| Original research | Peer reviewed |
Cite as: Newman SJ, Rohrbach BW, Wilson ME, et al. Characterization of histopathologic lesions among pigs with overgrown claws. J Swine Health Prod. 2015;23(2):91–96.
Also available as a PDF.
SummaryObjective: To characterize histologic lesions in pigs with and without claw overgrowth. Materials and methods: Hindlimb claws from a subset of 24 sows that were part of a larger field study were selected because of claw deformities associated with overgrowth and change in gait. Length measurements were available for 72 lateral or medial rear claws. Claws were examined histologically and the lesions categorized. Overgrowth was defined as a toe growth measuring > 50 mm in length. Results: Lateral rear claws were most consistently overgrown. However, the distribution and severity of lesions failed to suggest a common etiology for overgrowth. Inflammation, arteriosclerosis, lamellar epithelial changes, phalanx rotation, or combinations of these were not prominent gross or histologic changes. Implications: The pathogenesis of overgrowth in this collection of claws is unknown, but does not appear to represent primary laminitis in this species. As lameness continues to prompt a significant economic loss due to culling, further studies on claw overgrowth, its effect on motion, and its pathogenesis are warranted. | ResumenObjetivo: Caracterizar las lesiones histológicas en cerdos con y sin pezuñas aumentadas de tamaño. Materiales y métodos: Se seleccionaron pezuñas de patas traseras de un subgrupo de 24 hembras que formaban parte de un estudio de campo más grande, debido a las deformidades de las pezuñas relacionadas con aumento de crecimiento y paso alterado. Las medidas de longitud estuvieron disponibles para 72 pezuñas traseras medias o laterales. Se examinaron las pezuñas histológicamente y se categorizaron las lesiones. El aumento en el crecimiento se definió como el crecimiento del dedo con una medida > 50 mm de longitud. Resultados: Las pezuñas traseras laterales fueron consistentemente más grandes. Sin embargo, la distribución y la severidad de las lesiones no sugieren una etiología común de aumento en el crecimiento. La inflamación, la arteriosclerosis, los cambios epiteliales laminares, la rotación de falange, o las combinaciones de estos no fueron cambios prominentes macro ni histológicos. Implicaciones: La patogénesis del aumento de tamaño en esta serie de pezuñas es desconocida, pero no parece representar laminitis primaria en esta especie. Debido que la cojera continua provocando una pérdida económica significante debido al desecho, es necesario hacer más estudios sobre el aumento de tamaño de las pezuñas, sus efectos en el movimiento, y su patogénesis. | ResuméObjectif: Caractériser les lésions histologiques chez des porcs avec et sans onglons à croissance exagérée. Matériels et méthodes: Les onglons des pattes arrière d’un sous-groupe de 24 truies faisant partie d’un plus grand groupe d’étude furent sélectionnés à cause des difformités associées à la croissance exagérée des onglons et de changement dans la posture. Des mesures de la longueur de 72 onglons latéraux et médiaux étaient disponibles. Un examen histologique des lésions fut effectué et les lésions caractérisées. Une croissance exagérée était définie comme étant la croissance d’un orteil mesurant > 50 mm en longueur. Résultats: Les onglons latéraux arrière étaient ceux présentant le plus souvent une croissance exagérée. Toutefois, la distribution et la sévérité des lésions n’ont pas permis de trouver une étiologie commune pour cette croissance exagérée. L’inflammation, l’artériosclérose, des changements épithéliaux lamellaires, la rotation de phalange, ou toutes les combinaisons de ces changements n’étaient pas des changements macroscopiques ou histologiques évidents. Implications: La pathogénie de la croissance exagérée dans la présente collection d’onglons est inconnue, mais ne semble pas représenter une laminite primaire chez cette espèce. Étant donné que les maux de pattes continuent de représenter des pertes économiques significatives à cause de la réforme des animaux, des études supplémentaires sur la croissance exagérée des onglons, ses effets sur la mobilité, et sa pathogénie sont justifiées. |
Keywords: swine, claw, corium, hoof, laminitis
Search the AASV web site
for pages with similar keywords.
