Original research
Peer reviewed
Genetic interaction between porcine reproductive and respiratory syndrome virus (PRRSV) strains in cell culture and in animals
Michael P. Murtaugh, PhD; Shishan Yuan, PhD; Eric A. Nelson, PhD; and Kay S. Faaberg, PhD
MPM, SY, KSF: Department of Veterinary PathoBiology, University of Minnesota, 1971 Commonwealth Avenue, St Paul, MN 55108; EAN: Animal Disease Research and Diagnostic Laboratory, South Dakota State University, Brookings, SD 57007. Address correspondence to Michael Murtaugh, Department of Veterinary PathoBiology, University of Minnesota, 1971 Commonwealth Avenue, St Paul, MN 55108; Tel: 612-625-6735; Fax: 612-625-5203; E-mail: murta001@umn.edu.
Murtaugh MP, Yuan S, Nelson EA, et al. Genetic interaction between porcine reproductive and respiratory syndrome virus (PRRSV) strains in cell culture and in animals. J Swine Health Prod. 2002;10(1):15-21. Also available as a PDF
Summary
Objective: The need to obtain protective immunity and full reduction of disease associated with porcine reproductive and respiratory syndrome virus (PRRSV) infections has encouraged the use of multiple vaccine types to possibly obtain broader protection against genotypically variable PRRSV isolates. This strategy introduces potential risks of genetic rearrangements that might accelerate the natural rate of genetic change, facilitating the emergence of new PRRSV strains. Our goal was to determine if recombination was occurring between vaccine strains in porcine cells and in vivo.
Methods: Attenuated vaccine viruses were grown together in cultured cells or administered simultaneously to pigs. Cell culture fluids and pig sera were evaluated for recombinant viruses.
Results: Genetic recombination occurred between attenuated vaccine strains of PRRSV grown together in porcine alveolar macrophages and in simian MA-104 cell cultures. However, animals inoculated with both strains simultaneously did not provide evidence of viral recombination between vaccine strains in vivo.
Implications: Practices which favor co-infection of cells with different virus strains might accelerate the rate of genetic change in PRRSV due to recombination. However, the probability of recombination of vaccine strains in animals appears to be low and recombinants appear to grow poorly. Mixing of vaccine strains for simultaneous administration in swine must be considered with the knowledge that recombination could occur. Vaccinating infected herds also introduces the possibility of recombination between vaccine and field strains. The likelihood of producing viable recombinant vaccine viruses is low, since they did not persist in cell culture and were not observed in vivo.
Keywords: swine, porcine reproductive
and respiratory virus, macrophage, recombination, vaccine
swine, porcine reproductive
and respiratory virus, macrophage, recombination, vaccine
Received: November 15, 2000
Accepted: June 6, 2001
Porcine reproductive and respiratory syndrome (PRRS), a disease of swine caused by the RNA virus, PRRSV, is now the most important disease of swine worldwide. The disease manifestations, which vary greatly, include abortions and weak-born piglets in sows, interstitial pneumonia in piglets, and abnormal sperm in boars.1-3 Even as more recent manifestations arise, for example, neurovirulence and the "abortion-storm" syndrome,4-6 others, such as blue ear disease, are rarely reported.
The variation in clinical signs and apparent emergence of new syndromes are due in part to changes in the PRRSV genome. Therefore, the appearance of PRRSV variants with new genetic compositions increases the likelihood of increased variation in the virus and in the disease it causes, including disease characteristics, modes and ease of transmission, changes in cell permissiveness and species specificity, capability of persistence, and escape from vaccinal immunity.
Vaccination with modified live vaccines is an important method of control for PRRS in the United States today. The need to obtain protective immunity and full reduction of disease associated with field infections has encouraged the use of multiple vaccine products to obtain broader protection against genotypically variable PRRSV isolates. While this approach is valid in theory, it introduces potential risks by providing an opportunity for different variants of PRRSV to infect the same cell and to exchange genetic information during viral replication. A consequence is the possible emergence of new virus strains with broad scale genetic differences compared to the original vaccine viruses. Large genetic re-arrangements have the potential to greatly accelerate the rate of genetic change compared to either simple mutation or to the process of recombination that may occur between identical genomes present in cells infected with a single viral strain.
