,Original research |
Peer reviewed |
Intradermal vaccination for Mycoplasma hyopneumoniae
Vacunación intracutánea contra Mycoplasma hyopneumoniae
Vaccination contre le Mycoplasma hyopneumoniae
Gary F. Jones, DVM, PhD; Vicki Rapp-Gabrielson, PhD; Rebecca Wilke, MS; Eileen L. Thacker, DVM, PhD, Diplomate ACVM; Brad J. Thacker, DVM, PhD, Diplomate ABVP; Linda Gergen, MS; Diane Sweeney, PhD; Terri Wasmoen, PhD
GFJ, VR-G, RW, LG, DS, TW: Schering-Plough Animal Health, Elkhorn, Nebrask; ELT, BJT: Iowa State University, College of Veterinary Medicine, Ames, Iowa; Corresponding author: Dr G. F. Jones, 21401 West Center Road, Elkhorn, NE 68022; Tel: 402-289-6115; Fax: 402-289-6082; E-mail: gary.jones@spcorp.com
Cite as: Jones GF, Rapp-Gabrielson V, Wilke R, et al. Intradermal vaccination for Mycoplasma hyopneumoniae. J Swine Health Prod. 2005;13(1):19-27.
Also available as a PDF.
SummaryObjectives: To evaluate the ability of needleless intradermal (ID) vaccines to induce protection against clinical disease and to compare the serological response of pigs to intramuscular (IM) and ID vaccination with Mycoplasma hyopneumoniae bacterins. Materials and methods: In each study, pigs seronegative for M hyopneumoniae were vaccinated either once or twice with M hyo-pneumoniae bacterins. The serological response to ID and IM vaccination was evaluated by the DAKO M hyopneumoniae blocking ELISA, and ID vaccine efficacy was tested by experimental respiratory challenge with a heterologous strain of virulent M hyopneumoniae. Results: Injection site reactions were minimal for all vaccines. Pigs vaccinated ID, either by needle or needleless injector, had significantly higher M hyopneumoniae antibody titers than did pigs vaccinated by IM injection. Pigs vaccinated by needleless ID injection had lower lung lesion scores and higher IgA and IgG titers in bronchoalveolar lavage fluid than did nonvaccinates following challenge. Protective immunity was established by 21 days postvaccination after a single ID dose of vaccine. Intradermal vaccination of swine for M hyopneumoniae by needleless injector was safe and efficacious, and did not require shaving or other preparation of the skin. Implications: Intradermal vaccination of swine with needleless injectors is feasible if vaccines are formulated for small doses (eg, 0.2 mL). | ResumenObjetivos: Evaluar la habilidad de las vacunas intracutáneas (ID por sus siglas en inglés), aplicadas sin aguja, para inducir protección contra la enfermedad clínica y comparar la respuesta serológica de los cerdos a la vacunación intramuscular (IM por sus siglas en inglés) y a la vacunación ID con bacterinas contra Mycoplasma hyopneumoniae. Materiales y Métodos: En cada investigación, cerdos seronegativos a M hyopneumoniae fueron vacunados una o dos veces con bacterinas contra Mycoplasma hyopneumoniae. La respuesta serológica a la vacunación ID e IM fue evaluada con la prueba de ELISA de bloqueo DAKO para M hyopneumoniae, y la eficacia de la vacunación ID fue probada con un reto experimental por vía respiratoria con una cepa virulenta heteróloga de M hyopneumoniae. Resultados: Las reacciones in situ a la inyección fueron mínimas para todas las vacunas. Los cerdos vacunados ID con aguja o con inyector sin aguja, presentaron títulos de anticuerpos contra M hyopneumoniae significativamente más altos que los que tuvieron los cerdos vacunados con la inyección IM. Después del reto, los cerdos vacunados con la inyección ID sin aguja tuvieron un menor número de lesiones de pulmón y títulos más altos de IgA e IgG en el fluido de lavado bronquioalveolar que los cerdos no vacunados. La inmunidad protectora se estableció 21 días después de una dosis única de la vacuna ID. La vacunación ID de cerdos contra M hyopneumoniae con inyector sin aguja fue segura y eficaz y no requirió rasurado u otra preparación de la piel. Implicaciones: La vacunación intracutánea de cerdos con inyectores sin aguja es posible si las vacunas se formulan para dosis pequeñas (por ejemplo, 0.2 mL). | ResuméObjectifs: Évaluer la capacité des vaccines intradermel (ID par ses initiales en anglais) sans aiguille, pour causer la protection contre le maladie clinique et pour comparer la réponse sérologique du porc à la vaccination intramusculaire (IM par ses initiales en anglais) et ID avec les vaccines de Mycoplasma hyopneumoniae. Matériaux et méthodes: Dans chaque recherche, les porcs séronégative au M hyopneumoniae ont été vaccinés un ou deux fois avec les vaccines de M hyopneumoniae. La réponse sérologique à la vaccination ID et IM a été évaluée par la ELISA bloquant DAKO par M hyopneumoniae, et la efficacité de la vaccine ID a été épreuvé par le défi expérimental respiratoire avec une souche virulente hétérologue de M hyopneumoniae. Résultats: Les réactions sur le site de la injection ont été minimales pour toutes les vaccines. Les porcs vaccinés ID, avec l'aiguille ou avec le injecteur sans aiguille, ont eu titres significativement plus haut de anticorps de M hyopneumoniae que les porcs vaccinés avec la injection IM. Après le défi, les porcs vaccinés avec la injection ID sans aiguille ont eu un plus bas nombre de lésions pulmonaires et plus haut titres de IgA et IgG dans le fluide de lavage bronchoalveolar que les porcs que n'ont pas été vaccinés. L'immunité protectrice a été établie aprés 21 jours d'une seule dose de la vaccine ID. La vaccination ID de porcs contre le M hyopneumoniae avec injecteur sans aiguille a été sauf et afficace, et n'a pas eu besoin de rasage ou des autres préparation de la peau. Implications: La vaccination ID de porcs avec injecteurs sans aiguille est réalisable si les vaccines sont formulées pour petites doses (par exemple 0.2mL). |
Keywords: swine, intradermal
vaccination, Mycoplasma hyopneumoniae
Search the AASV web site
for pages with similar keywords.