Received: March 24, 2014
Accepted: September 16, 2014
Lameness resulting from overgrown claws in large commercial swine operations in the United States and other countries has become an important economic and welfare issue.1 Routine claw trimming can provide some relief, but is labor intensive. Early culling is another detrimental outcome. Causes of claw overgrowth may be multiple and the result of an extended duration from point of insult until pathologic assessment. Causes include toxic,2 parturition-associated,2,3 or nutritional etiologies,2 and may be affected by management, housing, body weight, weight distribution on individual claws, trauma, or combinations of these.2,4
In cattle with inflammatory disease of the corium, defined as laminitis, claws are often overgrown and contain horizontal and vertical wall grooves, cracks, and white-line separations.5 These gross changes are also observed in swine, but an association with a primary inflammatory condition is less clearly determined in pigs due to the few descriptive histologic studies reported in the peer-reviewed literature for swine.2-4 A recent publication from Brazil characterized laminitis in pigs by radiographic evaluation, and distal phalanx rotation was confirmed in one or multiple digits.4 Additionally, the lateral claw was consistently longer than the medial claw.4 In another study, claws of 11 pigs with chronic lameness were histologically assessed.2 Five had sclerosis and dyskeratosis, which those authors associated with faulty horn formation and lameness. In a series of acute lameness cases from 24 post-parturient pigs, histological examination showed hyperemia, hemorrhage, thrombosis, swelling and disorganization of epithelial cells of the stratum germinativum layer, and, rarely, retraction of the keratin layer from the most basal four or five layers of epithelium, resulting in a cleft.3 Causes of swine claw lesions were often undetermined, but considerations included toxicosis2 and parturition-related2,3 or nutritional reasons.2
Continued studies of claw lesions are necessary to characterize claw overgrowth in pigs and determine contributing causes for development of claw overgrowth. The objectives of this study were to evaluate the available rear claws from 24 sows and assess histologic lesions in hopes of discovering more about the pathologic changes associated with overgrown claws, and to compare the lesions to those seen in laminitis.
Materials and methods
The specimens for this study were obtained from a larger study that was conducted in accordance with the Institutional Animal Care and Use Committee (IACUC) guidelines.
A group of 24 commercial Landrace-Large White F-1 crossbred sows, ranging from third to seventh parity (median, fourth parity) were housed at a large facility and selected for this study because of gross overgrowth of one or both hind claws in the absence of significant lameness. In these sows, the caregivers noticed a change in locomotion that they attributed to the necessity for these sows to lift their limbs differently, but none were assigned lameness scores prior to slaughter. The examined claws were selected from a representative cohort of sows from a larger group with varying degrees of claw overgrowth. The total number of sows was based on convenience and included the number that could be transported to slaughter simultaneously.
Sows were fed a corn-soybean-based gestation and lactation ration or a diet supplemented with Availa Zn (50 mg zinc-amino acid complex per kg feed), Mn (20 mg manganese-amino acid complex per kg feed), and Cu (10 mg copper-amino acid complex per kg feed) (Zinpro Performance Minerals, Eden Prairie, Minnesota) for partial replacement of inorganic trace minerals.
All pigs were housed in swine barns designed with heating and cooling from a forced-air system. Sows were housed on fully slatted concrete floors during breeding and gestation periods, and in farrowing stalls with cast-iron slatted flooring during periparturient and lactation periods. Wells providing water for the farm site were charged with chlorine monthly. During gestation, water was delivered via trough waterers filled twice daily. Water in farrowing crates was from nipple waterers. Parasite control programs were not used in these sows.