The objective of this study was to determine whether genetic recombination occurs between attenuated vaccine viruses grown together in cell culture, either in simian MA-104 cells or porcine alveolar macrophages, or in animals.
Materials and methods
Preparation of virus stocks
The vaccine strains of PRRSV used in this study were RespPRRS (Boehringer Ingelheim Vetmedica, Inc, St Joseph, Missouri) and Prime Pac PRRS (Schering Plough Animal Health, Kenilworth, New Jersey). Vaccine vials were reconstituted with the provided diluent according to the manufacturers' directions. Virus was inoculated onto MA-104 cells grown in Eagle's modified essential medium (EMEM; Life Technologies, Gaithersburg, Maryland) containing 10% fetal bovine serum, at a multiplicity of infection (MOI) of 0.1. Multiplicity of infection is the virus:cell ratio at the time a virus suspension is added to a culture of cells. The MA-104 cell line was established from rhesus monkey embryonic kidney tissue and is available from the American Type Culture Collection (Rockville, Maryland) and many veterinary and human diagnostic laboratories. When cytopathic effect (CPE) was greater than 80%, cell culture supernatants were harvested, clarified by centrifugation, and stored at -80°C. The titer of virus stocks was 1.3x107 median tissue culture infective doses (TCID50) per mL.
Cell culture experiments
The MA-104 cell line was co-infected with RespPRRS and Prime
Pac PRRS vaccine strains at a combined MOI of 1.0, using a 1:3
ratio of RespPRRS to Prime Pac PRRS, and incubated until CPE was
maximal 3 days after infection. First passage supernatants containing
progeny virus were harvested and used to reinfect fresh MA-104
cells at an MOI of 1.0; this process was repeated for a total
of four passages. Aliquots of each passage were frozen at
-80°C. The experiment was replicated three times.
Alveolar macrophages were harvested by lung lavage from healthy pigs at approximately 6 weeks of age, plated overnight in culture media, and inoculated with individual vaccine virus strains at an MOI of 0.1. Supernatants were harvested when CPE was greater than 80%. These macrophage-adapted viral stocks were titered on MA-104 cells and used for experiments involving growth on macrophages. Alveolar macrophages were inoculated with each virus alone or with both viruses together at a relative MOI ranging from 1 to 10 of Prime Pac PRRS:RespPRRS, including addition of Prime Pac PRRS 2 hours or 4 days before addition of RespPRRS. The experiment of co-culturing two vaccine viruses on alveolar macrophages was performed six times.
Macrophage and MA-104 cell supernatants were subjected to polymerase chain reaction (PCR) testing to identify strain-specific nucleotide sequences that would identify parental and recombinant viruses.
Animal inoculation
Eight 4-week-old piglets were obtained from a PRRSV-naive herd, and each piglet was assigned to one of three groups: Prime Pac PRRS and RespPRRS combined (n=4), Prime Pac PRRS alone (n=2), or RespPRRS alone (n=2). Each group of piglets was maintained in a separate isolation room and allowed to acclimate for 7 days prior to inoculation. To avoid cross-contamination, caretakers and technicians showered before entering each room when it was impossible for different personnel to deal with each group of pigs. The room containing dual-vaccinated animals was always entered last. Blood samples were collected from all animals the day prior to inoculation, and sera were tested by the HerdChek PRRS ELISA (IDEXX Laboratories, Westbrook, Maine) to verify PRRSV-seronegative status.