Received: February
2, 2004
Accepted: June
22, 2004
Mycoplasma hyopneumoniae, the causative agent of enzootic pneumonia of swine, is one of the most common pathogens isolated in diagnostic laboratories from swine with respiratory disease.1 Vaccination has proven to be an important and cost-effective method of reducing the incidence of disease due to M hyopneumoniae, and may become more important as bacterial resistance to antibiotic therapy and public sensitivity to the use of antibiotics in food animals increases.2,3
Economically important diseases can be transmitted by repeated use of vaccination needles,4 and broken needles in pork are a serious industry problem.5 Needleless injection devices capable of vaccinating animals are commercially available and would preclude the use of needles and allow intradermal (ID) or deeper injections.6,7 It is postulated that protective immunity to M hyopneumoniae requires cell-mediated and mucosal components.8,9 Intradermal vaccination has been reported to significantly increase IgG serum titers of vaccinated animals6 and elicit both cell-mediated7 and mucosal immune responses.10 Dendritic cells of myloid lineage that localize in the basal and suprabasal layers of the epidermis uniquely express CD1, a family of glycoproteins that present nonpeptide lipid antigens to T cells.11,12 Activation of these cells is important in inducing an adaptive immune response to antigens. Intradermal vaccination may enhance the uptake of M hyopneu-moniae antigens by dendritic cells and thus increase the immune response induced by vaccination. In addition, M hyopneumoniae bacterins may be good candidates for ID vaccination, as the outer membranes of Mycoplasma species are high in lipids.
The objectives of the studies presented here were to evaluate the feasibility of ID vaccination of swine for M hyopneumoniae using needleless injection devices. Serum antibody titers to M hyopneumoniae and protection against experimental challenge with virulent M hyopneumoniae were used to evaluate immune responses to ID vaccination with M hyopneumoniae bacterins formulated for ID delivery.
Materials and methods
Bacterins
Four bacterins were formulated for use in Studies One and Three, and three were formulated for Study Two. The bacterins used in each study were formulated using the same lot of antigen with an oil-in-water adjuvant (Emunade; Schering-Plough Animal Health, Union, New Jersey), aqueous adjuvant B (Noveon, Cleveland, Ohio), or aqueous adjuvant C (Superfos Biosector, Vedbaek, Denmark). The amount of antigen per dose of bacterin was constant within each study, except where indicated, while the volume per dose was varied to accommodate the injection methods and devices used (Table 1).
Vaccinations
All vaccinations were administered in the neck, with the booster vaccinations given on the opposite side as the initial vaccination. Pigs were vaccinated in each study on Day 0, and pigs given a second vaccination were revaccinated on Day 14 or 15. Intramuscular injections were given with an 18-gauge, 1-inch needle. Intradermal injections were given using a 26-gauge, 3/8-inch needle or a spring-powered needleless injector (Dermo-Jet and Vaccijet Model 02, Robbins Instruments, Inc, Chatham, New Jersey). The Dermo-Jet was used in Study One and the Vaccijet 02 was used in Studies Two and Three. The injectors each delivered a set volume: the Dermo-Jet delivered 0.1 mL and the Vaccijet 02 delivered 0.2 mL. Injection pressure, and therefore injection depth and amount of fluid which penetrated the skin, was regulated by selection of spacers fitting the nozzle of the Dermo-Jet and by adjustments in the spring tension of the Vaccijet 02. Vaccinations given with the Dermo-Jet were administered as three closely spaced injections to assure that approximately 0.1 mL of bacterin penetrated the skin surface. Each vaccination given with the Vaccijet 02 was given as a single 0.2 mL injection.