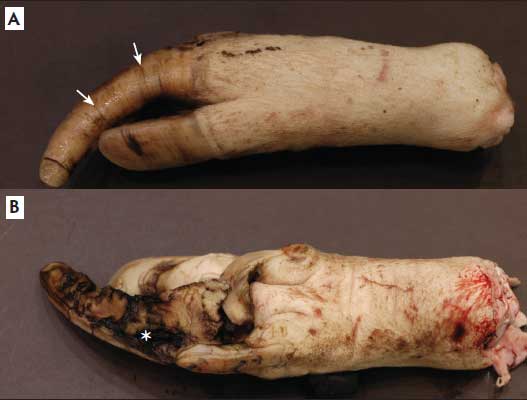
The two rear feet collected at slaughter from each of 24 sows were evaluated at the University of Tennessee, Department of Biomedical and Diagnostic Sciences, Anatomic Pathology Division (Knoxville, Tennessee). Rear claws were chosen for assessment because of their propensity to be overgrown.4,6-9 Photographs of a range of normal, overgrown, and deformed claws were taken by the pathologist to categorize the lesions prior to histologic sample collection (overgrown claws shown in Figure 1). Control rear claws collected from six sows with visibly normal rear feet at slaughter were used for comparisons. Claws were sectioned sagittally with a band saw to obtain thin slices for formalin fixation. Two authors (SJN, SVA) assessed the slices for phalanx rotation. In some instances, feet were further sectioned into numerous smaller pieces to facilitate easier formalin storage prior to measurement. Hence, only claws with complete measurements of length from the coronary band to the tip of the toe were included in the study (total 72 of 96 claws).
Figure 1: Right hind hoof of a sow with severely overgrown claws. Both dorsal (A) and ventral (B) claw surfaces are shown. Note horizontal grooves (A; arrows) and accompanying ulcerative lesions ventrally (B; asterisk).
Measurements for each claw were determined and recorded. Determination of what defined overgrowth (claw lengths > 50 mm) was extrapolated from work on claw-length measurements in gilts published by one of the authors (SVA).10 Claws were then divided into representative, full-thickness, 2- to 5-mm sagittal sections, and the samples were fixed in 10% buffered formalin. Representative sections of each claw were obtained by using a razor blade to remove the soft tissue between the bone of the third phalanx and the hoof wall. The samples were subsequently oriented within the tissue-processing cassette so that laminae were cut in longitudinal sections to allow for best viewing. In those instances where very small regions of lamellar or coronary corium were present, tissue sections were re-oriented by melting the paraffin blocks, then placing the tissues at a 90° angle to the original orientation in order to maximize the information obtained from each sample. Tissue samples were routinely processed, embedded in paraffin, sectioned at 3 µm, and stained with hematoxylin and eosin (HE).11 A single board-certified pathologist (SJN) reviewed the slides in a blinded manner. Histopathological findings for each claw were recorded as present or absent. Pathologic lesions were evaluated for the following categories: lymphoplasmacytic inflammation, neutrophilic inflammation, eosinophilic inflammation, or overall inflammation (if a combination of types were present); and epithelial apoptosis, epithelial necrosis, epithelial vacuolation, or overall epithelial changes (if a combination of types were present). Additionally, an assessment for arteriosclerosis of chronic laminitis was conducted using HE-stained sections.
Statistical analysis
A chi-square test was used when an expected cell value in the 2 × 2 table was < 5, but when an expected cell value was ≥ 5, the more statistically appropriate test, the Fisher’s exact test, was used to evaluate the association between overgrowth and histopathological findings. All analyses were performed with a commercial software program (SAS version 9.2; SAS Institute, Cary, North Carolina); P values < .05 were considered significant in all tests.
Results
Grossly, 39 claws (54%) were considered overgrown, and 33 claws (46%) were normal length. The range of lengths for the overgrown claws was 51 to 79 mm (median, 59.5 mm), and the range of lengths for normal claws was 38 to 50 mm (median, 45.0 mm). Lateral claws were more commonly overgrown (26 of 39; 67%) than medial claws (13 of 33; 39%). Examples of overgrown claws are shown in Figure 1. Two authors (SJN, SVA) examined all sagittal sections of claws and did not identify distal phalanx rotation in any of them. Because phalanx rotation was not identified in any of the samples, statistical assessment was not performed.
Coronary corium was available from the majority of the samples (66 of 72; 92%), and lamellar corium was available from 17 of 72 (24%). All claws had at least one corium, either lamellar or coronary, available for assessment, and 14 of 72 (19%) had both. Examples of representative overgrown and normal length claws and the distribution of histologic lesions (Figure 2) are presented along with P values for comparisons in Table 1.
Figure 2: Histologic sections of the hooves of sows with overgrown claws. A: Within the lamellar epithelium are apoptotic cells, cellular vacuolation (arrowheads), and excessive accumulations of keratin (×20). B: Moderate numbers of perivascular eosinophils (arrows) are present (×40). C: Inflammatory response (asterisks) in the epithelium and subepithelium (×4). All sections stained with hematoxylin and eosin.