For pigs receiving only one vaccine strain, vaccines were diluted according to label directions in the provided diluent. One label dose (2 mL) was administered intramuscularly and two label doses were administered intranasally upon inspiration, with 2 mL in each nare, using syringes equipped with aspirator tips. Thus, each pig received three label doses. For dually inoculated pigs, vaccines were resuspended in one-half the recommended volume of diluent and equal volumes of the two vaccines were combined. The combined vaccine preparation was then administered exactly as described above so that each pig received three label doses of each vaccine. Blood samples were collected from all animals 1, 2, 4, 7, 9, 13, and 17 days after inoculation, and serum samples were immediately frozen at -80°C for storage until evaluation by PCR.
PCR testing
Viral RNA was isolated from cell culture supernatants and serum using the QIAamp Viral RNA kit (Qiagen Inc, Valencia, California) for cDNA synthesis, as previously described.7 Polymerase chain reaction was used to amplify an 1182-base region from nucleotides 12886 to 14067 of RespPRRS strain virus (Genbank accession number AF066183) and Prime Pac PRRS strain virus (Genbank AF066384), corresponding to part of open reading frame (ORF) 3, all of ORF 4, and part of ORF 5, using primers which were specific for RespPRRS strain or for Prime Pac PRRS strain at the 5' and 3' ends of the sequence.8 At the 5' end, the strain-specific primers (Resp5 and Prime5) differed at eight of 23 bases, and at the 3' end, the strain-specific primers (Resp3 and Prime3) differed at six of 25 positions. Thus, RespPRRS strain was specifically detected using primers Resp5 and Resp3, and Prime Pac PRRS was specifically detected using primers Prime5 and Prime3. Recombinant progeny virus consisting of ResPRRS at the 5' end and Prime Pac PRRS at the 3' end was detected using primers Resp5 and Prime3, and recombinant virus consisting of Prime Pac PRRS at the 5' end and RespPRRS at the 3' end was detected using Prime5 and Resp3.
Nested PCR amplification of the ORF 7 region of PRRSV was performed as described.9
Controls for PCR testing
To establish conclusively that the PCR products obtained using primer pairs from different vaccine strains were actually from recombined progeny virus, samples were treated with ribonuclease A at a concentration of 10 µg per mL before RNA purification. Purified viral RNA was added to media supernatants at 10 µg per mL, repurified as described above, and analyzed by denaturing gel electrophoresis to determine whether the media supernatants contained ribonuclease activity that would degrade RNA that was not protected within viral particles. To determine whether recombinant products could be generated by enzyme switching from one template to another, RNA from the two vaccine viruses was mixed together before cDNA synthesis and PCR testing was performed. Lastly, media supernatants from cultures infected with either vaccine virus alone were tested using homologous primer pairs (Resp5 and Resp3, or Prime5 and Prime3) and using heterologous primer pairs to establish the specificity of the PCR reactions. This series of control experiments was performed one time.
To obtain additional information about the nature of recombination, the PCR products obtained with the Resp5 and Prime3 primers were cloned, and five colonies were selected and sequenced using standard methods.
To determine whether recombinant virus progeny were continually produced in successive passages or were able to compete with parental vaccine strains for growth on MA-104 cells, media supernatants from passages one through four were examined for evidence of recombinant viruses.
Diagnostic tests
Anti-PRRSV antibody response was determined by the HerdChek ELISA, and serum was tested for the presence of virus by nested PCR. Samples with S:P>0.4 were considered positive, and samples with S:P>0.10 were considered suggestive of positive. The DNA sequencing was performed by the Advanced Genetic Analysis Center at the University of Minnesota on an ABI377 automated DNA sequencer using a Taq DiDeoxy Terminator Cycle Sequencing kit (Applied Biosystems, Foster City, California). Sequence analysis was performed with DNASTAR, GCG, and EUGENE software.
Results
Effect of dual infection on recombination in MA-104 cell culture
Polymerase chain reaction amplification of MA-104 culture supernatants from the first co-infection passage using primer pairs specific for RespPRRS (Resp5 and Resp3) or Prime Pac PRRS (Prime5 and Prime3) gave readily visible product bands of 1182 bp on an agarose gel (Figure 1). In addition, PCR reactions containing primers Resp5 and Prime3, or Prime5 and Resp3, which would amplify only chimeric viral progeny, also gave product bands from first passage media supernatants.