Testing the needleless injectors
Sections of skin, including the subcutaneous fascia and underlying muscle, were collected at necropsy from the necks of pigs at 4 and 14 weeks of age. The hair was shaved from half of each skin sample and the samples were injected with fluid (0.7% Dextran Blue dye in water) by the injectors. Skin from 14-week-old pigs was tested with the Dermo-Jet at a single pressure setting (12 replicates), and skin from a 4-week-old pig was tested at two pressure settings of the Vaccijet 02 (six replicates per pressure setting). The amount of the fluid that penetrated the skin was estimated by weighing skin sections before and after injection, and after blotting residual fluid from the skin surface. The skin was dissected and the depth of injected dye measured.
Study animals, environment, and nutrition
Healthy crossbred pigs (Landrace x Yorkshire x Duroc cross) were purchased from a conventional herd that was seronegative and culture negative for M hyopneumoniae. Feed was appropriate for age and weight, and both water and feed were provided ad libitum. Antimicrobial medication was removed from the diet at least 10 days prior to experimental challenges. Pigs were housed in mechanically ventilated barns, with all treatment groups in a study housed in the same room. Pigs enrolled in challenge studies were weighed a day before vaccination, before challenge, and before necropsy. Bacterins or vaccines, other than the test M hyopneu-moniae bacterins, were not administered. Animals were used and cared for with appropriate regard for humane concerns.
Study design
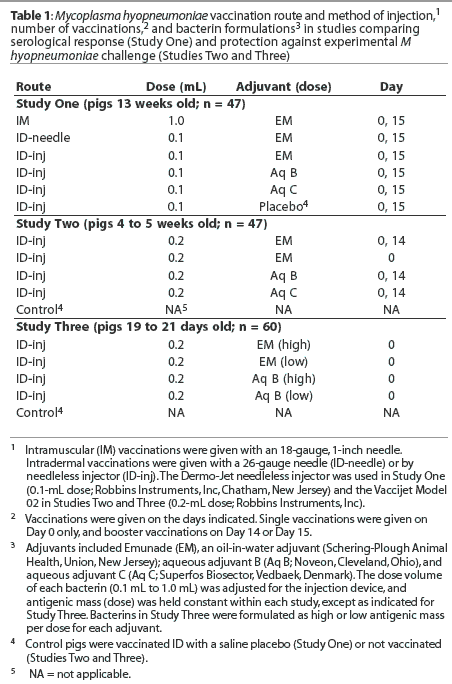
Study One: Serological response to IM and ID vaccination. Pigs 13 weeks old (n = 47) were blocked by sex and randomly allotted to six treatment groups (Table 1) by ear tag number (n = 7 to 8 per group), and housed in pens of two pigs each. The pigs were vaccinated on Day 0 and Day 15 with M hyopneumoniae bacterins formulated to contain approximately 55% of the minimum antigen dose of a bacterin licensed for IM vaccination (H. Jayappa, Schering-Plough Animal Health, unpublished data, 1999). Emunade-adjuvanted bacterins were administered to three groups by conventional IM injection, by ID injection given by needle, or by Dermo-Jet. The remaining groups were vaccinated ID by Dermo-Jet with bacterins formulated with aqueous adjuvants B or C, or with a saline placebo. Pigs were observed daily and examined for injection-site reactions for 36 days. Blood for serum was collected from the pigs before vaccination on Day 0 and on Days 14, 22, 29, and 36. Pigs vaccinated ID with adjuvanted M hyopneumoniae bacterins were necropsied on Day 36. Palpable injection site reactions were dissected, fixed, and submitted to the Veterinary Diagnostic Laboratory of Iowa State University (ISU-VDL, Ames, Iowa) for microscopic examination.
