Patterson JL, Cameron AC, Smith TA, et al. The effect of gonadotrophin treatment at weaning on primiparous sow performance. J Swine Health Prod. 2010;18(4):196–199
| Brief communication | Peer reviewed |
Cite as: Patterson JL, Cameron AC, Smith TA, et al. The effect of gonadotrophin treatment at weaning on primiparous sow performance. J Swine Health Prod. 2010;18(4):196–199.
Also available as a PDF.
SummaryIn sows treated at weaning with gonadotrophins, weaning-to-estrus interval was shorter than in untreated controls, but the proportions of sows bred within a 3-day period or 7 days after weaning, farrowing rate, total born, and born alive did not differ with treatment. | ResumenEn hembras tratadas al destete con gonadotropinas, el intervalo de destete a estro fue más corto que en los controles no tratados, pero las proporciones de hembras inseminadas dentro de un periodo de 3 días ó 7 días después del destete, el índice de partos, total de nacidos, y nacidos vivos no difirió con el tratamiento. | ResuméChez des truies traitées au moment du sevrage avec des gonadotrophines, l’intervalle sevrage-Å“strus était plus court que chez les animaux témoins non-traités, mais les proportions de truies saillies à l’intérieur d’une période de 3 jours ou 7 jours après le sevrage, le taux de mise-bas, le nombre total d’animaux nés, et de nés-vivants ne différaient pas entre les traitements. |
Keywords: swine, sows, weaning, gonadotrophin
Search the AASV web site
for pages with similar keywords.
Received: November 11, 2009
Accepted: February 22, 2010
The principal goal of commercial breeding swine herds is to consistently meet weekly breeding targets. Weaned sows failing to return to estrus within 7 days after weaning contribute to missed breeding targets and increase nonproductive sow days. Historically, extended weaning-to-estrus intervals (WEI) have been a concern in primiparous sows and associated with short lactation length, larger litter size weaned, season, and inadequate feed intake during lactation.1,2 However, in well-managed contemporary sow farms, more than 90% of sows may return to estrus within 3 to 5 days after weaning,3 and in an experimental setting, even feed restriction of primiparous sows during the last week of lactation had no effect on WEI.4
The primary objective of the present study was therefore to determine the response to gonadotrophin treatment at weaning in contemporary parity-one commercial sows with lactation lengths typical of the North American swine industry.
Materials and methods
All animal procedures were carried out in accordance with the recommended guidelines of the Canadian Council for Animal Care.5 The study was conducted under the supervision of the herd veterinarian (D. Augsburger) and with technical support from staff certified for the use of swine in research (J. Patterson, A. Cameron, T. Smith, G. Foxcroft; University of Alberta) or staff with extensive experience in the application of research protocols in a commercial farm setting (R. Schott, PIC; L. Greiner, Innovative Swine Solutions).
This study was conducted from January to April 2008 during the start-up phase of a 6000-sow commercial farrow-to-wean facility located in western Illinois. Throughout the trial, the herd was checked monthly for serological changes using an enzyme-linked immunosorbent assay (ELISA) for porcine reproductive and respiratory syndrome (PRRS) virus antibody. Positive ELISAs were then confirmed via a polymerase chain reaction (PCR) for PRRS virus. This herd was PRRS-negative both by ELISA and PCR, and no other health issues were noted throughout the trial period. In order to estimate postfarrowing weight, a subset of sows (n = 94) were weighed before they entered the farrowing rooms and again within 24 hours after farrowing. Total born, born alive, stillborn, and mummies were recorded for all sows. Simple regression analysis was used to predict postfarrowing weight on the basis of pre-farrowing weight and litter size (r2 = 0.88, P < .001).
Primiparous crossbred sows were blocked by genetic line (Pig Improvement Company [PIC] C22, n = 285; and PIC C29, n = 122), estimated postfarrowing weight, and total born at parity one. Sows were then randomly allocated within block to either receive a 5-mL dose of a combination of 400 IU equine chorionic gonadotrophin (eCG) and 200 IU human chorionic gonadotrophin (hCG) (PG600; Intervet Schering-Plough Animal Health, USA, De Soto, Kansas) intramuscularly in the neck on the morning of weaning (Day 0; PG group; n = 189), or to be uninjected controls (CONT group; n = 218). In addition to individual herd-specific ear tags, sows were provided a unique treatment ear tag after assignment to treatment.