Controls for PCR testing
Samples treated with ribonuclease A before RNA purification still produced PCR bands. Conversely, viral RNA added to media supernatants was degraded. Mixing RNA from both vaccine viruses together, before cDNA synthesis and PCR, gave rise to PCR products with matched primer sets specific for each virus, but not with mismatched sets specific for recombinant products. Lastly, media supernatants from cultures infected with either vaccine virus alone produced PCR products only with the homologous primer pair, not with the primer pair specific for the other vaccine virus nor with the two primer pairs specific for recombinant viral progeny.
All clones of PCR products obtained with the Resp5 and Prime3 primers appeared to be derived from independent recombination events, on the basis of the location in which template switching occurred (Figure 2). Furthermore, four of the recombinant clones were consistent with a single crossover in the region examined, while the fifth clone showed evidence of a triple crossover.
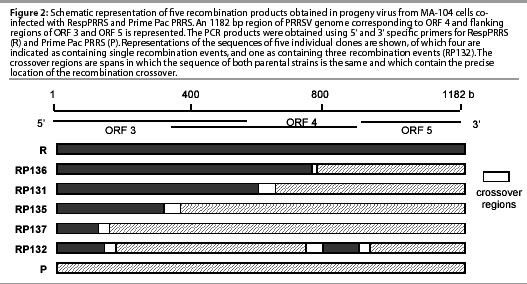
The PCR products from RespPRRS-specific primers were abundant at all passages and PCR products from the recombinant primer pair Resp5-Prime3 were readily apparent for three passages, and still detected at passage four, though at a low level (data not shown). The PCR products from the Prime5-Resp3 primer pair were present at a low but equivalent abundance for three passages and were not detected in passage four. Similarly, the Prime Pac PRRS-specific primer pair amplified DNA from the first three passages but not from the fourth.
Effect of dual infection on recombination in porcine macrophages
Infection of alveolar macrophages with either virus alone resulted in the production of progeny virus, but the amplified PCR products were more abundant from culture supernatants of macrophages infected with RespPRRS than from macrophages infected with Prime Pac PRRS (data not shown). Co-culture of the two viruses on macrophages under various conditions resulted in the detection of recombinant progeny virus. Infection of macrophages with Prime Pac PRRS virus for 4 days then co-culture with RespPRRS for 1 day produced recombinant virus in the culture supernatant (Figure 3, lane 2), as did co-culture for 5 days at an MOI ratio of 3 Prime Pac PRRS:1 RespPRRS (Figure 3, lane 3). The amount of recombinant virus was similar to the amount of Prime Pac PRRS progeny in this co-culture (lane 5). The yield of Prime Pac PRRS progeny viral RNA was substantially greater in the absence of RespPRRS, as shown by the comparison of lanes 5 and 6.

Although recombination occurred between the two PRRSV strains when grown on alveolar macrophages, its frequency was observed to be lower than on MA-104 cells. Infection of macrophages with a relative MOI ranging from 1 to 10 of Prime Pac PRRS:RespPRRS, including addition of Prime Pac PRRS 2 hours before addition of RespPRRS, consistently resulted in high levels of RespPRRS and low levels of Prime Pac PRRS (Figure 4). Recombinant progeny were observed in only one of six cases, and the amplification product was barely visible in the gel (Figure 4, lane 7).