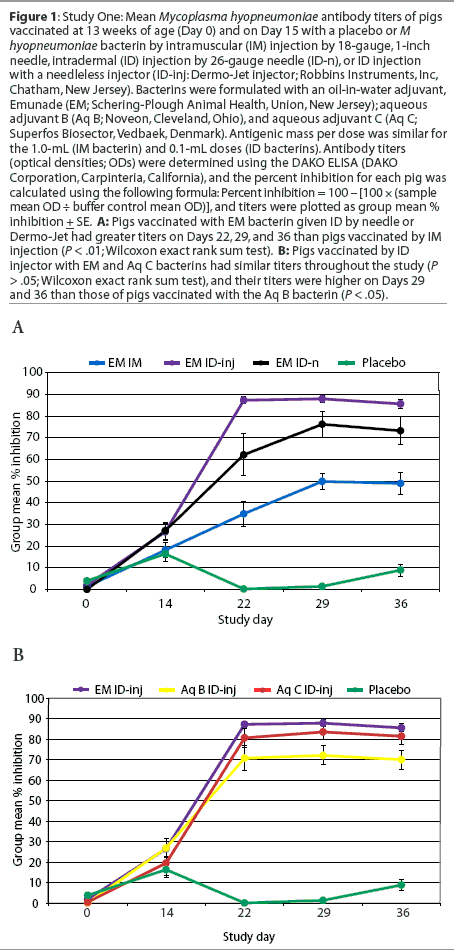
Study Two: Comparison of ID adjuvant formulations by experimental challenge. Pigs 4 to 5 weeks old (n = 47) were blocked by weight and litter and randomly allotted to a nonvaccinated group and four groups vaccinated once or twice ID by needleless injector (Table 1). Pigs of all groups were commingled during the challenge phase of the study. The bacterins used were formulated with a dose of 0.2 mL that contained approximately three times the amount of antigen per dose as the bacterins used in Study One. The pigs were experimentally challenged 20 days after the second vaccination with a derivative of M hyopneumoniae strain 11 as previously described.12 Briefly, each pig was given an intratracheal inoculation of 10 mL of a 1:100 dilution of tissue lung homogenate containing 107 color-changing units per ml (CCU: the reciprocal of the highest dilution of a culture that causes a red-to-yellow color change after 7 to 10 days incubation in medium containing phenol red); thus, the inoculating dose was 106 CCU of M hyopneumoniae per pig. Blood for serum was collected before vaccination (Day -4) and on Days 14, 21, 28, 34, and 59. The pigs were euthanized and necropsied, and the lungs scored, 28 days after challenge (Day 62). Portions of lung tissue from five pigs with typical lesions were selected by the prosector, without knowledge of the treatment group assignments, for further examination. Tissue samples were fixed in formalin and submitted to ISU-VDL for histological and immunohistochemical examination.
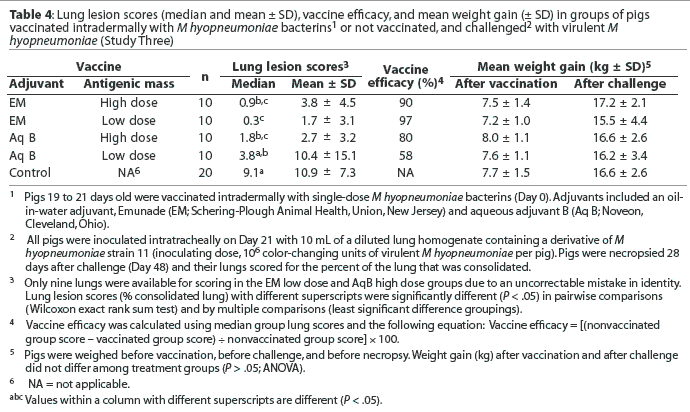
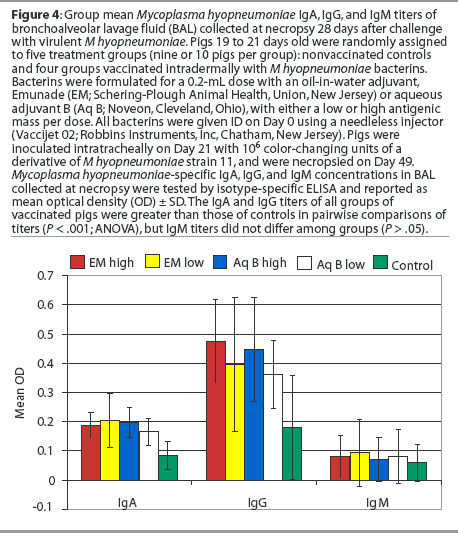
Study Three: Comparison of ID antigen dose and adjuvant by experimental challenge. Pigs 19 to 21 days of age (n = 60) were blocked by weight and litter and randomly allotted to a nonvaccinated control group and four vaccinated groups (Table 1). The adjuvants which provided the best clinical and serological responses in Study Two (oil-in-water and aqueous adjuvant B) were used to formulate bacterins with an antigenic mass equal to a predetermined low or high dose. The high dose bacterin contained 114% of the minimum protective dose for a single dose of a conventional two-dose IM bacterin, as recently established for that lot of antigen (H. Jayappa, Schering-Plough Animal Health, unpublished data, 2002). Pigs in the vaccinated groups were vaccinated once by ID inoculation on Day 0. Vaccinates and nonvaccinates were experimentally challenged on Day 21 as described in Study Two. Blood for serum was collected before vaccination (Day -1) and on Days 20 and 48. The pigs were euthanized and necropsied 28 days after challenge (Day 49), and the lungs were scored and lavaged with phosphate buffered saline to obtain bronchoalveolar lavage fluid (BAL).