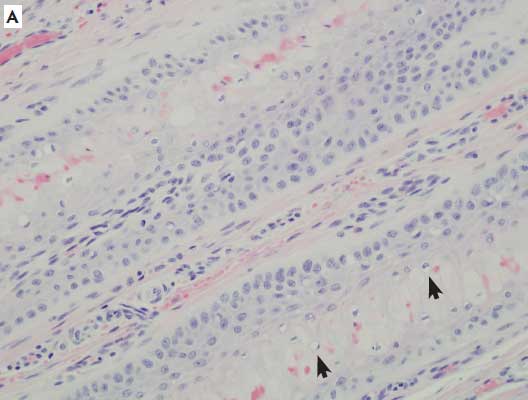
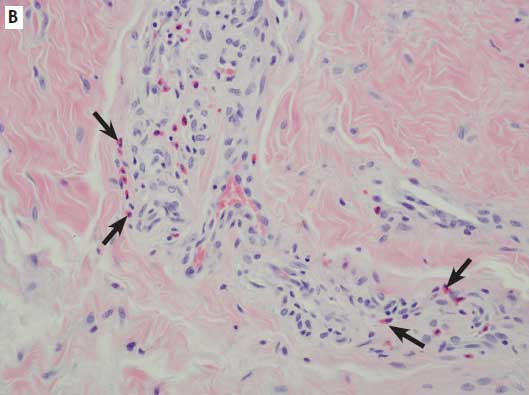
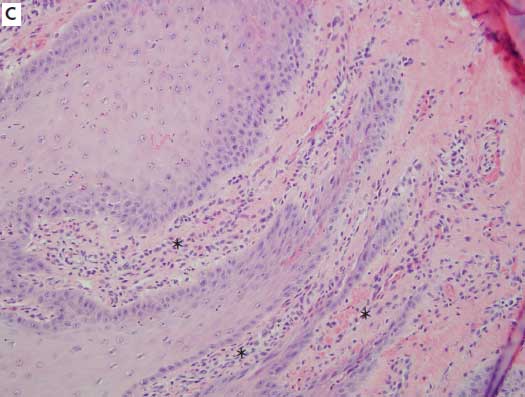
Table 1: Comparison of histological characteristics among a sample of normal (n = 33) and overgrown (n = 39) porcine claws*
| No. overgrown (%) | P† | ||
|---|---|---|---|
| Lesion | Yes | No | |
| Apoptosis | 4 (10) | 0 (0) | .12 |
| Eosinophilic inflammation | 8 (21) | 5 (15) | .56 |
| Epithelial necrosis | 2 (5) | 0 (0) | .50 |
| Lymphoplasmacytic inflammation | 8 (21) | 9 (27) | .50 |
| Neutrophilic inflammation | 6 (15) | 4 (12) | .75 |
| Vacuolation | 6 (15) | 3 (9) | .49 |
| Combined epithelial parameters | 9 (23) | 3 (9) | .12 |
| Combined inflammatory parameters | 14 (36) | 12 (36) | .97 |
Pathologic lesions were evaluated for the following categories: lymphoplasmacytic inflammation, neutrophilic inflammation, eosinophilic inflammation, or overall inflammation (if a combination of types were present); and epithelial apoptosis, epithelial necrosis, epithelial vacuolation, or overall epithelial changes (if a combination of types were present).
† A chi-square test was used when an expected cell value in the 2 × 2 table was < 5; Fisher’s exact test was used when an expected cell value was ≥ 5. For all comparisons, P < .05 was considered significant.
Overgrown claws were not significantly associated with any individual inflammatory cell infiltrate nor a combination of inflammatory cell types (inflammation). Additionally, overgrown claws were not significantly more associated with changes in the epithelium of the corium or lamellar laminae or a combination of epithelial changes (epithelium). Vascular changes (arteriosclerosis) were assessed in a subset of traditionally processed claws and were not identified; hence, statistical assessments were not performed.