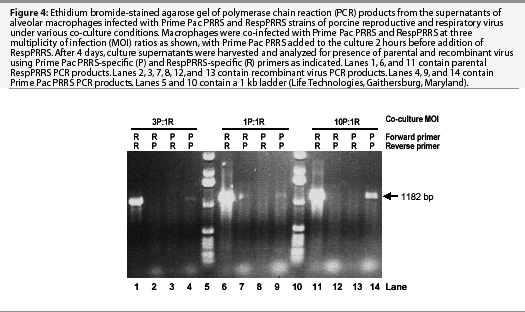
Effect of dual vaccination on recombination in vivo
Dual infection with the two attenuated virus strains resulted in viremia and seroconversion in 5-week-old piglets. Viremia was demonstrated by nested PCR analysis in serum of all animals on Days 3 and 7. By Day 17, ELISA tests showed seroconversion in nearly all animals, with the highest serological response in animals inoculated with RespPRRS strain alone (Table 1). Strain-specific PCR detected Prime Pac PRRS virus in serum in one of four dually infected animals and in one of two singly infected animals 24 hours after infection. Neither the RespPRRS strain nor recombinant viruses were detected by strain-specific PCR in any animals 24 hours after infection, and all animals were virus-negative by strain-specific PCR at all time points beyond 24 hours to the end of the experiment.

Discussion
Porcine respiratory and reproduction syndrome was first described in the mid-1980's as mystery swine disease, and was widespread in North America and Europe at the time PRRSV was described by Wensvoort et al1 in Europe and by Collins et al2 in North America. The origins of PRRSV are unclear, since North American and European forms of the virus are somewhat distinct and are equivalently distant from lactate dehydrogenase-elevating virus (LDV) of mice, the closest relative of PRRSV.10 Moreover, the earliest evidence of PRRSV infection in swine dates from 1979 in Canada and 1985 in the United States.11,12 Thus, it appears that PRRSV has emerged recently both as a new virus and as the cause of a new disease of swine.
Genetic analysis and clinical observations suggest that the virus is changing rapidly. The mechanisms of change involve both mutation and genetic recombination.13 Recombination is particularly significant since it can occur at a high rate in coronaviruses, which are in the same order, the Nidovirales, as PRRSV by virtue of similar genetic organization, replication strategy, and methods of gene transcription and protein translation.14 Thus, information about coronaviruses might be applicable to PRRSV. Recombination also produces large rearrangements of groups of genes which may give rise to new recombinant viral forms.
Our results show that recombination occurs in macrophages as well as in MA-104 cells. Several controls were performed to prove that the PCR products obtained using primer pairs from different vaccine strains were actually from recombined progeny virus. Samples treated with ribonuclease A before RNA purification still produced PCR bands, demonstrating that the RNA was in intact virions and not released from lysed cells. Conversely, viral RNA added to media supernatants was degraded, showing that PCR products were derived only from RNA packaged into progeny recombinant viral particles. Mixing RNA from both vaccine viruses together before cDNA synthesis and PCR gave rise to PCR products only with matched primer sets specific for each virus, but not with mismatched sets specific for recombinant products, indicating that recombinant products were not generated by reverse transcriptase or Taq polymerase switching from one template to another.
The frequency of recombination, particularly in macrophages, was low and consistent with a lower replication rate for Prime Pac PRRS virus in cells dually infected with the RespPRRS strain of PRRSV. Also, recombinant vaccine virus progeny competed poorly with parental types for growth in cells, as they disappeared from culture supernatants within four passages. These results suggest that attenuation of growth is the principal outcome of recombination. Thus, chimeric progeny compete poorly with the parental strains for growth, both in MA-104 cells and macrophages.
Further evidence in support of this conclusion is that recombination was not observed in vivo. Growth of both vaccine viruses in pigs was confirmed by nested PCR detection of viral nucleic acids in serum and by seroconversion. The largely negative results obtained from strain-specific PCR, including no indication of recombinant viruses, suggest that the vaccine viruses grew poorly in vivo. Reduced viremia following administration of PRRSV vaccine strains compared to infection with field strains has been observed previously.15 Our observations and those of Christopher-Hennings et al15 suggest that attenuated vaccine viruses may grow poorly in pigs compared to wild type PRRSV.