From the day after weaning, all sows were provided twice daily (8:00 am and 3:00 pm) fence-line contact with mature boars for stimulation and detection of estrus. Sows were bred by artificial insemination (AI) according to established herd protocols, with doses of 3.0 × 109 sperm cells per insemination dose. Semen was stored in a long-term extender (Androhep Plus; Minitube of America, Verona, Wisconsin) and delivered to the farm four times per week. Farm policy stipulated that unused semen be discarded on day 5 after delivery. At breeding for the sows utilized in this study, semen was on average 2.1 ± 0.1 days old.
For sows first detected in heat on or before Day 4, breeding was delayed for 24 hours, and then sows were bred every am and pm until they were no longer standing. For sows first detected in heat on Day 5, breeding was delayed 8 to 16 hours (next am or pm heat check) and then sows were bred every am and pm until they were no longer standing. Sows first detected in heat on Day 6 or later after weaning were bred at first detection of standing heat and then every am and pm until they were no longer standing.
For detection of nonpregnant sows, pregnancy diagnosis was confirmed by ultrasonography at day 30 after the last insemination, and heat detection was performed daily from approximately day 18 after breeding until sows were moved into the farrowing rooms at day 112 of gestation.
Gilts were fed approximately 1.8 to 2.0 kg of a commercial gestation diet once per day. Fourteen days before their predicted farrowing date, daily feed allowance was increased by 0.9 kg until sows were moved into farrowing rooms. In the farrowing rooms, sows were fed 1.8 kg daily until day 2 after farrowing and 2.7 kg on day 3, and were then fed to appetite with a commercial lactation diet for the remainder of lactation.
To ensure the reliability of the results, sows receiving only a single insemination were not included in the analysis (n = 32; PG = 21, CONT = 11), and the results are based on data from 375 sows inseminated at least twice. Reproductive parameters analyzed were estrus synchronization rate, determined as the number of sows with first observed estrus within 7 days after weaning; weaning-to-estrus interval (WEI); proportion of sows bred that farrowed; total litter-size born; and born-alive piglets at farrowing. In addition, within each treatment, the synchrony of sows returning to estrus was determined as the proportion of sows in estrus within a 3-day period. A fertility index6 was calculated as total pigs produced per 100 weaned sows bred within a designated breeding week (percentage of sows bred by 7 days × farrowing rate × litter size). Estimated postfarrowing weight, measured weaning weight, and estimated lactation weight loss were recorded for all sows.
The fixed effects of treatment (CONT versus PG), genotype (C22 versus C29), and their interaction on the various parameters measured were analyzed using a linear mixed effect model (SAS Institute Inc, Cary, North Carolina). Data were examined for normality and homogeneity of variance. Effects of treatment on the percentage of sows exhibiting estrus within set periods after weaning and breeding and farrowing rates were analyzed using a chi-squared test. Results are presented as least squares means ± SE or as proportions, and P < .05 was considered statistically significant.
Results
Parity one total litter size (12.2 ± 0.2 versus 12.0 ± 0.3), estimated farrowing weight (194.0 ± 1.7 versus 192.1 ± 1.9 kg), and weaning weight (189.6 ± 1.4 versus 189.3 ± 1.6 kg) did not differ (P > .05) in CONT and PG sows, respectively, indicating balanced allocation of sows across treatment. Lactation length (19.5 ± 0.1 versus 19.1 ± 0.1 days) and estimated weight loss in lactation (4.6 ± 1.3 versus 2.8 ± 1.0 kg) did not differ by treatment (P > .05) in CONT and PG sows, respectively.
Considering data from all sows assigned to treatment, there was no effect of genetic line or a treatment × genetic line interaction on the characteristics measured, therefore, only the main effect of treatment is presented (Table 1). Treatment did not affect the proportion of sows in estrus within 7 days after weaning (estrus synchronization rate), or within a peak 3-day breeding window (Table 1). However, the timing of this 3-day breeding window (Days 4, 5, and 6 for CONT versus Days 3, 4, and 5 for PG sows) reflected a shorter WEI in PG than in CONT sows (P < .001; Figure 1). Pregnancy rate, total born, and born alive did not differ by treatment. As a simple overall comparison of sow productivity between CONT and PG sows, a combined fertility index was calculated and indicated no gain in productivity in response to gonadotrophin (GT) treatment.