Serology and analysis of bronchoalveolar lavage fluids
Sera were assayed for antibodies to M hyo-pneumoniae using the DAKO ELISA (DAKO Corporation, Carpinteria, California) according to the manufacturer's directions. Results for each pig were expressed as the ratio of the optical density (OD) of the sample to the OD of the buffer control. The percent inhibition for each pig was calculated using the following formula: Percent inhibition = 100 - [100 x (sample mean OD buffer control mean OD)]. Zero was used as the percent inhibition if the calculated value was a negative number. Seropositive was defined as a percent inhibition > 50%, and seronegative as < 35%. Percent inhibition between 35% and 50% was defined as suspect.
The M hyopneumoniae-specific IgA, IgG, and IgM concentrations in BAL collected in Study Three were measured by a previously described ELISA.12 The microtiter plates were coated with a membrane preparation of M hyopneumoniae strain 11 and the bronchoalveolar lavage fluid was tested undiluted. The isotype of M hyopneumoniae-specific antibodies was assessed using peroxidase-labeled goat anti-porcine IgA, IgG, and IgM antibodies (all heavy-chain specific). Optical density was determined at 405 nm with an ELISA plate reader.
Necropsy
Pigs were euthanized by lethal injection. The lungs were removed from the thorax and the proportion of each lobe involved in pneumonic lesions estimated visually. The estimated per cent of pneumonic tissue in each lobe was multiplied by weighting factors as previously described,13 and the % lung lesion score calculated as the sum of the calculated lobe values (Table 2).
The presence of antigens of swine influenza virus (H1N1 and H3N2) and M hyopneumoniae in lung samples collected from pigs of Study Two was evaluated by immunohistochemical examination using monoclonal and polyclonal antibodies, respectively, according to procedures established in the diagnostic laboratory.
Statistical analysis and calculations
The M hyopneumoniae serum IgG titers, reported as percent inhibition, of pigs vaccinated IM and ID with the Emunade-adjuvanted vaccines in Study One were compared by Wilcoxon exact rank sum test. The titers of pigs in Study One vaccinated ID by injector with bacterins containing Emunade and aqueous adjuvants B and C were compared by the Wilcoxon exact rank sum test. Titers of pigs vaccinated ID with vaccines containing aqueous B and C adjuvants were not compared to titers of IM-vaccinated pigs because of the difference in adjuvants. Lung consolidation scores were analyzed by Wilcoxon exact rank sum test for pairwise comparisons. Overall experimental error was controlled by performing a secondary parametric analysis of lung consolidation scores with multiple comparisons (Dunnett, Tukey, least significant difference, and Student-Newman-Keuls) using arcsine-square root transformed scores. BAL titers were analyzed by analysis of variance (ANOVA). BAL titers were also analyzed after adjustment for multiplicity (bootstrapping or permutations) to correct for study error. Statistical analysis was performed using SAS (Version 6.12; SAS Inc, Cary, North Carolina). Statistical significance was declared at a P <= .05.
Vaccine efficacy in reducing lung scores was calculated, using group median lung scores, as follows: Vaccine efficacy = [(median nonvaccinated group score - median vaccinated group score) median nonvaccinated group score] x 100.
Results
Study One
Injector testing. Preliminary testing demonstrated that the Dermo-Jet ejected 0.08 g of fluid per dose. On shaved skin, 0.07 +/- 0.01 g stayed on or in the skin, and 0.04 +/- 0.01 g penetrated the surface of the skin to a mean depth of 6.6 +/- 1.1 mm. The dye penetrated a mean of 4.8 +/- 0.7 mm into unshaven skin (P = .04, Student's t test). Hair on the skin interfered with blotting of fluid from the skin surface on unshaven skin, so an estimate of the dose that penetrated the skin was based on the ratio of the depth of penetration (mm) into shaven and unshaven skin (4.8 : 6.6). It was estimated that approximately 0.03 g of fluid per injection penetrated the unshaven skin; therefore, vaccinations with the Dermo-Jet were given as three closely spaced injections to approximate an injection of 0.1 mL.
Vaccination and histological evaluation. No injection site reaction of clinical importance occurred. The injection site reactions that were found were small and resolved within 2 weeks after the first vaccination. Small scars in the area of the injection sites were found within the thickness of the skin in four of 24 pigs vaccinated with the Dermo-Jet: three on the side of the second vaccination and one on the side of the first vaccination. The microscopic lesions were small and focal, varying from mild to moderate lymphoplasmacytic perivascular inflam-mation in the superficial and deep dermis to moderately sized granulomas within the skin. A very small focus of lymphocytes and macrophages was found in the skeletal muscle underlying the skin lesion of one pig.
Serology. All pigs were seronegative for M hyopneumoniae-specific antibodies prior to vaccination. Antibody titers for pigs vaccinated ID by needle were similar to those of pigs vaccinated by IM injection on Day 15 (P = .06), and were significantly higher on Day 22 (P = .04), Day 29 (P = .01) and Day 36 (P = .02) (Figure 1A). Titers of pigs vaccinated by ID injector with bacterins containing Emunade and aqueous C adjuvants were similar throughout the study, and on Days 29 and 36 were higher than titers of pigs vaccinated with the bacterin containing aqueous B adjuvant (P < .05; Figure 1B).
