evaluate the caustic potential of various forms of lime individually and in combination with sodium bicarbonate.
Experimental study design
Materials and methods
Two mixed-breed white pigs weighing approximately 30 kg (66 lb) were obtained from a university research herd. Pigs were handled in accordance with the "Guide for the Care and Use of Laboratory Animals," NIH publication 86-23, 1985, and under the supervision of the University of Missouri Animal Care and Use Committee. These pigs were normal by physical examination and observation for each of 6 days prior to initiating the experiment.
Three powdered lime preparations were evaluated for caustic potential according to a modified Draize test.10 The test preparations were
- calcium hydroxide (hydrated lime),
- calcium oxide (quicklime), and
- a mixture of equal parts by dry weight of calcium hydroxide and sodium bicarbonate.
One gram of each of the powdered preparations was applied to
an area of moistened skin (1 sq in) on the right lateral trunk
of each pig. The powdered preparations were secured to the skin
by covering with a non-adhering dressing and an elastic
bandage around the trunk. Pigs were monitored visually
for signs of discomfort, and the bandages were removed after 6
hours.
The test sites were examined for signs of irritation, then washed
with a commercial laundry detergent and water and allowed to dry.
Thirty minutes later, sodium bicarbonate was applied to the site
previously covered with calcium hydroxide. This site was re-bandaged
as previously described, and the pigs were monitored for signs
of discomfort. The bandages were removed after 3 hours and the
sites were examined for signs of irritation.
Seven days after the first experiment, a second experiment was performed to evaluate each compound applied as an aqueous paste. One gram of each test compound was mixed with 2 mL of water to make a paste, which was applied to the left side of each pig as previously described. Test sites were observed and examined as in first experiment. The calcium hydroxide test site on each pig was cleaned and an aqueous paste of sodium bicarbonate was applied. This site was bandaged and examined after 3 hours as described for the first experiment.
Results
In the first experiment, neither pig developed any detectable dermal reaction to any of the three test materials applied as dry powder, nor were any signs of discomfort observed. No detectable dermal reaction was observed at the site where the sodium bicarbonate had been applied after the calcium hydroxide.
In the second experiment, both pigs showed signs of mild discomfort as evidenced by rubbing their left sides against the pens. They appeared otherwise normal. When the bandages were first removed, both pigs showed a severe dermal reaction at the site of the calcium hydroxide and sodium bicarbonate combination paste. In each case, there was a dark necrotic eschar 4-5 cm in diameter, with mild peripheral erythema. One pig developed an area of erythema without necrosis, approximately 1 cm in diameter, at the site where calcium oxide paste had been applied.
Discussion
In this case, the lime preparation spread on the pens was labeled calcium hydroxide. The owner of the pigs reported abnormal behavior and deaths presumed to be caused by caustic effects of the lime. However, in our experiments, we could not demonstrate caustic effects from calcium hydroxide alone. We do not have a completely satisfactory explanation for the abnormal behavior and deaths of the first five pigs. Perhaps a wider distribution of lime over the pigs' bodies and a longer exposure time might have created some discomfort; however, we do not believe that exposure to the hydrated lime alone caused the dermal injury and high death losses seen in this case.
When sodium bicarbonate was applied to the skin of the pigs in an effort to neutralize the effects of the lime, it produced the opposite effect. Calcium hydroxide (Ca(OH)2) and sodium bicarbonate (NaHCO3), both weak alkalis, combine to form sodium hydroxide (NaOH), a strong alkali known as lye. This chemical reaction is represented by the following equation:
Ca(OH)2 + NaHCO3 --> NaOH + CaCO3 + H2O
We believe the sodium hydroxide was responsible for the injury and death of most of the pigs. This hypothesis is supported by our experimental observations. The paste made of hydrated lime and sodium bicarbonate caused lesions comparable to those observed in the field case. We believe that the lesions observed in the nasal cavity, lungs, and stomach of the field case were incidental and unrelated to the primary cause of death, that is, caustic alkali burns.
We included quicklime (calcium oxide) in the patch testing in case the lime used on the farm had actually been quicklime, although it was labeled "hydrated lime." One pig showed some reaction to the quicklime and water patch, possibly because of the exothermic reaction of quicklime and water, but this reaction was mild compared to the one at the hydrated lime-sodium bicarbonate site.