Discussion
Determining causes and significance of overgrown claws in pigs is an ongoing area of research because of its perceived impact on foot health and lameness. Lameness resulting from overgrown claws in large commercial swine operations in the United States and other countries has become an important economic and welfare issue and can be detrimental because it results in increased and early cull rates. Routine claw trimming can provide some relief, but is labor-intensive. van Amstel et al10 determined that 39 mm was the length after which the claws were considered overgrown for gilts. On the basis of one author’s (SVA’s) experience trimming claws in adult sows, a length of 50 mm is recommended, as trimming shorter than that resulted in bleeding from the quick corium in some cases. Hence, for the purpose of this study, overgrown claws were identified as those measuring ≥ 50 mm and totaled 39 of the 72 claws (54%) available for examination.
Of the total of overgrown claws in this study, 67% were lateral rear claws. Lateral rear claw prevalence for lesions and overgrowth has been reported previously.4,6-9 The lateral claw is often more overgrown than the medial due to normal weight-bearing stress in sows in commercial operations,9 although it has also been reported that the medial hind claws are more overgrown than the lateral hind claws when the pig has laminitis.4 Unequal weight distribution between claws causes the rate of horn growth to exceed the rate of wear, and the imbalance in this weight distribution results in different lengths of claws, with the lateral claws being more severely affected.4 This disparity of claw size was also noted by authors of another study10where claw sizes were measured over a period of 55 days; the average size of the lateral rear claws was 1266 mm2, whereas that of the medial rear claws was 959 mm2. Once size disparity exists, it can produce abnormal conformation or postural and stride abnormalities that may cause ongoing lameness.10
The lack of definitive gross lesions (distal phalanx rotation) or histologic lesions, ie, inflammation, arteriosclerosis, or epithelial changes within the laminae, suggests that overgrown claws in this group of pigs are likely due to a variety of other factors, including management, flooring, uneven weight bearing, or diet. Overgrown claws in weaned pigs have been associated with damaged, slippery, or rough floor surfaces, suggesting that, for weaned pigs, the quality of the flooring may be the most important aspect of housing.2 The use of mats as a floor covering has also been associated with overgrown claws, but the documented overgrowth did not correlate to clinical lameness in that study.12 That result is similar to the absence of lameness despite significant overgrowth in many of the animals in this study. Nonetheless, ambulation may be altered by such overgrowth, and the need for corrective trimming may be warranted.
Pigs in this study were mature sows that had been housed at the same facility for one or more parities. During the breeding and gestation periods, all sows were housed on fully slatted concrete floors, and during the periparturient and lactation periods, they were housed in farrowing stalls with cast-iron slatted flooring. Hence, it is difficult to determine how much effect, if any, housing environment had on any recognized lesions, as all pigs were handled similarly.
While claw overgrowth can be seen in instances of laminitis,4 the phalanx rotation typically associated with laminitis was not identified, nor were histologic lesions involving vessels, as might be expected in more chronic cases. Histologic assessment of inflammation of the laminae (laminitis) and laminar epithelial changes were not consistently seen in the overgrown claws examined in this case series.
One of the goals of the study was to determine the incidence of histologic lesions, both in the epithelium of the laminae and the dermis of the claw, from a range of rear claws, many of which were overgrown. Main categories of assessment included epithelial changes, such as vacuolation, apoptosis, and necrosis; and infiltration of a variety of inflammatory cells, including lymphocytes, plasma cells, eosinophils, and neutrophils. In some samples, while inflammation was present, it was often non-specific and mild. Overall inflammation and inflammation subtypes were seen more in overgrown claws (14) than in normal claws (12), but the total numbers were insufficient to obtain statistical significance. The range of inflammatory infiltrates was narrow and did not allow a grading scale based on severity to be determined. The infiltrates varied in type, but there was no consistent inflammatory cell predominance. The presence of eosinophils is often associated with allergy-hypersensitivity or parasitism in most species, but the pig has a propensity to respond with eosinophils non-specifically within tissues.13 Typically, neutrophils are seen in acute active inflammation. The presence of lymphocytes and plasma cells often indicates more chronic, nonspecific processes.