Study Two
Injector testing. The Vaccijet 02 delivered 0.18 g of Emunade-adjuvanted vaccine and 0.19 g of aqueous-adjuvanted vaccine per injection. At a pressure setting that deposited the dye primarily into the dermis and upper layers of subcutaneous tissues, approximately 0.1 mL of dye (range, 0.02 to 0.11 mL) penetrated the unshaven skin from the neck of a 4-week-old pig. The replications on pig skin were limited (six replications per pressure setting), and the skin was from a single pig; thus, the measurements should be regarded as crude estimates.
Vaccination. A palpable intradermal bleb, typical of ID injections, was produced with each vaccination. No injection site reaction of clinical importance occurred, and all reactions resolved within 1 week after the vaccination. All pigs were seronegative for M hyopneumoniae on Day 0, and the nonvaccinated pigs remained seronegative until after challenge.
Serology. The serological response in pigs of groups vaccinated twice was similar to that seen in Study One, and pigs vaccinated once with the Emunade-adjuvanted bacterin remained seronegative until after challenge (Figure 2).
Lung lesion scores and growth. Lung lesion scores were greater in the nonvaccinated control pigs than in pigs vaccinated with a single dose of the Emunade-adjuvanted vaccine (P < .01) or with two doses of vaccines with the aqueous adjuvant B (P < .01) or Emunade (P = .02, Table 3). Lung lesion scores of pigs vaccinated twice with the aqueous adjuvant C bacterin were not statistically different (P = .21) from those of the nonvaccinated pigs. When tested by multiple comparisons to control for overall experimental error, only the groups vaccinated with the Emunade-adjuvanted vaccine, either once or twice, had lesion scores significantly lower than the nonvaccinates. Vaccine efficacy, based on lung lesion scores, ranged from 67% (aqueous adjuvants) to 92% (Emunade single dose). Overall differences in weight gain during the vaccination and challenge portions of the study were not significant (Table 3).

Histological examinations and immunohistochemistry. Lung samples examined were negative for antigens of swine influenza virus, and four of five were positive for antigens of M hyopneumoniae. The microscopic lesions present, characterized by moderate to severe multifocal suppurative and histiocytic bronchopneumonia with moderate to severe peribronchiolar lymphoid hyperplasia, were consistent with pneumonia caused by M hyopneumoniae.
Study Three
Vaccinations. A bleb typical of intradermal injections could be palpated after vaccination in most of the injected pigs, although substantial amounts of the bacterins appeared to remain on the surface of the skin. Injection site reactions of clinical importance did not occur.
Serology. All pigs were seronegative for M hyopneumoniae on Day -1. One of the 20 pigs vaccinated with the aqueous adjuvant B vaccine, and none of the 20 pigs vaccinated with Emunade-adjuvanted vaccines, had seroconverted by 20 days after vaccination. All vaccinated pigs and eight of 20 nonvaccinated pigs had seroconverted by Day 48, 28 days after challenge (Figure 3).
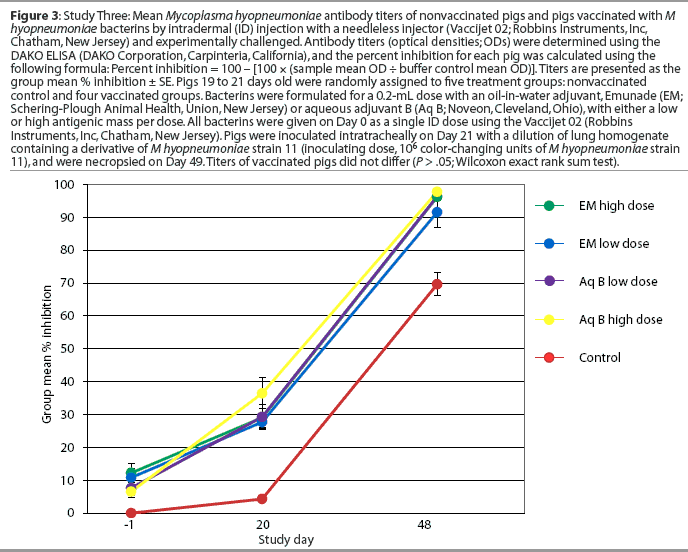
Lung lesion scores and growth. Lung lesion scores ranged from 0% to 41%. Lung lesion scores of the nonvaccinated control pigs were significantly greater than those of pigs vaccinated ID with the Emunade-adjuvanted high (P < .01) and low (P < .001) antigen dose bacterins and the high antigen dose aqueous B adjuvanted bacterin (P < .001; Table 4). The differences in lung scores remained significant when analyzed by multiple comparisons analysis to control for overall experimental error. The difference between the lung lesion scores of pigs vaccinated with the low-antigen dose, aqueous-adjuvanted bacterin and the nonvaccinates approached significance (P < .10). Pigs vaccinated with the low-antigen dose, Emunade-adjuvanted bacterin had significantly lower lung lesion scores than did those vaccinated with the low-antigen dose, aqueous B-adjuvanted bacterin (P = .01). Vaccine efficacy ranged from 58% to 97%. Overall differences in weight gain during the vaccination and challenge periods of the study were not significant (Table 4).