When caustic alkalis such as sodium hydroxide contact tissues, the hydroxide anion saponifies fatty acids in cell membranes, destroying membrane integrity, and also causes swelling and shortening of collagen. Focal softening and liquefaction of the tissue ensue, a condition referred to as liquefactive necrosis. In severe burns, the release of inflammatory mediators may cause hemodynamic disturbances, including circulatory shock and ischemic-reperfusion injury. Death may occur due to circulatory failure or multiple organ failure.11 Multiple organ failure is believed to be the most common cause of death in burn patients even in the absence of uncontrolled infection.12 In these pigs, death was probably caused by shock syndrome and multiple organ failure after more than 50% of their skin surface was severely burned by caustic chemicals.
In this case, lime was used in the relative absence of organic
matter or bedding, contrary to uses suggested in the literature.
Organic matter could be expected to
buffer some of the alkalizing effects of lime. Although we do
not believe that the lime alone was responsible for the injury
to these pigs, it might be prudent to exercise caution when using
lime in animal environments without organic matter for bedding.
The initial response of the producer in this case was to wash the pigs with water and laundry detergent. This response was appropriate, as the cornerstone of treatment for any chemical skin burn is copious lavage with water. Burns caused by strong alkalis may require continuous lavage for one to two hours before tissue pH returns to normal.13 Neutralization of a chemical with a weak acid-base opposite is not recommended, because the search for the neutralizing agent may delay immediate lavage, and the neutralization reaction may be exothermic, causing further damage.13 This case illustrates a third reason for not attempting neutralization treatment: the wrong neutralizing agent may be chosen.
Implications
- Hydrated lime and sodium bicarbonate are common agricultural and household chemicals. Each compound individually is a mild alkali with minimal or slight caustic potential.
- Hydrated lime and sodium bicarbonate combine in water to form sodium hydroxide, a strong alkali and caustic agent capable of causing severe alkali burns.
- Suspected chemical skin burns should be treated with copious lavage with water. Neutralization treatment is not recommended.
References--refereed
1. Boynton RS. Chemistry and Technology of Lime and Limestone. New York: John Wiley & Sons; 1966:182-185.
2. Boynton RS. Chemistry and Technology of Lime and Limestone. New York: John Wiley & Sons; 1966: 385-386.
3. Hogan JS, Smith KL. Bacteria counts in sawdust bedding. J Dairy Sci. 1997;80:1600-1605.
4. Ellenhorn MJ, Schonwald S, Ordog G, Wasserberger J. Ellenhorn's Medical Toxicology: Diagnosis and Treatment of Human Poisoning. 2nd ed. Baltimore: Williams & Wilkins; 1997:1093-1097.
5. Nebergall WH, Schmidt FC, Holtzclaw HF. College Chemistry with Qualitative Analysis. 5th ed. Lexington, Massachusetts: D.C. Heath and Company; 1976:930-932.
6. Early SH, Simpson RL. Caustic burns from contact with wet cement. JAMA. 1985;254:528-529.
7. Erdmann D, Hussmann J, Kucan JO. Treatment of a severe alkali burn. Burns. 1996;22:141-146.
8. Rozenbaum D, Baruchin AM, Dafna Z. Chemical burns of the eye with special reference to alkali burns. Burns. 1991;17:136-140.
9. Anonymous. Het gebruik van landbouwpoederkalk in varkensstallen. [The use of powdered agricultural calcium in pig sties]. Tijdschrift voor Diergeneeskunde. 1986;111:870.
10. Draize JH, Woodward G, Calvery HO. Methods for the study of irritation and toxicity of substances applied topically to the skin and mucous membranes. J Pharmacol Exp Ther. 1944;82:377-390.
11. Huang YS, Yang ZC, Liu XS, Chen FM, He BB, Li A, Crowther RS. Serial experimental and clinical studies on the pathogenesis of multiple organ dysfunction syndrome (MODS) in severe burns. Burns. 1998;24:706-716.
12. Sheridan RL, Ryan CM, Yin LM, Hurley J, Tompkins RG. Death in the burn unit: Sterile multiple organ failure. Burns. 1998;24:307-311.
13. Stewart CE. Chemical skin burns. Am Family Physician. 1985;31:149-157.