Table 1: Effects of treatment with exogenous gonadotrophins (PG)* or no treatment (CONT) at weaning on subsequent fertility in primiparous sows bred with at least two inseminations within 7 days after weaning in a commercial farrow-to-wean facility
* Treated sows received a combination dose of 400 IU equine chorionic gonadotrophin and 200 IU human chorionic gonadotrophin (PG600; Intervet Schering-Plough Animal Health, USA, De Soto, Kansas) administered intramuscularly in the neck on the morning of weaning. The trial was conducted from January to April 2008. † Number of sows with first estrus observed within 7 days after weaning. ‡ Results are presented as least squares means ± SE or proportions, and P < .05 was considered statistically significant. Data were analyzed using a linear mixed effect model and using a chi-squared procedure in SAS (SAS Institute Inc, Cary, North Carolina). The main effect of treatment (CONT versus PG) is shown; the effect of genotype (PIC C22 versus PIC C29), and the treatment × genotype interaction are not shown (P > .05). § Percentage of sows bred over a 3-day period. NA = not applicable
|
||||||||||||||||||||||||||||||||||||||||||
Figure 1: Effects of treatment with exogenous gonadotrophins (PG) or no treatment (CONT) on the overall distribution of weaning-to-estrus interval (WEI). Treatment described in Table 1. The red and blue lines reflect the advancement in WEI for PG and CONT sows, respectively. Treatment did not affect the proportion or the accumulative percentage of sows in estrus within 7 days after weaning or within a peak 3-day breeding window. However, the timing of this 3-day breeding window (Days 4, 5, and 6 for CONT; Days 3, 4, and 5 for PG sows) reflected a shorter WEI in PG than in CONT sows (P < .001). 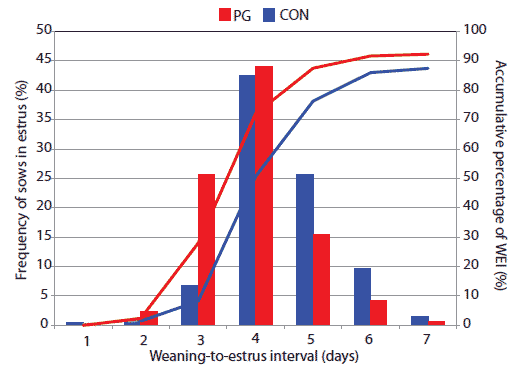 |
Discussion
Results from a previous study7 showed that a single insemination between 0 and 24 hours before ovulation resulted in optimal fertilization, and sows inseminated after ovulation had a significantly lower percentage of normal embryos. Furthermore, it has been suggested that the optimal time for insemination lies in the interval from 28 hours before to 4 hours after ovulation.8 In the present study, the timing of at least one insemination would fall within the above optimal interval in any sow receiving multiple inseminations. However, in sows receiving only a single insemination due to a short heat duration or inadequate heat detection, the optimal breeding window might be missed and the sow might even be bred a considerable time after ovulation. Therefore, in analyzing the results of the present study, data from 32 sows (PG = 21, CONT = 11) inseminated only once were removed.
In the present study, synchronization of estrus in GT treatment (PG) was not superior to that in no treatment (CONT), as measured by the percentage of sows bred over a 3-day period. However, PG sows were first detected in heat on Days 3, 4, and 5, while CONT sows were first detected in heat on Days 4, 5, and 6, which is consistent with earlier reports that GT treatment shortens the WEI.2,6,9,10 This shorter WEI has not been associated with any change in estrus duration, timing of ovulation with respect to estrus duration, or size of the largest preovulatory follicle.10 Therefore, in the current study, time of ovulation was assumed to be advanced by a similar amount of time equal to the advance in the WEI in PG sows. Most commercial herds have established multiple-insemination breeding protocols that ensure that at least one AI occurs near ovulation in > 90% of inseminated sows for optimal fertilization.11 However, it has been reported that it may be advantageous to inseminate sows with semen < 72 hours old rather than ≥ 72 hours old.10 Therefore, depending on the schedule of semen delivery to the farm, it may be advantageous to advance the onset of breeding in association with use of GT treatment to match optimal semen age and quality. Other estrus-synchronization12 and ovulation-synchronization13,14 protocols have been successfully applied in the swine industry. Although the synchrony did not differ by treatment in this study, the early and synchronous return to estrus in GT-treated sows after weaning is consistent with these emerging protocols and the introduction of single fixed-time insemination protocols.
Consistent with earlier reports, GT treatment had no effect on estrus synchronization or farrowing rate.2,9 In the current study, and consistent with other studies,2,10 subsequent litter size was not affected by treatment, in contrast to reports showing smaller6,9 or larger2 litter size after GT treatment. These differences in the response to GT treatment may have been influenced by the very synchronous return to estrus seen in the control sows in the present study, which was a result of good lactational management and marginal overall loss in estimated sow body weight during lactation.