Bronchoalveolar lavage fluids. The concentrations of IgA and IgG antibodies (P < .001), but not IgM antibodies (P > .05), in the BAL from all groups of vaccinated pigs were significantly greater than those of nonvaccinated pigs in pairwise comparisons of titers (Figure 4). The results were the same for IgA and IgM when the data was adjusted for multiplicity (bootstrap or permutations) to correct for study error. The IgG titers of the pigs vaccinated with the Emunade adjuvanted vaccines or the high-antigen-dose, aqueous-adjuvanted vaccine remained significantly greater than those of the nonvaccinates when the data were adjusted for multiplicity (P = .001, Emunade high dose; P = .045, Emunade low dose; P = .005, aqueous B high dose). The IgG titers of pigs vaccinated with the aqueous adjuvant, low-antigen-dose bacterin were not significantly greater than those of the nonvaccinates (P = .18) when similarly analyzed.
Discussion
The results of Study One demonstrated that a needleless injector could deliver an ID vaccination in 13-week-old pigs without special preparation of the skin surface. Three injections were required to inoculate approximately 0.1 mL of the bacterins ID using the Dermo-Jet, as a substantial amount of the ejected volume did not penetrate the surface of the skin. Three injections per dose was acceptable for an experimental study, but would not be acceptable in clinical practice. Intradermal vaccination by use of a syringe and needle, which allowed control of the amount and site of deposition of the Emunade vaccine, was included to prevent potential confounding of the analysis, as the volume and depth of delivery of the Dermo-Jet vaccinations were variable and the number of injection sites differed. The humoral immune response to the Emunade-adjuvanted bacterin was greater in pigs vaccinated ID by needle or Dermo-Jet than in those vaccinated by IM injection. Histological examination of small scars found 3 weeks after the last vaccinations in four of 24 pigs vaccinated by Dermo-Jet supported our impression that the injections given by the needleless injection device had been predominantly into superficial layers of the dermis and subcutis. Use of the Vaccijet 02 allowed vaccination with a nominal 0.2-mL dose and adjustment of injection pressure in Studies Two and Three. The pressure required to inject into the skin of 4-week-old pigs was less than that required for older pigs, and a greater proportion of the vaccine appeared to penetrate the skin, probably due to the thick skin and coarse hair normal in older pigs. Clinically significant injection-site reactions were not observed in pigs vaccinated by any of the injection routes or vaccine formulations used in the three studies.
All vaccines given in a two-dose regimen elicited significant and similar IgG titers in 4-week-old pigs in Study Two, but lung lesions of pigs vaccinated with aqueous adjuvant C did not differ significantly from those of nonvaccinated controls following virulent challenge. A single dose of the Emunade-adjuvanted vaccine did not elicit a serological response until after challenge, but the immune response was protective. The antigenic mass per dose was greater than a minimum protective dose established for that antigen lot when administered in two IM doses, but the antigenic mass expected to penetrate the skin was slightly less. This was further supported by the results of Study Three, where pigs vaccinated with a single dose of an Emunade-adjuvanted vaccine, with antigen dose equal to the minimum protective dose of a two-dose IM bacterin, were protected against experimental challenge within 21 days of vaccination. As in earlier studies,3,14 antibody titers did not correlate with protection against infection with M hyopneumonia in this study. However, the significantly higher antibody levels in the ID-vaccinated pigs, compared to the IM-vaccinated pigs, together with the solid protection against challenge in the single-dose ID-vaccinated pigs, demonstrates that ID vaccination for M hyopneumoniae is feasible and protective.
Significant differences in weight gain between treatment groups were not found in either Study Two or Three. Similar results have been reported by others using this challenge model in IM-vaccination studies conducted with pigs with other respiratory pathogens, and under conditions of adequate housing and management.3,8
It has been postulated that a local mucosal effect and cell-mediated response may be necessary components of protective immunity to M hyopneumoniae.8,9 Measures of cell-mediated immunity were not examined in this study, but IM vaccination of pigs with killed M hyopneumoniae bacterins can induce interferon-gamma secretion by lymphocytes, and appears to reduce secretion of tumor necrosis factor-[alpha] following virulent challenge.9 Resistance to tracheal infection by M galisepticum in chickens has been associated with postvaccination tracheal IgG titers, but not IgA or IgM titers.15 The level of IgG in BAL fluid was the only immune measurement that differentiated between the responses elicited by high (97%) and low (57%) efficacy M hyopneumoniae bacterins in this study, suggesting a similar protective response in pigs. The protection afforded by M hyopneumoniae bacterins that failed to elicit a significant serological response to vaccination could be explained if either cell-mediated immunity or IgG in tracheal or lung secretions were important factors in resistance of swine to M hyopneumoniae.