We have identified field isolates of PRRSV which appear to have arisen by recombination, demonstrating that this genetic phenomenon occurs in PRRSV and can result in functional progeny which can compete successfully against existing strains.8,13 Whether the recombinant strains are attenuated relative to the parental field strains is not known. In poultry, new strains of infectious bronchitis virus, an avian coronavirus, have emerged by recombination and show differences in pathogenicity relative to the parental strains.16-19
We have now shown directly that PRRS vaccine strains recombine readily when grown together in MA-104 cell culture, but less frequently in porcine macrophages. Recombination presumably is facilitated by co-infection, since it allows for the simultaneous presence of different virus types in the same cell. The viral RNA polymerase then can use different replication templates to produce chimeric RNA genomes which are a mixture of the initial types. The administration of modified-live vaccine strains to pigs in infected herds also introduces the possibility of recombination between vaccine and field strains of PRRSV. The probability of recombination between vaccine and field strains cannot be estimated, but may be low by comparison to the in vitro experiment reported here. To date, we have analyzed more than 1000 field isolates by DNA sequencing of ORF 5 and have observed no evidence of recombination between vaccine and field strains of PRRSV (K.S. Faaberg and M.P. Murtaugh, unpublished data, 2001).
The Prime Pac PRRS vaccine used in this study is not presently available in the United States. Nevertheless, the implications of our findings, ie, that vaccine viruses have the capacity to recombine with each other, are relevant to current swine health practice since at least two live attenuated vaccines, RespPRRS and Ingelvac ATP (Boehringer Ingelheim Vetmedica), are commercially available. Moreover, in vivo recombination has been reported between attenuated PRRSV strains after simultaneous exposure.20 Even if simultaneous administration of two vaccines is not a common industry practice, the rotation of two vaccines to achieve better strain cross-protection21 may increase the number of PRRSV strains circulating in a herd and thus increase the possibility of recombination between vaccinal strains or between vaccine and field variants. Our evidence indicates that the rate of recombina- tion between vaccinal strains in vivo is undetectably low, but in swine herds with large numbers of animals there is an increased likelihood that rare events might occur. Vaccination of infected herds also would be expected to increase the number and the genetic diversity of PRRSV variants within a herd, and accordingly increase the possibility of recombination. Whether recombinant viruses have novel phenotypes cannot be predicted. In our experiments, recombinant forms that appeared in cell culture were unable to compete effectively with the parental vaccine forms and disappeared within a few generations. By contrast, recombination of field isolates was common and recombinants were able to predominate in the circulation of infected pigs under the conditions reported,20 indicating that the factors which affect the frequency and outcome of recombination are not fully known.
Implications
- Mixing different vaccine strains of PRRSV in one syringe for vaccination is not advisable since it may favor recombination due to the presence of a high concentration of virus.
- The principal outcome of recombination between vaccine strains is attenuation of growth of the recombinant virus.
- The likelihood of producing viable recombinant vaccine viruses is low, since, under the conditions of this study, they were not able to persist in cell culture and were not observed in vivo.
Acknowledgements
Partial funding for this study was provided by PIC, Inc, Franklin, Kentucky, grant #99-071 from the Minnesota Pork Producers Association, and Boehringer Ingelheim Vetmedica, Inc, St Joseph, Missouri.
References -- refereed
1. Wensvoort G, Terpstra C, Pol JMA, ter Laak EA, Bloemradd M, de Kluyver EP, Kragten C, van Buiten L, den Besten A, Wagenaar F, Broekhuijsen JM, Moonen PLJM, Zetstra T, de Boer EA, Tibben HJ, de Jong MF, van't Veld P, Groenland GJR, van Gennep JA, Voets MT, Verheijden JHM, Braamskamp J. Mystery swine disease in the Netherlands: the isolation of Lelystad virus. Vet Quart. 1991;13:121-130.
2. Collins JE, Benfield DA, Christianson WT, Harris L, Hennings JC, Shaw DP, Goyal SM, McCullough S, Morrison RB, Joo HS, Gorcyca DE, Chladek DW. Isolation of swine infertility and respiratory syndrome virus (isolate ATCC VR-2332) in North America and experimental reproduction of the disease in gnotobiotic pigs. J Vet Diagn Invest. 1992;4:117-126.