Epithelial changes indicative of acute or ongoing damage were not a prominent finding among study pigs. Epithelial changes were seen more in overgrown claws (nine) than in normal claws (three), but again, the total numbers were too small for comparison, and statistical significance could not be achieved. When present, epithelial changes demonstrated some histologic characteristics similar to those reported in acute laminitis in other species, but were mild and rarely included all characteristics. Porcine claw lesions did not appear to have a common pathogenesis or histologic lesion, and inflammation and epithelial changes were infrequent overall. In chronic cases, it is difficult to know whether the changes seen are cause or effect.
This study was intended to generate hypotheses rather than test a specific hypothesis, and while this represents a preliminary study, small sample size may have prevented clear associations. It would be important to examine a larger number of overgrown and normal claws and assess the incidence and severity of histologic lesions in an effort to clarify the common etiology for porcine claw overgrowth. Studies for claw lesions that include larger numbers of pigs from different sources representing a variety of housing conditions and feeding management are necessary to characterize the histologic lesions associated with overgrowth, determine the role of laminitis, and determine contributing causes of overgrowth development, including the effect of diet.
Implications
• Claw overgrowth may not be associated with significant inflammation or epithelial changes in the laminae.
• Additional studies are warranted to determine whether histologic changes similar to laminitis are seen in overgrown claws.
• Additional studies are warranted to determine underlying causes of overgrown claws.
Acknowledgements
Special thanks to Sharon Schlosshan for technical assistance and Misty Bailey for editorial assistance.
Conflict of interest
None reported.
Disclaimer
Scientific manuscripts published in the Journal of Swine Health and Production are peer reviewed. However, information on medications, feed, and management techniques may be specific to the research or commercial situation presented in the manuscript. It is the responsibility of the reader to use information responsibly and in accordance with the rules and regulations governing research or the practice of veterinary medicine in their country or region.
References
1. Anil SS, Anil L, Deen J. Evaluation of patterns of removal and associations among culling because of lameness and sow productivity traits in swine breeding herds. JAVMA. 2005;226:956–961.
2. Nilsson SA. Laminitis in pigs. Nord Vet Med. 1964;16:128–139.
3. Maclean CW. Acute laminitis in sows. Vet Rec. 1966;83:71–75.
4. Guimaraes AMS, Althaus LK, Tullio DM, DeConto I, Silva AW, Ferrari MV, Biondi AW, Alberton GC. Laminitis in culled sows from commercial farms of southern Brazil. Arch Vet Sci. 2008;13:140–144.
5. Bergsten C. Causes, risk factors and prevention of laminitis and related claw lesions. Acta Vet Scand. 2003;44(S1):S157–S166.
6. Jorgensen B. Osteochondrosis/osteoarthrosis and claw disorders in sows, associated with leg weakness. Acta Scand. 2000;41:123–138.
7. Knauer M, Stalder KM, Karriker L, Bass TJ, Johnson C, Serenius T, Layman L, McKean JD. A descriptive survey of lesions from cull sows harvested at two Midwestern US facilities. Prev Vet Med. 2007;82:198–212.
8. Lahrmann KH, Steinberg C, Dahms S, Heller P. Prevalence of herd specific factors and limb disorders, and their associations in intensive swine production. Berliner und Munchener Tierarztliche Wochenschrift (Berlin). 2003;116:67–73.
9. Gjein H, Larssen RB. Housing of pregnant sows in loose and confined systems – a field study. 2. Claw lesions: morphology, prevalence, location and relation to age. Acta Vet Scand. 1995;36:433–442.
10. van Amstel S, Doherty T. Claw horn growth and wear rates, toe length and claw size in commercial pigs: A pilot study. J Swine Health Prod. 2010;18:239–243.
11. Cassera FL. Nuclear and cytoplasmic staining. In: Cassera FL. Histotechnology: A Self-Instructional Text. 2nd ed. Chicago, Illinois: American College of Clinical Pathology Press; 1997:83–109.
12. Calderón Díaz JA, Fahey AG, Kilbride AL, Green LE, Boyle LA. Longitudinal study of the effect of rubber slat mats on locomotory ability, body, limb and claw lesions, and dirtiness of group housed sows. J Anim Sci. 2013;91:3940–3954.
13. Maxie MG, Youssef S. Nervous system. In: Maxie MG, ed. Jubb, Kennedy, and Palmer’s Pathology of Domestic Animals. 5th ed. New York, New York: Saunders Elsevier; 2007:281–457.