The results of these studies indicate that the use of needleless ID injectors can be effective in administering vaccines to pigs, which may decrease spread of other economically important swine pathogens such as porcine reproductive and respiratory syndrome virus or porcine circovirus type 2. Further studies investigating the use of these technologies are needed to determine the optimal vaccination strategies for use of needleless injectors in the swine industry.
Implications
- Intradermal vaccination of pigs for M hyopneumoniae is feasible and protective.
- Intradermal vaccination of swine with needleless injectors is effective using vaccines formulated for small volumes (0.2 mL).
- Intradermal M hyopneumoniae vaccination, with penetration of the surface of the skin by a relatively small antigenic mass, was protective against experimental challenge with virulent M hyopneumoniae bacteria.
- Needless injectors used in these studies often left substantial amounts of the vaccine on the surface of the skin, which might be unavoidable and might delay acceptance of the technology.
- The suitability of the currently available needleless injectors for use in commercial swine operations was beyond the scope of these studies.
References
*1. Halbur PG. Porcine respiratory diseases. Proc IPVS. Birmingham, UK. 1998:1-10.
2. Maes D, Deluyker H, Verdonck M, Castyck F, Miry C, Vrijens B, Verbeke W, Viaene J, de Kruif A. Effect of vaccination against Mycoplasma hyopneu-moniae in pig herds with an all-in/all-out production system. Vaccine. 1999;17:1024-1034.
3. Thacker EL, Thacker BJ, Boettcher TB, Jayappa H. Comparison of antibody production, lymphocyte stimulation, and protection induced by four commercial Mycoplasma hyopneumoniae bacterins. Swine Health Prod. 1998;6:107-112.
4. Otake S, Dee SA, Rossow KD, Moon RD, Pijoan C. Transmission of porcine reproductive and respiratory syndrome by needles. Vet Rec. 2002;150:114-115.
*5. Stier RF. The dirty dozen: Ways to reduce the 12 biggest foreign materials problems. Food Safety. 2003;9:44-50.
6. Schrijver RS, Langedijk JPM, Keil GM, Middel WGJ, Maris-Veldhuis M, Van Oirschot JT, Rijesewijk FAM. Comparison of DNA application methods to reduce BRSV shedding in cattle. Vaccine. 1998;16:130-134.
7. Dègano P, Sarphie DF, Banghams CRM. Intradermal DNA immunization of mice against influenza A virus using the novel PowderJect" system. Vaccine. 1998;16:394-398.
8. Boettcher TB, Thacker BJ, Halbur PG, Waters WR, Nutsch R, Thacker EL. Vaccine efficacy and immune response to Mycoplasma hyopneumoniae challenge in pigs vaccinated against porcine reproductive and respiratory syndrome virus and M hyopneumoniae. Swine Health Prod. 2002;10:259-264.
9. Thacker EL, Thacker BJ, Kuhn M, Hawkins PA, Waters WR. Evaluation of local and systemic immune responses induced by intramuscular injection of Mycoplasma hyopneumoniae bacterin to pigs. Am J Vet Res. 2000;61:1384-1389.
10. Enioutina EY, Visic D, Daynes RA. The induction of systemic and mucosal immune responses to antigen-adjuvant compositions administered into the skin: alterations in the migratory properties of dendritic cells appears to be important for stimulating mucosal immunity. Vaccine. 2000;18:2753-2767.
11. Bendelac A, Medzhitov R. Adjuvants of immunity: harnessing innate immunity to promote adaptive immunity. J Exp Med. 2002;195:19-23.
12. Cutler CW, Jotwani R, Pulendran B. Dendritic cells: immune saviors or Achilles' heel? Infect Immun. 2001;69:4703-4708.
13. Christensen G, Sorensen V, Mousing J. Diseases of the respiratory system. In: Straw B, D'Allaire S, Mengeling W, Taylor DJ, eds. Diseases of Swine. 8th ed. Ames, Iowa: Iowa University Press; 1999:913-940.
14. Djordevic SP, Eamens GJ, Romalis LF, Nichols PJ, Taylor V, Chin J. Serum and mucosal antibody responses and protection in pigs vaccinated against Mycoplasma hyopneumoniae with vaccines containing a denatured membrane antigen pool and adjuvant. Aust Vet J. 1997;75:504-511.
15. Papazisi L, Silbart LK, Frasca S Jr, Rood D, Liao X, Gladd M, Javed MA, Geary SJ. A modified live Mycoplasma gallisepticum vaccine to protect chickens from respiratory disease. Vaccine. 2002;20:3709-3719.
* Non-refereed references.