3. Christopher-Hennings J, Nelson EA, Hines RJ, Nelson JK, Swenson SL, Zimmerman JJ, Chase CL, Yaeger MJ, Benfield DA. Persistence of porcine reproductive and respiratory syndrome virus in serum and semen of adult boars. J Vet Diagn Invest. 1995;7:456-464.
4. Thanawongnuwech R, Halbur PG, Andrews JJ. Immunohistochemical detection of porcine reproductive and respiratory syndrome virus antigen in neurovascular lesions. J Vet Diagn Invest. 1997;9:334-337.
5. Rossow KD, Shivers JL, Yeske PE, Polson DD, Rowland RR, Lawson SR, Murtaugh MP, Nelson EA, Collins JE. Porcine reproductive and respiratory syndrome virus infection in neonatal pigs characterized by marked neurovirulence. Vet Rec. 1999;144:444-448.
7. Nelsen CJ, Murtaugh MP, Faaberg KS. Porcine reproductive and respiratory syndrome virus comparison: divergent evolution on two continents. J Virol. 1999;73:270-280.
8. Yuan S, Nelsen CJ, Murtaugh MP, Schmitt BJ, Faaberg KS. Recombination between North American strains of porcine reproductive and respiratory syndrome virus. Virus Res. 1999;61:87-98.
9. Christopher-Hennings J, Nelson EA, Nelson JK, Hines RJ, Swenson SL, Hill HT, Zimmerman JJ, Katz JB, Yaeger MJ, Chase CC. Detection of porcine reproductive and respiratory syndrome virus in boar semen by PCR. J Clin Micro. 1995;33:1730-1734.
10. Murtaugh MP, Elam MR, Kakach LT. Comparison of the structural protein coding sequences of the VR-2332 and Lelystad virus strains of the PRRS virus. Arch Virol. 1995;140:1451-1460.
11. Carman S, Sanford SE, Dea S. Assessment of seropositivity to porcine reproductive and respiratory syndrome (PRRS) virus in swine herds in Ontario: 1978 to 1982. Can Vet J. 1995;36:776-777.
12. Zimmerman JJ, Yoon K-J, Wills RW, Swenson SL. General overview of PRRSV: a perspective from the United States. Vet Microbiol. 1997;55:187-196.
13. Kapur V, Elam MR, Pawlovich TM, Murtaugh MP. Genetic variation in PRRS virus isolates in the midwestern United States. J Gen Virol. 1996;77:1271-1276.
14. Lai MMC. RNA recombination in animal and plant viruses. Microbiol Rev. 1992;56:61-79.
15. Christopher-Hennings J, Nelson EA, Nelson JK, Benfield DA. Effects of a modified-live virus vaccine against porcine reproductive and respiratory syndrome in boars. Am J Vet Res. 1997;58:40-45.
16. Cavanagh D, Davis P, Cook J, Li D. Molecular basis of the variation exhibited by avian infectious bronchitis coronavirus (IBV). Adv Exp Med Biol. 1990;276:369-372.
17. Wang L, Junker D, Collisson EW. Evidence of natural recombination within the S1 gene of infectious bronchitis virus. Virology. 1993;192:710-716.
18. Wang L, Junker D, Hock L, Ebiary E, Collisson EW. Evolutionary implications of genetic variations in the S1 gene of infectious bronchitis virus. Virus Res. 1994;34:327-338.
19. Jia W, Karaca K, Parrish CR, Naki SA. A novel variant of avian infectious bronchitis virus resulting from recombination among three different strains. Arch Virol. 1995;140:259-271.
References -- non refereed
6. Halbur PG, Bush E. Update on abortion storms and sow mortality. Swine Health Prod. 1997;5:73.
20. Mengeling WL, Clouser DF, Vorwald AC, Lager, KM. Genetic recombination among strains of porcine reproductive and respiratory syndrome virus. Proc Conf Res Workers Anim Dis. 2000;81:134.
21. Loula T. What's new with PRRS on commercial farms? Proc Allen D Leman Swine Conf. 1998;25:172-173.