Summer infertility and high ambient temperatures often result in extended WEI, increased regular returns to estrus, and decreased litter size; furthermore, primiparous females may be most susceptible.15,16 As this study was conducted during the months of January to April, when summer infertility would not normally be considered a risk in North America, this may explain the lack of gain in the fertility index (total pigs produced per weaned sow) in response to GT treatment, in contrast to an increase in fertility index in an earlier study.6 Use of GT treatment during periods of seasonal infertility should be studied further.
Implications
- Given the excellent reproductive performance of weaned primiparous sows under the conditions of this experiment, there appears to be little benefit of using GT to improve postweaning performance.
- In the application of fixed-time AI protocols in swine breeding management, there may still be advantages of gonadotrophin treatment at weaning.
References
1. Hughes PE. Effects of parity, season and boar contact on the reproductive performance of weaned sows. Livest Prod Sci. 1998;54:151–157.
2. Vargas AJ, Bernardi ML, Wentz I, Borchardt Neto G, Bortolozzo FP. Time of ovulation and reproductive performance over three parities after treatment of primiparous sows with PG600. Theriogenology. 2006;66:2017–2023.
3. Poleze E, Bernardi ML, Amaral Filha WS, Wentz I, Bortolozzo FP. Consequences of variation in weaning-to-estrus interval on reproductive performance of swine females. Livest Sci. 2006;103:124–130.
4. Vinsky MD, Novak S, Dixon WT, Dyck MK, Foxcroft GR. Nutritional restriction in lactating primiparous sows selectively affects female embryo survival and overall litter development. Reprod Fertil Dev. 2006;18:347–355.
5. Canadian Council on Animal Care. Guidelines on: the care and use of farm animals in research, teaching and testing. Ottawa, Ontario, Canada; 2009. Available at: http://www.ccac.ca/en/CCAC_Programs/Guidelines_Policies/GDLINES/Guidelis.htm. Accessed 6 May 2010.
6. Kirkwood RN, Aherne FX, Foxcroft GR. Effect of gonadotropin at weaning on reproductive performance of primiparous sows. Swine Health Prod. 1998;6:51–55.
7. Soede NM, Wetzels CCH, Zondag W, de Koning MAI, Kemp B. Effects of time of insemination relative to ovulation, as determined by ultrasonography, on fertilization rate and accessory sperm count in sows. J Reprod Fert. 1995;104:99–106.
8. Nissen AK, Soede NM, Hyttel P, Schmidt M, D’Hoore L. The influence of time of insemination relative to time of ovulation on farrowing frequency and litter size in sows, as investigated by ultrasonography. Theriogenology. 1997;47:1571–1582.
9. Kirkwood RN. Pharmacological intervention in swine reproduction. Swine Health Prod. 1999;7:29–35.
10. Knox RV, Rodriguez-Zas SL, Miler GM, Willenburg KL, Robb JA. Administration of P.G. 600 to sows at weaning and the time of ovulation as determined by transrectal ultrasound. J Anim Sci. 2001;79:796–802.
11. Belstra BA, Flowers WL, See MT. Factors affecting temporal relationships between estrus and ovulation in commercial sow farms. Anim Reprod Sci. 2004;84:377–394.
12. Patterson J, Wellen A, Hahn M, Pasternak A, Lowe J, DeHaas S, Kraus D, Williams N, Foxcroft G. Responses to delayed estrus after weaning in sows using oral progestagen treatment. J Anim Sci. 2008;86:1996–2004.
13. Zak LJ, Patterson J, Hancock J, Rogan D, Foxcroft GF. Benefits of synchronizing ovulation with porcine luteinizing hormone (pLH) in a fixed time insemination protocol in weaned multiparous sows. In: Rodriquez-Martinez H, Vallet JL, Ziecik AJ, eds. Control of Pig Reproduction VIII. 8th ed. Nottingham, United Kingdom: Nottingham University Press; 2009;66:305–306.
*14. Johnston ME, Gaines AM, Swanson ME, Webel SK. Timed insemination following GnRH agonist administration in weaned sows. Proc VIII Int Conf Pig Repro. Banff, Alberta. 2009;170.
15. Bates RO, Kelpinski J, Hines B, Ricker D. Hormonal therapy for sows weaned during fall and winter. J Anim Sci. 2000;78:2068–2071.
16. Almond PK, Bilkei G. Seasonal infertility in large pig production units in an Eastern-European climate. Aust Vet J. 2005;83:344–346.
* Non-refereed reference.